Abstract
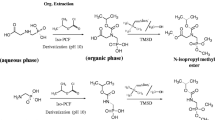
The interest in compound-specific isotope analysis for product authenticity control and source differentiation in environmental sciences has grown rapidly during the last decade. However, the isotopic analysis of very polar analytes is a challenging task due to the lack of suitable chromatographic separation techniques which can be used coupled to isotope ratio mass spectrometry. In this work, we present the first method to measure carbon isotope compositions of the widely applied herbicide glyphosate and its metabolite aminomethylphosphonic acid (AMPA) by liquid chromatography coupled to isotope ratio mass spectrometry. We demonstrate that this analysis can be carried out either in cation exchange or in reversed-phase separation modes. The reversed-phase separation yields a better performance in terms of resolution compared with the cation exchange method. The measurement of commercial glyphosate herbicide samples show its principal applicability and reveals a wide range of δ13C values between −24 and −34 ‰ for different manufacturers. The absolute minimum amounts required to perform a precise and accurate determination of carbon isotope compositions of glyphosate and AMPA were in the sub-microgram range. The method proposed is sensitive enough to further perform the experiments that are necessary to better understand the carbon isotope fractionation associated to the natural degradation of glyphosate into AMPA. Furthermore, it can be used for contaminant source allocation and product authenticity as well.






Similar content being viewed by others
References
Vereecken H (2005) Mobility and leaching of glyphosate: a review. Pest Management Science 61(12):1139–1151. doi:10.1002/ps.1122
Coupe RH, Kalkhoff SJ, Capel PD, Gregoire C (2011) Fate and transport of glyphosate and aminomethylphosphonic acid in surface waters of agricultural basins. Pest Management Science 68(1):16–30. doi:10.1002/ps.2212
Ermakova IT, Shushkova TV, Leont'evskii AA (2008) Microbial degradation of organophosphonates by soil bacteria. Microbiology 77(5):615–620. doi:10.1134/s0026261708050160
Borggaard OK, Gimsing AL (2008) Fate of glyphosate in soil and the possibility of leaching to ground and surface waters: a review. Pest Management Science 64(4):441–456. doi:10.1002/ps.1512
Nowack B (2003) Environmental chemistry of phosphonates. Water Res 37(11):2533–2546. doi:10.1016/s0043-1354(03)00079-4
Singh BK, Walker A (2006) Microbial degradation of organophosphorus compounds. FEMS Microbiol Rev 30(3):428–471. doi:10.1111/j.1574-6976.2006.00018.x
Schmidt TC, Zwank L, Elsner M, Berg M, Meckenstock RU, Haderlein SB (2004) Compound-specific stable isotope analysis of organic contaminants in natural environments: a critical review of the state of the art, prospects, and future challenges. Anal Bioanal Chem 378(2):283–300. doi:10.1007/s00216-003-2350-y
Elsner M, Jochmann MA, Hofstetter TB, Hunkeler D, Bernstein A, Schmidt TC, Schimmelmann A (2012) Current challenges in compound-specific stable isotope analysis of environmental organic contaminants. Anal Bioanal Chem 403(9):2471–2491. doi:10.1007/s00216-011-5683-y
Elsner M (2010) Stable isotope fractionation to investigate natural transformation mechanisms of organic contaminants: principles, prospects and limitations. J Environ Monit 12(11):2005–2031. doi:10.1039/c0em00277a
Kujawinski DM, Stephan M, Jochmann MA, Krajenke K, Haas J, Schmidt TC (2010) Stable carbon and hydrogen isotope analysis of methyl tert-butyl ether and tert-amyl methyl ether by purge and trap-gas chromatography-isotope ratio mass spectrometry: method evaluation and application. J Environ Monit 12(1):347–354. doi:10.1039/b914514a
Stalikas CD, Konidari CN (2001) Analytical methods to determine phosphonic and amino acid group-containing pesticides. Journal of Chromatography A 907(1–2):1–19. doi:10.1016/s0021-9673(00)01009-8
Reinnicke S, Bernstein A, Elsner M (2010) Small and reproducible isotope effects during methylation with trimethylsulfonium hydroxide (TMSH): a convenient derivatization method for isotope. Anal Chem 82(5):2013–2019. doi:10.1021/ac902750s
Corr LT, Berstan R, Evershed RP (2007) Optimisation of derivatisation procedures for the determination of delta C-13 values of amino acids by gas chromatography/combustion/isotope ratio mass spectrometry. Rapid Communications in Mass Spectrometry 21(23):3759–3771. doi:10.1002/rcm.3252
Krummen M, Hilkert AW, Juchelka D, Duhr A, Schluter HJ, Pesch R (2004) A new concept for isotope ratio monitoring liquid chromatography/mass spectrometry. Rapid Communications in Mass Spectrometry 18(19):2260–2266
Dunn PJH, Honch NV, Evershed RP (2011) Comparison of liquid chromatography-isotope ratio mass spectrometry (LC/IRMS) and gas chromatography-combustion-isotope ratio mass spectrometry (GC/C/IRMS) for the determination of collagen amino acid delta(13) C values for palaeodietary and palaeoecological reconstruction. Rapid Communications in Mass Spectrometry 25(20):2995–3011. doi:10.1002/rcm.5174
Godin J-P, McCullagh JSO (2011) Review: current applications and challenges for liquid chromatography coupled to isotope ratio mass spectrometry (LC/IRMS). Rapid communications in mass spectrometry: RCM 25(20):3019–3028. doi:10.1002/rcm.5167
Zhang L, Kujawinski DM, Jochmann MA, Schmidt TC (2011) High-temperature reversed-phase liquid chromatography coupled to isotope ratio mass spectrometry. Rapid Communications in Mass Spectrometry 25(20):2971–2981. doi:10.1002/rcm.5069
Heuer V, Elvert M, Tille S, Krummen M, Mollar XP, Hmelo LR, Hinrichs KU (2006) Online delta C-13 analysis of volatile fatty acids in sediment/porewater systems by liquid chromatography-isotope ratio mass spectrometry. Limnology and Oceanography-Methods 4:346–357
McCullagh JSO (2010) Mixed-mode chromatography/isotope ratio mass spectrometry. Rapid Communications in Mass Spectrometry 24(5):483–494. doi:10.1002/rcm.4322
Zhang L, Kujawinski DM, Federherr E, Schmidt TC, Jochmann MA (2012) Caffeine in your drink: natural or synthetic? Anal Chem 84(6):2805–2810. doi:10.1021/ac203197d
Winfield TW, Bashe WJ, Baker TV (1990) Determination of glyphosate in drinking water by direct-aqueous-injection HPLC, post-column derivatisation, and flourescence detection. Environmental Monitoring Systems, Labratory Office of Research and Development, U.S. Environmental Protection Agency, Cincinnati, Ohio
Werner RA, Brand WA (2001) Referencing strategies and techniques in stable isotope ratio analysis. Rapid Communications in Mass Spectrometry 15(7):501–519. doi:10.1002/rcm.258
Elsig J, Leuenberger MC (2010) C-13 and O-18 fractionation effects on open splits and on the ion source in continuous flow isotope ratio mass spectrometry. Rapid Communications in Mass Spectrometry 24(10):1419–1430. doi:10.1002/rcm.4531
Piez KA, Eagle H (1956) C-14 isotope effect on the ion-exchange chromatography of amino acids. J Am Chem Soc 78(20):5284–5287. doi:10.1021/ja01601a035
Caimi RJ, Brenna JT (1997) Quantitative evaluation of carbon isotopic fractionation during reversed-phase high-performance liquid chromatography. Journal of Chromatography A 757(1–2):307–310. doi:10.1016/s0021-9673(96)00694-2
Meier-Augenstein W (1999) Applied gas chromatography coupled to isotope ratio mass spectrometry. Journal of Chromatography A 842(1–2):351–371. doi:10.1016/s0021-9673(98)01057-7
Voskamp M (2005) Untersuchungen zur Sorption an Huminstoffen: Molekulargewicht und Kohlenstoff-Isotopenfraktionierung. University of Leipzig, Leipzig
Ricci MP, Merritt DA, Freeman KH, Hayes JM (1994) Acquisition and processing of data for isotope-ratio-monitoring mass-spectrometry. Org Geochem 21(6–7):561–571. doi:10.1016/0146-6380(94)90002-7
Jochmann MA, Blessing M, Haderlein SB, Schmidt TC (2006) A new approach to determine method detection limits for compound-specific isotope analysis of volatile organic compounds. Rapid Communications in Mass Spectrometry 20(24):3639–3648. doi:10.1002/rcm.2784
Kujawinski DM, Zhang L, Schmidt TC, Jochmann MA (2012) When other separation techniques fail: compound-specific carbon isotope ratio analysis of sulfonamide containing pharmaceuticals by high-temperature-liquid chromatography-isotope ratio mass spectrometry. Anal Chem 84(18):7656–7663. doi:10.1021/ac300116w
Dill GM, Sammons RD, Feng PCC, Kohn F, Kretzmer K, Mehrsheikh A, Bleeke M, Honegger JL, Farmer D, Wright D, Haupfear EA (2010) Glyphosate: discovery, development, applications, and properties. In: Nandula VK (ed) Glyphosate resistance in crops and weeds. John Wiley & Sons, Inc., pp 1–33. doi:10.1002/9780470634394.ch1
Chamberlain K, Evans AA, Bromilow RH (1996) 1-octanol/water partition coefficient (K-ow) and pK(a) for ionisable pesticides measured by a pH-metric method. Pestic Sci 47(3):265–271. doi:10.1002/(sici)1096-9063(199607)47:3<265::aid-ps416>3.0.co;2-f
Acknowledgments
The study was partly funded by ONEMA (The French National Agency for Water and Aquatic Environments) and BRGM. Additionally, we acknowledge financial support from German Research Foundation (DFG).
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in the topical collection Isotope Ratio Measurements: New Developments and Applications with guest editors Klaus G. Heumann and Torsten C. Schmidt.
Rights and permissions
About this article
Cite this article
Kujawinski, D.M., Wolbert, J.B., Zhang, L. et al. Carbon isotope ratio measurements of glyphosate and AMPA by liquid chromatography coupled to isotope ratio mass spectrometry. Anal Bioanal Chem 405, 2869–2878 (2013). https://doi.org/10.1007/s00216-012-6669-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-012-6669-0