Summary
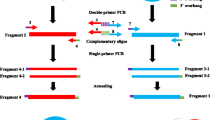
Gene conversion, the non-reciprocal transfer of sequence information between homologous DNA sequences, has been reported in lower eukaryotes, mammals and in Escherichia coli. In an E. coli rec + strain, we established a plasmid carrying two different deleted neo genes (neoDL and neoDR) in an inverted orientation and then selected for homologous recombination events that had reconstructed an intact neo + gene. We found some plasmids that had apparently experienced intramolecular gene conversion. Further evidence, however, suggests that they are products of multiple rounds of reciprocal crossing-over,apparently involving two plasmid molecules. First, most of the Neo+ clones contained multiple types of Neo+ plasmids, although the frequency of producing the neo + clones was low. Second, all the neo + clones also contained, as a minority, one particular form of dimer, which can be formed by reciprocal crossing-over between neoDL of one plasmid molecule and neoDR of another plasmid molecule. Third, in reconstruction experiments, we cloned and purified this dimer and transferred it back into the rec + cells. The dimer gave rise to clones containing multiple types of neo + recombinant monomers, including those apparent gene conversion types, and containing only few molecules of this dimer plasmid.
Similar content being viewed by others
References
Amundsen SK, Taylor AF, Chaudhury AM, Smith GR (1986) recD: The gene for an essential third subunit of exonuclease V. Proc Natl Acad Sci USA 83:5558–5562
Bachman BJ (1987) Derivation genotypes of some mutant derivatives of Escherichia coli K-12. In: Neidhardt FC, Ingraham JL, Low KB, Margasanik B, Schaechter M (eds) Escherichia coli and Salmonerlla typhimurium. Cellular and molecular biology. ASM, Washington DC, pp 1190–1219
Balbas P, Soberon X, Merino E, Zurita M, Lomeli H, Valle F, Flores N, Bolivar F (1986) Plasmid vector pBR322 and its special-purpose derivatives — a review. Gene 50:3–40
Benbow RM, Zuccarelli AJ, Sinsheimer RL (1975) Recombinant DNA molecules of bacteriophage phiX174. Proc Natl Acad Sci USA 72:235–239
Boliver F, Rodriguez RL, Greene PJ, Betlach MC, Heyneker HL, Boyer HW (1977) Construction and characterization of new cloning vehicles. Gene 2:95–133
Chen EY, Howley PM, Levinson AD, Seeburg PH (1982) The primary structure and genetic organization of the bovine papillomavirus type 1 genome. Nature 299:529–534
Clark AJ, Margulies AD (1965) Isolation and characterization of recombination deficient mutants of E. coli K12. Proc Natl Acad Sci USA 53:451–459
Cohen A, Laban A (1983) Plasmidic recombination in Escherichia coli K12: the role of the recF gene. Mol Gen Genet 189:471–474
Csonka LN, Clark AJ (1979) Deletions generated by the transposon Tn10 in the srl recA region of the Escherichia coli K-12 chromosome. Genetics 93:321–343
Das Gupta C, Wu AM, Kahn R, Cunningham RP, Radding (1981) Concerted strand exchange and formation of Holliday structures by E. coli recA protein. Cell 25:507–516
Doherty MJ, Morrison PT, Kolodner R (1983) Genetic recombination of bacterial plasmid DNA: Physical and genetic analysis of the products of plasmid recombination in Escherichia coli. J Mol Biol 167:539–560
Emmerson PT, Howard-Flanders P (1967) Cotransduction with thy of a gene required for geentic recombination in Escherichia coli. J Bacteriol 93:1729–1731
Esposito MS (1978) Evidence that spontaneous mitotic recombination occurs at the two-strand stage. Proc Natl Acad Sci USA 75:4436–4440
Evans GA, Margulies DH, Camerini-Otero RD, Ozato K, Seidman JG (1982) Structure and expression of a mouse major histocompatibility antigen gene, H-2Ld. Proc Natl Acad Sci USA 79:1994–1998
Feiss M, Kobayashi I, Widner W (1983) Separate sites for binding and nicking of bacteriophage lambda DNA by terminase. Proc Natl Acad Sci USA 80:955–959
Fishel RA, James AA, Kolodner R (1981) recA-independent general genetic recombination of plasmids. Nature 294:184–186
Goldmark RJ, Linn S (1970) An endonuclease activity from Escherichia coli absent from certain rec - strain. Proc Natl Acad Sci USA 67:434–441
Hanahan D (1985) Techniques for transformation of E. coli. In: Glover DM (ed) DNA cloning, vol I. IRL Press, Oxford, pp 109–135
Holliday R (1964) A mechanism for gene conversion in fungi. Genet Res 5:282–304
Holmes DS, Quigley M (1981) A rapid boiling method for the preparation of bacterial plasmids. Anal Biochem 114:193–198
Horii Z, Clark AJ (1973) Genetic analysis of the RecF pathway of genetic recombination in Escherichia coli K12. J Mol Biol 80:327–344
Ikeda H, Kobayashi I (1979) recA-mediated recombination of bacteriophage lambda: structure of recombinant and intermediate DNA molecules and their packaging in vitro. Cold Spring Harbor Symp Quant Biol 43:1009–1021
James AA, Morrison PT, Kolodner R (1982) Genetic recombination of bacterial plasmid DNA; Analysis of the effect of recombination-deficient mutation on plasmid recombination. J Mol Biol 160:411–430
Kaiser K, Murray NE (1979) Physical characterization of the “Rac prophage” in E. coli K12. Mol Gen Genet 175:159–174
Kitani Y, Olive LS (1970) Alteration of gene conversion patterns in Sordaria fimicola by supplementation with DNA bases. Proc Natl Acad Sci USA 66:1290–1297
Kobayashi I, Ikeda H (1978) On the role of recA gene product in genetic recombination: An analysis by in vitro packaging of recombinant DNA molecules formed in the absence of protein synthesis. Mol Gen Genet 166:25–29
Kobayashi I, Ikeda H (1983) Double Holliday structure: A possible in vivo intermediate form of general recombination in Escherichia coli. Mol Gen Genet 191:213–220
Kobayashi I, Murialdo H, Crasemann JM, Stahl MM, Stahl FW (1982) Orientation of cohesive end site cos determines the active orientation of chi sequence in stimulating recA recBC-mediated recombination in phage lambda lytic infections. Proc Natl Acad Sci USA 79:5981–5985
Kobayashi I, Stahl MM, Stahl FW (1984) The mechanism of the chi-cos interaction in RecA-RecBC-mediated recombination in phage lambda. Cold Spring Harbor Symp Quant Biol 49:497–506
Laban A, Cohen A (1981) Interplasmidic and intraplasmidic recombination in Escherichia coli K-12. Mol Gen Genet 184:200–207
Lusky M, Botchan M (1981) Inhibition of SV40 replication in simian cells by specific pBR322 DNA sequences. Nature 293:79–81
Meselson MS, Radding CM (1975) A general model for genetic recombination. Proc Natl Acad Sci USA 72:358–361
Mitchell MB (1955) Aberrant recombination of pyridoxine mutants of Neurospora. Proc Natl Acad Sci USA 41:215–220
Oishi M (1969) An ATP-dependent deoxyribonuclease from Escherichia coli with a possible role in genetic recombination. Proc Natl Acad Sci USA 64:1292–1299
Ponticelli AS, Schultz DW, Taylor AF, Smith GR (1985) Chi dependent DNA strand cleavage by RecBC enzyme. Cell 41:145–151
Rosamond J, Telander KM, Linn S (1979) Modulation of the recBC enzyme of Escherichia coli K12 by Ca++. J Biol Chem 254:8646–8652
Rosenberg SM, Stahl MM, Kobayashi I, Stahl FW (1985) Improved in vitro packaging of coliphage lambda DNA: A one strain system free from endogeneous phage. Gene 38:165–175
Rossignol JL, Nicolas A, Hamza H, Langin T (1984) Origins of gene conversion reciprocal exchange in Ascobolus. Cold Spring Harbor Symp Quant Biol 49:13–21
Rubnitz J, Subramani S (1986) Extrachromosomal and chromosomal gene conversion in mammalian cells. Mol Cel Biol 6:1608–1614
Saito I, Stark GR (1986) Charomids: Cosmid vectors for efficient cloning and mapping of large or small restriction fragments. Proc Natl Acad Sci USA 83:8664–8668
Sarver N, Byrne YC, Howley PM (1982) Transformation and replication in mouse cells of bovine papilloma virus-pML2 plasmid vector that can be reduced in bacteria. Proc Natl Acad Sci USA 79:7147–7151
Shibata T, Das Gupta C, Cunningham RP, Radding CM (1979) Purified Escherichia coli recA protein catalyzes homologous pairing of superhelical DNA and single-stranded fragments. Proc Natl Acad Sci USA 76:1638–1642
Slightom J, Blechl AE, Smithies O (1980) Human fetal Ggamma-and Agamma globin genes: Complete nucleotide sequences suggest that DNA can be exchanged between these duplicated genes. Cell 21:627–638
Smith GR, Kunes SM, Schultz DW, Taylor A, Triman KL (1981) Structure of Chi hot spots of generalized recombination. Cell 24:429–438
Southern PJ, Berg P (1982) Transformation of mammalian cells to antibiotic resistance with a bacterial gene under control of the SV40 early region promoter. J Mol Appl Genet 1:327–341
Stahl FW (1969) The mechanics of inheritance. Prentice-Hall, New Jersey, p 13
Stahl FW (1979a) Genetic recombination. Freeman, San Francisco
Stahl FW (1979b) Special sites in generalized recombination. Annu Rev Genet 13:7–24
Stahl FW, Kobayashi I, Stahl MM (1985) In phage lambda, cos is a recombinator for Red pathway. J Mol Biol 181:199–209
Strathern A, Herkowitz I (1975) Defective prophage in Escherichia coli K12 strains. Virology 67:136–143
Summers DK, Sherratt DJ (1984) Multimerization of high copy number plasmids causes instability: ColE1 encodes a determinant essential for plasmid monomerization and stability. Cell 36:1097–1103
Szostak JW, Orr-Weaver TL, Rothstein RJ, Stahl FW (1983) The double-strand break repair model for recombination. Cell 33:25–33
Telander-Muskavitch KM, Linn S (1980) Electron microscopy of E. coli recBC enzyme reaction intermediates. In: Alberts B, Fox CF (eds) Mechanistic Studies of DNA Replication and Genetic Recombination. Academic Press, New York, pp 901–908
Thompson BJ, Escarmis C, Parker B, Slater WC, Doniger J, Tessman I, Warner RC (1975) Figure-8 configuration of dimers of S13 and phiX174 replicative form DNA. J Mol Biol 91:409–419
Weiss EH, Mellor A, Golden L, Fahrner K, Simpson E, Hurst J, Flavell RA (1983) The structure of a mutant H-2 gene suggests that the generation of polymorphism in H-2 genes may occur by gene conversion-like events. Nature 301:671–674
Whitehouse HLK (1982) Genetic recombination. Wiley, New York
Author information
Authors and Affiliations
Additional information
Communicated by M. Takanami
Rights and permissions
About this article
Cite this article
Yamamoto, K., Yoshikura, H., Takahashi, N. et al. Apparent gene conversion in an Escherichia coli rec + strain is explained by multiple rounds of reciprocal crossing-over. Mol Gen Genet 212, 393–404 (1988). https://doi.org/10.1007/BF00330842
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00330842