Abstract
Aims and objective
Mast cells play a central role in allergic and chronic inflammation. Extracts from Clausena lansium (Lour.) Skeels (Rutaceae) possess many pharmacological effects including anti-inflammatory, anti-oxidant, anti-cancer, and anti-trichomonal activities. In addition, the leaves and fruit are used in Chinese folk medicine. We have isolated and identified four known cinnamamides from this plant: lansiumamide C, lansamide I, lansiumamide B, and SB-204900. However, the biological activities of these compounds are not yet understood. The purpose of this paper is to clarify the pharmacological effects of these compounds on mast cells.
Methods
We measured inflammatory molecules in A23187-stimulated rat basophilic leukemia cells (RBL-2H3) treated with these compounds using HPLC, ELISA, and immunoblotting methods. In addition, some signaling molecules were investigated by immunoblotting.
Results
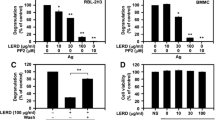
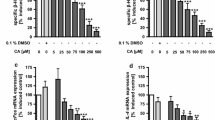
Lansamide I, lansiumamide B, and SB-204900 significantly decreased histamine release. Furthermore, lansiumamide B- and SB-204900-treated cells also reduced the protein and/or mRNA levels of TNF-α. SB-204900 markedly suppressed the phosphorylation of p38 MAPK.
Conclusion
Our findings suggest that lansiumamide B and SB-204900 attenuate mast-cell-induced inflammation.





Similar content being viewed by others
References
Metcalfe DD. Mast cells and mastocytosis. Blood. 2008;112:946–56.
Kawakami T, Ando T, Kimura M, Wilson BS, Kawakami Y. Mast cells in atopic dermatitis. Curr Opin Immunol. 2009;21:666–78.
Nigrovic PA, Lee DM. Synovial mast cells: role in acute and chronic arthritis. Immunol Rev. 2007;217:19–37.
Abramson J, Pecht I. Regulation of the mast cell response to the type 1 Fcε receptor. Immunol Rev. 2007;217:231–54.
Rivera J. Molecular adapters in FcεRI signaling and the allergic response. Curr Opin Immunol. 2002;14:688–93.
Sarkar FH, Li Y, Wang Z, Padhye S. Lesson learned from nature for the development of novel anti-cancer agents: implication of isoflavone, curcumin, and their synthetic analogs. Curr Pharm Des. 2010;16:1801–12.
Park HH, Lee S, Son HY, Park SB, Kim MS, Choi EJ, et al. Flavonoids inhibit histamine release and expression of proinflammatory cytokines in mast cells. Arch Pharm Res. 2008;31:1303–11.
Murata T, Itoigawa M, Ito C, Nakao K, Tsuboi M, Kaneda N, et al. Induction of apoptosis in human leukaemia HL-60 cells by furanone-coumarins from Murraya siamensis. J Pharm Pharmacol. 2008;60:385–9.
Matsui T, Ito C, Itoigawa M, Okada T, Furukawa H. Effect of natsudaidain isolated from Citrus plants on TNF-α and cyclooxygenase-2 expression in RBL-2H3 cells. J Pharm Pharmacol. 2009;61:109–14.
Matsui T, Ito C, Itoigawa M, Okada T, Furukawa H. Anti-inflammatory activity of phenylpropanoids and phytoquinoids from Illicium species in RBL-2H3 cells. Planta Med. 2007;73:662–5.
Prasad KN, Hao J, Yi C, Zhang D, Qiu S, Jiang Y, et al. Antioxidant and anticancer activities of wampee (Clausena lansium (Lour.) Skeels) peel. J Biomed Biotechnol. 2009;2009:1–6.
Tang K, Zhang JT. Mechanism of (−) clausenamide induced calcium transient in primary culture of rat cortical neurons. Life Sci. 2004;74:1427–34.
Adebajo AC, Iwalewa EO, Obuotor EM, Ibikunle GF, Omisore NO, Adewunmi CO, et al. Pharmacological properties of the extract and some isolated compounds of Clausena lansium stem bark: anti-trichomonal, antidiabetic, anti-inflammatory, hepatoprotective and antioxidant effects. J Ethnopharmacol. 2009;122:10–9.
Maneerat W, Ritthiwigrom T, Cheenpracha S, Laphookhieo S. Carbazole alkaloids and coumarins from Clausena lansium roots. Phytochem Lett. 2012;5:26–8.
Feng Z, Li X, Zheng G, Huang L. Synthesis and activity in enhancing long-term potentiation (LTP) of clausenamide stereoisomers. Bioorg Med Chem Lett. 2009;19:2112–5.
Ito C. Search for novel anti-inflammatory agents having the inhibitory activity on histamine release from higher plants. Bull Res Inst Meijo Univ. 2012;17:39–42.
Milner PH, Coates NJ, Gilpin ML, Spear SR. SB-204900, a novel oxirane carboxamide from Clausena lansium. J Nat Prod. 1996;59:400–2.
Lin J-H. Cinnamamide derivatives from Clausena lansium. Phytochemistry. 1989;28:621–2.
Oh HA, Kim HM, Jeong HJ. Distinct effects of imperatorin on allergic rhinitis: imperatorin inhibits caspase-1 activity in vivo and in vitro. J Pharmacol Exp Ther. 2011;339:72–81.
Zhang D, Huang C, Yang C, Liu RJ, Wang J, Niu J, et al. Antifibrotic effects of curcumin are associated with overexpression of cathepsins K and L in bleomycin treated mice and human fibroblasts. Respir Res. 2011;12:154.
Kim D-C, Lee W, Bae J-S (2011) Vascular anti-inflammatory effects of curcumin on HMGB1-mediated responses in vitro. Inflamm Res 60:1161–1168.
Yin Y, Gong FY, Wu XX, Sun Y, Li YH, Chen T, et al. Anti-inflammatory and immunosuppressive effect of flavones isolated from Artemisia vestita. J Ethnopharmacol. 2008;120:1–6.
Theoharides TC, Alysandratos KD, Angelidou A, Delivanis DA, Sismanopoulos N, Zhang B, et al. Mast cells and inflammation. Biochim Biophys Acta. 2012;1822:21–33.
Wasiuk A, de Vries VC, Hartmann K, Roers A, Noelle RJ. Mast cells as regulators of adaptive immunity to tumours. Clin Exp Immunol. 2008;155:140–6.
Passante E, Frankish N. The RBL-2H3 cell line: its provenance and suitability as a model for the mast cell. Inflamm Res. 2009;58:737–45.
Gilfillan AM, Beaven MA. Regulation of mast cell responses in health and disease. Crit Rev Immunol. 2011;31:475–529.
Baumgartner RA, Ozawa K, Cunha-Melo JR, Yamada K, Gusovsky F, Beaven MA. Studies with transfected and permeabilized RBL-2H3 cells reveal unique inhibitory properties of protein kinase C γ. Mol Biol Cell. 1994;5:475–84.
Ozawa K, Yamada K, Kazanietz MG, Blumberg PM, Beaven MA. Different isozymes of protein kinase C mediate feedback inhibition of phospholipase C and stimulatory signals for exocytosis in rat RBL-2H3 cells. J Biol Chem. 1993;268:2280–3.
Ozawa K, Szallasi Z, Kazanietz MG, Blumberg PM, Mischak H, Mushinski JF, et al. Ca2+-dependent and Ca2+-independent isozymes of protein kinase C mediate exocytosis in antigen-stimulated rat basophilic RBL-2H3 cells. Reconstitution of secretory responses with Ca2+ and purified isozymes in washed permeabilized cells. J Biol Chem. 1993;268:1749–56.
Klemm S, Ruland J. Inflammatory signal transduction from the Fcε RI to NF-κ B. Immunobiology. 2006;211:815–20.
Kawakami Y, Kitaura J, Yao L, McHenry RW, Kawakami Y, Newton AC, et al. A Ras activation pathway dependent on Syk phosphorylation of protein kinase C. Proc Natl Acad Sci USA. 2003;100:9470–5.
Liu Y, Graham C, Parravicini V, Brown MJ, Rivera J, Shaw S. Protein kinase C θ is expressed in mast cells and is functionally involved in Fcε receptor I signaling. J Leukoc Biol. 2001;69:831–40.
Kandere-Grzybowska K, Kempuraj D, Cao J, Cetrulo CL, Theoharides TC. Regulation of IL-1-induced selective IL-6 release from human mast cells and inhibition by quercetin. Br J Pharmacol. 2006;148:208–15.
Kempuraj D, Madhappan B, Christodoulou S, Boucher W, Cao J, Papadopoulou N, et al. Flavonols inhibit proinflammatory mediator release, intracellular calcium ion levels and protein kinase C theta phosphorylation in human mast cells. Br J Pharmacol. 2005;145:934–44.
Hochdörfer T, Kuhny M, Zorn CN, Hendriks RW, Vanhaesebroeck B, Bohnacker T, et al. Activation of the PI3 K pathway increases TLR-induced TNF-α and IL-6 but reduces IL-1β production in mast cells. Cell Signal. 2011;23:866–75.
Huang P, Han J, Hui L. MAPK signaling in inflammation-associated cancer development. Protein Cell. 2010;1:218–26.
Coulthard LR, White DE, Jones DL, McDermott MF, Burchill SA. p38MAPK: stress responses from molecular mechanisms to therapeutics. Trends Mol Med. 2009;15:369–79.
Hundley TR, Prasad AR, Beaven MA. Elevated levels of cyclooxygenase-2 in antigen-stimulated mast cells is associated with minimal activation of p38 mitogen-activated protein kinase. J Immunol. 2001;167:1629–36.
Guan Z, Buckman SY, Pentland AP, Templeton DJ, Morrison AR. Induction of cyclooxygenase-2 by the activated MEKK1 → SEK1/MKK4 → p38 mitogen-activated protein kinase pathway. J Biol Chem. 1998;273:12901–8.
Hwang D, Jang BC, Yu G, Boudreau M. Expression of mitogen-inducible cyclooxygenase induced by lipopolysaccharide: mediation through both mitogen-activated protein kinase and NF-κB signaling pathways in macrophages. Biochem Pharmacol. 1997;54:87–96.
Gibbs BF, Plath KE, Wolff HH, Grabbe J. Regulation of mediator secretion in human basophils by p38 mitogen-activated protein kinase: phosphorylation is sensitive to the effects of phosphatidylinositol 3-kinase inhibitors and calcium mobilization. J Leukoc Biol. 2002;72:391–400.
Azzolina A, Guarneri P, Lampiasi N. Involvement of p38 and JNK MAPKs pathways in Substance P-induced production of TNF-α by peritoneal mast cells. Cytokine. 2002;18:72–80.
Teixeira CC, Fuchs FD. The efficacy of herbal medicines in clinical models: the case of jambolan. J Ethnopharmacol. 2006;108:16–9.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Andras Falus.
Rights and permissions
About this article
Cite this article
Matsui, T., Ito, C., Furukawa, H. et al. Lansiumamide B and SB-204900 isolated from Clausena lansium inhibit histamine and TNF-α release from RBL-2H3 cells. Inflamm. Res. 62, 333–341 (2013). https://doi.org/10.1007/s00011-012-0586-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-012-0586-8