Abstract.
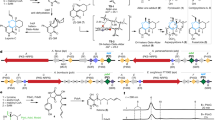
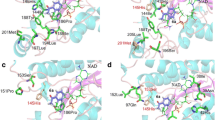
Structural and mechanistic studies on the crotonase superfamily (CS) are reviewed with the aim of illustrating how a conserved structural platform can enable catalysis of a very wide range of reactions. Many CS reactions have precedent in the ‘carbonyl’ chemistry of organic synthesis; they include alkene hydration/isomerization, aryl-halide dehalogenation, (de)carboxylation, CoA ester and peptide hydrolysis, fragmentation of β-diketones and C-C bond formation, cleavage and oxidation. CS enzymes possess a canonical fold formed from repeated ββα units that assemble into two approximately perpendicular β-sheets surrounded by α-helices. CS enzymes often, although not exclusively, oligomerize as trimers or dimers of trimers. Two conserved backbone NH groups in CS active sites form an oxyanion ‘hole’ that can stabilize enolate/oxyanion intermediates. The range and efficiency of known CS-catalyzed reactions coupled to their common structural platforms suggest that CS variants may have widespread utility in biocatalysis.
Similar content being viewed by others
Author information
Authors and Affiliations
Corresponding authors
Additional information
R. B. Hamed, E. T. Batchelar: These authors contributed equally to this work.
Received 18 January 2008; received after revision 25 March 2008; accepted 1 April 2008
Rights and permissions
About this article
Cite this article
Hamed, R.B., Batchelar, E.T., Clifton, I.J. et al. Mechanisms and structures of crotonase superfamily enzymes – How nature controls enolate and oxyanion reactivity. Cell. Mol. Life Sci. 65, 2507–2527 (2008). https://doi.org/10.1007/s00018-008-8082-6
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-008-8082-6