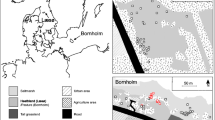
Abstract
Polygyny is common in social insects despite inevitable decreases in nestmate relatedness and reductions to the inclusive fitness returns for cooperating non-reproductive individuals. We studied the prevalence and mode of polygyny in the African acacia-ant Crematogaster mimosae. These ants compete intensively with neighboring colonies of conspecifics and with three sympatric ant species for resources associated with the whistling-thorn acacias in which they all obligately nest. We used the genotypes of alate males at ten microsatellite loci to reconstruct queen genotypes and found that C. mimosae colonies are frequently secondarily polygynous, in that they include multiple closely related (and sometimes full-sib) queens, and (more rarely) unrelated queens. We also found that individual queens in both monogynous and polygynous colonies had mated with multiple males, making C. mimosae an interesting example of simultaneous polygyny and polyandry. The presence of polygyny in C. mimosae and the intense competition for nest-sites between C. mimosae and its conspecifics support the association between nest-site limitation and polygyny. Polygyny may allow for increased worker populations and a competitive advantage, as inter-colony conflicts are typically won by the colony with the larger number of workers.



Similar content being viewed by others
References
AntWeb [http://www.antweb.org] 2013. Retreived 14 January 2013
Bernasconi G. and Strassmann J.E. 1999. Cooperation among unrelated individuals: the ant foundress case. Trends Ecol. Evol. 14: 477-482
Beye M., Neumann P., Chapuisat M., Pamilo P. and Moritz R.F.A. 1998. Nestmate recognition and the genetic relatedness of nests in the ant Formica pratensis. Behav. Ecol. Sociobiol. 43: 67-72
Blaimer B.B. 2012a. A subgeneric revision of Crematogaster and discussion of regional species-groups (Hymenoptera: Formicidae). Zootaxa 3482: 47-67
Blaimer B.B. 2012b. Acrobat ants go global - Origin, evolution and systematics of the genus Crematogaster (Hymenoptera: Formicidae). Mol. Phylogenet. Evol. 65: 421-436
Bourke A.F.G. 1988. Worker reproduction in the higher eusocial Hymenoptera. Q. Rev. Biol. 63: 291-311
Bourke A.F.G. and Franks N.R. 1995. Social Evolution in Ants. Princeton University Press, Princeton
Brownstein M.J., Carpten J.D. and Smith J.R. 1996. Modulation of non-templated nucleotide addition by Taq DNA polymerase: primer modifications that facilitate genotyping. BioTechniques 20: 1004-1010
Chapuisat M., Goudet J. and Keller L. 1997. Microsatellites reveal high population viscosity and limited dispersal in the ant Formica paralugubris. Evolution 51: 475-482
Crozier R.H. and Pamilo P. 1996. Evolution of Social Insect Colonies: Sex Allocation and Kin Selection. Oxford University Press, Oxford
Dalecky A., Gaume L., Schatz B., McKey D. and Kjellberg F. 2005. Facultative polygyny in the plant-ant Petalomyrmex phylax (Hymenoptera : Formicinae): sociogenetic and ecological determinants of queen number. Biol. J. Linn. Soc. 86: 133-151
Debout G., Schatz B., Elias M. and McKey D. 2007. Polydomy in ants: what we know, what we think we know, and what remains to be done. Biol. J. Linn. Soc. 90: 319-348
Feldhaar H., Fiala B. and Gadau J. 2005. A shift in colony founding behaviour in the obligate plant-ant Crematogaster (Decacrema) morphospecies 2. Insect. Soc. 52: 222-230
Feldhaar H., Fiala B., bin Hashim R. and Maschwitz U. 2000. Maintaining an ant-plant symbiosis: secondary polygyny in the Macaranga triloba-Crematogaster sp. association. Naturwissenschaften 87: 408-411
Feldhaar H., Fiala B., Gadau J., Mohamed M. and Maschwitz U. 2003. Molecular phylogeny of Crematogaster subgenus Decacrema ants (Hymenoptera: Formicidae) and the colonization of Macaranga (Euphorbiaceae) trees. Mol. Phylogenet. Evol. 27: 441-452
Fournier D., Keller L., Passera L. and Aron S. 2003. Colony sex ratios vary with breeding system but not relatedness asymmetry in the facultatively polygynous ant Pheidole pallidula. Evolution 57: 1336-1342
Hamilton W.D. 1964. The genetical evolution of social behaviour I, II. J. Theor. Biol. 7: 1-52
Heinze J., Strätz M., Pederson J.S. and Haberl M. 2000. Microsatellite analysis suggests occasional worker reproduction in the monogynous ant Crematogaster smithi. Insect. Soc. 47: 299-301
Heinze J., Trunzer B., Hölldobler B. and Delabie J.H.C. 2001. Reproductive skew and queen relatedness in an ant with primary polygyny. Insect. Soc. 48: 149-153
Herbers J.M. 1986. Nest site limitation and facultative polygyny in the ant Leptothorax longispinosus. Behav. Ecol. Sociobiol. 19: 115-122
Herbers J.M. 1993. Ecological determinants of queen number in ants. In: Queen Number and Sociality in Insects (Keller L., Ed), Oxford University Press, New York, pp 262-293
Hölldobler B. and Wilson E.O. 1977. The number of queens: An important trait in ant evolution. Naturwissenschaften 64: 8-15
Hughes W.O.H. and Boomsma J.J. 2004. Genetic diversity and disease resistance in leaf-cutting ant societies. Evolution 58: 1251-1260
Hughes W.O.H. and Boomsma J.J. 2006. Does genetic diversity hinder parasite evolution in social insect colonies? J. Evol. Biol. 19: 132-143
Hughes W.O.H., Ratnieks F.L.W. and Oldroyd B.P. 2008. Multiple paternity or multiple queens: two routes to greater intracolonial genetic diversity in the eusocial Hymenoptera. J. Evol. Biol. 21: 1090-1095
Janzen D.H. 1973. Evolution of polygynous obligate acacia-ants in Western Mexico. J. Anim. Ecol. 42: 727-750
Jones O.R. and Wang J. 2010. colony: a program for parentage and sibship inference from multilocus genotype data. Mol. Ecol. Res. 10: 551-555
Kautz S., Ballhorn D.J., Kroiss J., Pauls S.U., Moreau C.S., Eilmus S., Strohm E. and Heil M. 2012. Host plant use by competing acacia-ants: mutualists monopolize while parasites share hosts. PLoS ONE 7: e37691
Kautz S., Pauls S.U., Ballhorn D.J., Lumbsch H.T. and Heil M. 2009. Polygynous supercolonies of the acacia-ant Pseudomyrmex peperi, an inferior colony founder. Mol. Ecol. 18: 5180-5194
Keller L. 1995. Social-life - The paradox of multiple queen colonies. Trends Ecol. Evol. 10: 355-360
Keller L. and Reeve H.K. 1994. Genetic variability, queen number and polyandry in social Hymenoptera. Evolution 48: 694-704
Kellner K., Trindl A., Heinze J. and D’Ettorre P. 2007. Polygyny and polyandry in small ant societies. Mol. Ecol. 16: 2363-2369
Komene Y., Higashi S., Ito F. and Miyata H. 1999. Effect of colony size on the number of gamergates in the queenless ponerine ant Rhytidoponera aurata. Insect. Soc. 46: 29-33
Konovalov D.A., Manning C. and Henshaw M.T. 2004. Kingroup: a program for pedigree relationship reconstruction and kin group assignments using genetic markers. Mol. Ecol. Notes 4: 779-782
Kronauer D.J.C. and Boomsma J.J. 2007. Multiple queens means fewer mates. Curr. Biol. 17: R753-R755
Kümmerli R. and Keller L. 2007. Reproductive specialization in multiple-queen colonies of the ant Formica exsecta. Behav. Ecol. 18: 375-383
Longino J.T. 1991. Taxonomy of the Cecropia-inhabiting Azteca ants. J. Nat. Hist. 25: 1571-1602
Manly B.F.J. 1991. Randomization and Monte Carlo Methods in Biology. Chapman and Hall, New York
Nonacs P. 1988. Queen number in colonies of social Hymenoptera as kin-selected adaptation. Evolution 42: 566-580
Palmer T.M. 2003. Spatial habitat heterogeneity influences competition and coexistence in an African acacia ant guild. Ecology 84: 2843-2855
Palmer T.M. 2004. Wars of attrition: colony size determines competitive outcomes in a guild of African acacia ants. Anim. Behav. 68: 993-1004
Palmer T.M., Stanton M.L., Young T.P., Goheen J.R., Pringle R.M. and Karban R. 2008. Breakdown of an ant-plant mutualism follows the loss of large herbivores from an African savanna. Science 319: 192-195
Palmer T.M., Young T.P. and Stanton M.L. 2002. Burning bridges: priority effects and the persistence of a competitively subordinate acacia-ant in Laikipia, Kenya. Oecologia 133: 372-379
Palmer T.M., Young T.P., Stanton M.L. and Wenk E. 2000. Short-term dynamics of an acacia ant community in Laikipia, Kenya. Oecologia 123: 425-435
Pamilo P. 1993. Polyandry and allele frequency differences between the sexes in the ant Formica aquilonia. Heredity 70: 472-480
Pirk C.W.W., Neumann P., Moritz R.F.A. and Pamilo P. 2001. Intranest relatedness and nestmate recognition in the meadow ant Formica pratensis (R.). Behav. Ecol. Sociobiol. 49: 366-374
Pedersen J.S. and Boomsma J.J. 1999a. Effect of habitat saturation on the number and turnover of queens in the polygynous ant, Myrmica sulcinodis. J. Evol. Biol. 12: 903-917
Pedersen J.S. and Boomsma J.J. 1999b. Positive association of queen number and queen-mating frequency in Myrmica ants: a challenge to the genetic-variability hypotheses. Behav. Ecol. Sociobiol. 45: 185-193
Puntilla P. 1996. Succession, forest fragmentation, and the distribution of wood ants. Oikos 75: 281-298
Queller D.C. and Goodnight K.F. 1989. Estimating relatedness using genetic markers. Evolution 43: 258-275
Quinet Y., Hamidi R., Ruiz-Gonzalez M.X., de Biseau J.-C. and Longino J.T. 2009. Crematogaster pygmaea (Hymenoptera: Formicidae: Myrmicinae), a highly polygynous and polydomous Crematogaster from northeastern Brazil. Zootaxa 2075: 45-54
Raymond M. and Rousset F. 2004. GenePop Version 3.4. http://wbiomed.curtin.edu.au/genepop/. Updated from Raymond and Rousset (1995) GenePop version 1.2: population genetics software for exact tests and ecumenicism. J. Hered. 83: 233-234
Rissing S.W. and Pollock G.B. 1987. Queen aggression, pleometrotic advantage and brood raiding in the ant Veromessor pergandei (Hymenoptera: Formicidae). Anim. Behav. 35: 975-981
Ross K.G. 1988. Differential reproduction in multiple-queen colonies of the fire ant Solenopsis invicta (Hymenoptera: Formicidae). Behav. Ecol. Sociobiol. 23: 341-355
Rubin B.E., Makarewich C.A., Talaba A.L., Stenzler L., Bogdanowicz S.M. and Lovette I.J. 2009. Isolation and characterization of microsatellite markers from the acacia-ant Crematogaster mimosae. Mol. Ecol. Res. 9: 1212-1214
Schmid-Hempel P. and Crozier R.H. 1999. Polyandry versus polygyny versus parasites. Philos. Trans. R. Soc. B 354: 507-515
Seppä P., Sundström L. and Punttila P. 1995. Facultative polygyny and habitat succession in boreal ants. Biol. J. Linn. Soc. 56: 533-551
Sommer K. and Hölldobler B. 1995. Colony founding by queen association and determinants of reduction in queen number in the ant Lasius niger. Anim. Behav. 50: 287-294
Stanton M.L., Palmer T.M. and Young T.P. 2002. Competition-colonization trade-offs in a guild of African acacia-ants. Ecol. Monogr. 72: 347-363
Stanton M.L., Palmer T.M., Young T.P., Turner M.L. and Evans A. 1999. Sterilization and canopy modification of a swollen thorn acacia tree by a plant-ant. Nature 401: 578-581
Sundström L. 1997. Queen acceptance and nestmate recognition in monogyne and polygyne colonies of the ant Formica truncorum. Anim. Behav. 53: 499-510
Trontti K., Thurin N., Sundström L. and Aron S. 2007. Mating for convenience or genetic diversity? Mating patterns in the polygynous ant Plagiolepis pygmaea. Behav. Ecol. 18: 298-303
Wiernasz D.C., Hines J., Parker D.G. and Cole B.J. 2008. Mating for variety increases foraging activity in the harvester ant, Pogonomyrmex occidentalis. Mol. Ecol. 17: 1137-1144
Wiernasz D.C., Perronia C.L. and Cole B.J. 2004. Polyandry and fitness in the western harvester ant, Pogonomyrmex occidentalis. Mol. Ecol. 13: 1601-1606
Young T.P., Stubblefield C.H. and Isbell L.A. 1997. Ants on swollen thorn acacias: species coexistence in a simple system. Oecologia 109: 98-107
Acknowledgments
We thank Amanda Talaba, John Lemboi, and Rafael Eraguy for collecting ants. The staff and administration at Mpala Research Centre provided excellent logistical support. Chris Makarewich, Amanda Talaba, Doug Morin and Laura Stenzler provided assistance and consultation in the lab. We thank Stefanie Kautz, Corrie Moreau, Max Winston, Wes Hochachka and two anonymous reviewers for suggestions on previous versions of the manuscript. This work was supported by the Explorer’s Club, the Bartels Scholars fund, the Cornell Lab of Ornithology, the Hughes Scholars program and by National Science Foundation grants DEB-0089706 and DEB-0444741 to T.M.P. and M.L.S. This research was carried out under the auspices of the Ministry of Education, Science, and Technology of the Republic of Kenya (Permit number MOEST 13/001/34 17).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rubin, B.E.R., Anderson, R.M., Kennedy, D. et al. Polygyny in the nest-site limited acacia-ant Crematogaster mimosae . Insect. Soc. 60, 231–241 (2013). https://doi.org/10.1007/s00040-013-0287-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00040-013-0287-5