Abstract
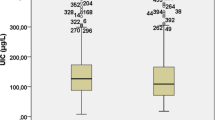
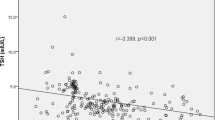
Background: Eleven years after the initiation of universal salt iodization program in Iran, the prevalence of goiter is still high in some areas. Aim: To investigate the role of thyroid autoimmunity in the etiology of residual goiter in schoolchildren of Isfahan, Iran. Material and methods: In a cross-sectional study, 2331 schoolchildren were selected by multi-stage random cluster sampling. Thyroid size was estimated in each child by inspection and palpation. Urinary iodine concentration (UIC), serum anti-thyroperoxidase antibody (anti-TPO Ab), and anti-thyroglobulin antibody (Anti-Tg Ab) were measured. Results: Overall, 32.9% of children had goiter. The median UIC was 1955.5 μg/dl. There was significant difference in prevalence of positive anti-TPO Ab in goitrous (grade 2) and non-goitrous children (9.7 vs 3.7%, p= 0.02). Goitrous children had higher prevalence of positive anti-Tg Ab than non-goitrous ones (15.1 vs 3.1%, p<0.001). Conclusions: According to the present study, goiter is still a public health problem in this region. This study suggests that thyroid autoimmunity is among the contributors of goiter persistence after elimination of iodine deficiency in Isfahan.
Similar content being viewed by others
References
WHO, UNICEF and ICCIDD. Assessment of iodine deficiency disorders and monitoring their elimination. A guide for programme managers. WHO/NHD/01.1. 2nd ed. Geneva: WHO, 2001.
Hashemipour M, Amini M, Aminorroaya A, et al. High prevalence of goiter in an iodine replete area: do thyroid auto-antibodies play a role? Asia Pac J Clin Nutr 2007, 16: 403–10.
Tsatsoulis A, Johnson EO, Andricula M, et al. Thyroid autoimmunity is associated with higher urinary iodine concentrations in an iodine-deficient area of Northwestern Greece. Thyroid 1999, 9: 279–83.
Luboshitzky R, Dgani Y, Atar S, Qupty G, Tamir A, Flatau E. Goiter prevalence in children immigrating from an endemic goiter area in Ethiopia to Israel. J Pediatr Endocrinol Metab 1995, 8: 123–5.
Premawardhana LD, Parkes AB, Mazziotti G, Lazarus JH. Autoimmune thyroiditis after elimination of iodine deficiency in Sri Lanka. Thyroid 2003, 13: 1187.
Prummel MF, Wiersinga WM. Thyroid peroxidase autoantibodies in euthyroid subjects. Best Pract Res Clin Endocrinol Metab 2005, 19: 1–15.
Gao TS, Teng WP, Shan ZY, et al. Effect of different iodine intake on schoolchildren’s thyroid diseases and intelligence in rural areas. Chin Med J (Engl) 2004, 117: 1518–22.
Mazziotti G, Premawardhana LD, Parkes AB, et al. Evolution of thyroid autoimmunity during iodine prophylaxis-the Sri Lankan experience. Eur J Endocrinol 2003, 149: 103–10.
Azizi F, Kimiagar M, Nafarabadi T, Yassai M. Current status of iodine deficiency disorders in the Islamic Republic of Iran. EMR Health Surv J 1990, 8: 23–7.
Aminorroaya A, Amini M, Rezvanian H, et al. Effects of iodized salt consumption on goiter prevalence in Isfahan: the possible role of goitrogens. Endocr Pract 2001, 7: 95–8.
Pino S, Fang SL, Braverman LE. Ammonium persulfate: a safe alternative oxidizing reagent for measuring urinary iodine. Clin Chem 1996, 42: 239–43.
Keshteli AH, Hashemipour M, Siavash M, Amini M. Selenium deficiency as a possible contributor of goiter in schoolchildren of Isfahan, Iran. Biol Trace Elem Res 2009, 129: 70–7.
Hashemipour M, Keshteli AH, Siavash Dastjerdi M, Amini M, Kelishadi R, Koleini N. Vitamin A status does not contribute to the residual goiter in schoolchildren of Isfahan, an iodine replenished area. Int J Food Sci Nutr 2009, 60: 19–27.
Zonenberg A, Kinalska I, Zarzycki W, Telejko B. Incidence of thyroid autoantibodies in the endemic goiter. Horm Metab Res 1994, 26: 238–42.
Doufas AG, Mastorakos G, Chatziioannou S, et al. The predominant form of non-toxic goiter in Greece is now autoimmune thyroiditis. Eur J Endocrinol 1999, 140: 505–11.
Bazrafshan HR, Mohammadian S, Ordookhani A, et al. Prevalence of goiter among schoolchildren from Gorgan, Iran, a decade after national iodine supplementation: association with age, gender, and thyroperoxidase antibodies. J Endocrinol Invest 2005, 28: 727–33.
Aminorroaya A, Momenzadeh M, Hovsepian S, Haghighi S, Amini M. Thyroid autoantibodies in women with and without thyroid disorders in an iodine-replete area. East Mediterr Health J 2008, 14: 325–32.
Zimmermann MB, Moretti D, Chaouki N, Torresani T. Introduction of iodized salt to severely iodine-deficient children does not provoke thyroid autoimmunity: a one year prospective trial in northern Morocco. Thyroid 2003, 13: 199–203.
Kabelitz M, Liesenkötter KP, Stach B, et al. The prevalence of anti-thyroid peroxidase antibodies and autoimmune thyroiditis in children and adolescents in an iodine replete area. Eur J Endocrinol 2003, 148: 301–7.
Loviselli A, Velluzzi F, Mossa P, et al. The Sardinian Autoimmunity Study: 3. Studies on circulating antithyroid antibodies in Sardinian schoolchildren: relationship to goiter prevalence and thyroid function. Thyroid 2001, 11: 849–57.
Milakovic M, Berg G, Eggertsen R, Lindstedt G, Nystrom E. Screening for thyroid disease of 15–17-year-old schoolchildren in an area with normal iodine intake. J Intern Med 2001, 250: 208–12.
Zois C, Stavrou I, Kalogera C, et al. High prevalence of autoimmune thyroiditis in schoolchildren after elimination of iodine deficiency in northwestern Greece. Thyroid 2003, 13: 485–9.
Simescu M, Varciu M, Nicolaescu E, et al. Iodized oil as a complement to iodized salt in schoolchildren in endemic goiter in Romania. Horm Res 2002, 58: 78–82.
Markou KB, Georgopoulos NA, Makri M, et al. Improvement of iodine deficiency after iodine supplementation in schoolchildren of Azerbaijan was accompanied by hypo and hyperthyrotropinemia and increased title of thyroid autoantibodies. J Endocrinol Invest 2003, 26 (Suppl): 43–8.
Heydarian P, Ordookhani A, Azizi F. Goiter rate, serum thyrotropin, thyroid autoantibodies and urinary iodine concentration in Tehranian adults before and after national salt iodization. J Endocrinol Invest 2007, 30: 404–10.
Wijeyaratne CN, Jayasinghe A, de Silva DG, Parkes AB, Lazarus JH, Premawardhana LD. Iodine prophylaxis, goitre and thyroid autoimmunity in Sri Lanka. Ceylon Med J 2005, 50: 20–3.
Premawardhana LD, Parkes AB, Mazziotti G, Lazarus JH. Autoimmune thyroiditis after elimination ofiodine deficiency in Sri Lanka. Thyroid 2003, 13: 1187.
Mazziotti G, Premawardhana LD, Parkes AB, et al. Evolution of thyroid autoimmunity during iodine prophylaxis—the Sri Lankan experience. Eur J Endocrinol 2003, 149: 103–10.
Mazziotti G, Amato G, Carella C. Is chronic autoimmune thyroiditis a systemic disease? Am J Med 2003, 115: 412–3.
Rezvanfar MR, Farahany H, Chehreiy A, Nemati M, Rostamy S, Karimy E. Urinary iodine excretion and antiperoxidase enzyme antibody in goitrous and healthy primary school children of Arak, Iran. J Endocrinol Invest 2007, 30: 274–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Khalili, N., Hashemipour, M., Keshteli, A.H. et al. The role of thyroid autoantibodies in the etiology of endemic goiter in schoolchildren of Isfahan, Iran. J Endocrinol Invest 32, 899–902 (2009). https://doi.org/10.1007/BF03345769
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF03345769