Abstract
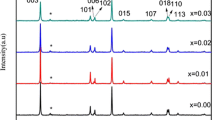
Three Al doped lithium nickel cobalt oxide (LiNi0.4Co0.4−xAl0.2+xO2) cathode materials for lithium ion batteries were synthesized by solid state reaction method at a temperature of 800 °C for 18 hours. The samples were crystalline as revealed by powder X-Ray diffraction (XRD). The ratios of the elements were determined by Energy Dispersive Analysis of X-rays (EDAX). The electrochemical properties obtained by charge/discharge cycling showed that the average discharge capacity for LiNi0.4Co0.4Al0.2O2 was 117 mAh/g. A good capacity retention was also shown by the material upon cycling.
Similar content being viewed by others
5. References
M. Wakihara and O. Yamamoto (Eds.), Lithium Ion Batteries, VCH, Kodanska, Tokyo, 1998.
G. Pistoia (Ed.), Lithium Batteries, New Materials, Development and Perspective, Elsevier, New York, 1994.
J.R. Dahn, E.W. Fuller, M. Obrovac and U. von Sacken, Solid State Ionics69, 265 (1994).
http://www.seimichemical.co.jp, http://www.smm.co.jp
T. Ohzuku, T. Yanagawa, M. Kouguchi and A. Ueda, J. Power Sources68, 131 (1997).
P. Biensan, J.P. Peres and F. Perton, (Eds.), The 1999 Joint International Meeting, 99-2, 1999.
S. Madhavi, G.V. Suba Rao, B.V.R. Chowdari and S.F.Y. Li, J. Power Sources93, 156 (2001).
K.K. Lee, W.S. Yoon, K.B. Kim, K.Y. Lee and S.T. Hong, J. Power Sources97, 308 (2001).
J.S. Weaving, F. Coowar, D.A. Teagle, J. Cullen, V. Dass, P. Bindah, R. Green and W.J. Macklin, J. Power Sources97, 733 (2001).
G.T.K. Fey, R.F. Shiu, V. Subramaniam, J.G. Chen and C.L. Chen J. Power Sources103, 265 (2002).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sethuprakhash, V., Basirun, W.J. & Arof, A.K. Structural and electrochemical properties of LiNi0.4Co0.4−xAl0.2+xO2 cathode materials for lithium-ion batteries. Ionics 11, 406–409 (2005). https://doi.org/10.1007/BF02430256
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02430256