Abstract
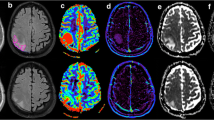
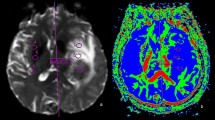
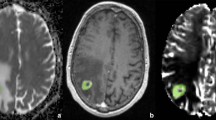
Brain metastasis is the second most common brain tumor in adults and still is a very common and devastating clinical problem for both cancer patients and treating physicians. Precise diagnosis of solitary brain metastasis is crucial for proper treatment planning and can directlyaffect patient morbidity and mortality. Although computed tomography is widely used for tumor diagnosis due to easy accessibility and low cost, magnetic resonance (MR) imaging is the method of choice for screening and differential diagnosis of solitary metastasis because of higher contrast resolution and availability of advanced imaging techniques such as diffusion-weighted and diffusion-tensor imaging, MR spectroscopy, perfusion and functional MR imaging. Major differential diagnosis of solitary metastasis includes high-grade glioma. Higher peritumoral rCBV values and Cho/Cr ratios of infiltrative gliomas, indicating existence of tumorcells, help to differentiate them from non-infiltrative metastasis. In conclusion, although the diagnosis of solitary brain tumors is still somewhat problematic, advanced MR imaging techniques provide valuable information to overcome the diagnostic hindrance and to increase effectiveness in therapy monitoring.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
REFERENCES
Andersen, C., and Jensen, F.T. (1998) Differences in blood–tumour-barrier leakage of human intracranial tumours: quantitative monitoring of vasogenic oedema and its response to glucocorticoid treatment. Acta Neurochir. (Wien) 140:919–924
Atlas, S.W., Lavi E., and Fisher P.G. (2002) Intraaxial brain tumors. In: Atlas, S.W. (Ed). MRI of the brain and spine, 3rd edn, vol 1. Lippincott Williams & Wilkins, Philadelphia, PA, pp. 565–693
Bastin, M.E., Carpenter, T.K., Armitage, P.A., Sinha, S., Wardlaw, J.M., and Whittle, I.R. (2006) Effects of dexamethasone on cerebral perfusion and water diffusion in patients with high-grade glioma. AJNR Am. J. Neuroradiol. 27:402–408
Bertossi, M., Virgintino, D., Maiorano, E., Occhiogrosso, M., and Roncali, L. (1997) Ultrastructural and morphometric investigation of human brain capillaries in normal and peritumoral tissues. Ultrastruct. Pathol. 21:41–49
Bogomolny, D.L., Petrovich, N.M., Hou, B.L., Peck, K.K., Kim, M.J., and Holodny, A.I. (2004) Functional MRI in the brain tumor patient. Top. Magn. Reson. Imaging 15:325–335
Bulakbasi, N. (2004) Clinical applications of proton MR spectroscopy in the diagnosis of brain tumours. Spectroscopy-An. Int. J. 18:143–153
Bulakbasi, N., Kocaoglu, M., Ors, F., Tayfun, C., and Ucoz, T. (2003) Combination of single-voxel proton MR spectroscopy and apparent diffusion coefficient calculation in the evaluation of common brain tumors. AJNR Am. J. Neuroradiol. 24:225–233
Bulakbasi, N., Guvenc, I., Onguru, O., Erdogan, E., Tayfun, C., and Ucoz, T. (2004) The added value of the apparent diffusion coefficient calculation to magnetic resonance imaging in the differentiation and grading of malignant brain tumors. J. Comput. Assist. Tomogr. 28:735–746
Bulakbasi, N., Kocaoglu, M., Farzaliyev, A., Tayfun, C., Ucoz, T., and Somuncu, I. (2005) Assessment of diagnostic accuracy of perfusion MR imaging in primary and metastatic solitary malignant brain tumors. AJNR Am. J. Neuroradiol. 26:2187–2199
Cha, S. (2004) Perfusion MR imaging of brain tumors. Top. Magn. Reson. Imaging 15:279–289
Cha, S. (2006) Update on brain tumor imaging: from anatomy to physiology. AJNR Am. J. Neuroradiol. 27:475–487
Chang, E.L., Hassenbusch, S.J.3rd., Shiu, A.S., Lang, F.F., Allen, P.K., Sawaya, R., and Maor, M.H. (2003) The role of tumor size in the radiosurgical management of patients with ambiguous brain metastases. Neurosurgery 53:272–280; discussion: 280–281
Cho, S.K., Na, D.G., Ryoo, J.W., Roh, H.G., Moon, C.H., Byun, H.S., and Kim, J.H. 2002. Perfusion MR imaging: clinical utility for the differential diagnosis of various brain tumors. Korean J. Radiol. 3:171–179
Ercan, N., Gultekin, S., Celik, H., Tali, T.E., Oner, Y.A., and Erbas, G. (2004) Diagnostic value of contrast-enhanced fluid-attenuated inversion recovery MR imaging of intracranial metastases. AJNR Am. J. Neuroradiol. 25:761–765
Erdogan, C., Hakyemez, B., Yildirim, N., and Parlak, M. (2005) Brain abscess and cystic brain tumor: discrimination with dynamic susceptibility contrast perfusion-weighted MRI. J. Comput. Assist. Tomogr. 29: 663–667
Essig, M., Waschkies, M., Wenz, F., Debus, J., Hentrich, H.R., and Knopp, M.V. (2003) Assessment of brain metastases with dynamic susceptibility-weighted contrast-enhanced MR imaging: initial results. Radiology 228:193–199
Ewing, J. (1940) Metastasis. In: Ewing, J. (ed) Neoplastic diseases: a treatise on tumors. W.B. Saunders, Philadelphia, PA, pp. 62–74
Fidler, I.J., Schackert, G., Zhang, R.D., Radinsky, R., and Fujimaki, T. (1999) The biology of melanoma brain metastasis. Cancer Metastasis Rev. 18:387–400
Field, A.S., Alexander, A.L., Wu, Y.C., Hasan, K.M., Witwer, B., Badie, B. (2004) Diffusion tensor eigenvector directional color imaging patterns in the evaluation of cerebral white matter tracts altered by tumor. J. Magn. Reson. Imaging. 20:555–562
Field, A.S., and Alexander, A.L. (2004) Diffusion tensor imaging in cerebral tumor diagnosis and therapy. Top. Magn. Reson. Imaging 15:315–324
Gavrilovic, I.T., and Posner, J.B. (2005) Brain metastases: epidemiology and pathophysiology. J. Neurooncol. 75:5–14
Gruber, S., Mlynarik, V., and Moser, E. (2003) High resolution 3D proton spectroscopic imaging of the human brain in 3T: SNR issues and application for anatomy-matched voxel sizes. Mag. Reson. Med. 49:299–306
Hartmann, M., Jansen, O., Heiland, S., Sommer, C., Munkel, K., and Sartor, K. (2001) Restricted diffusion within ring enhancement is not pathognomonic for brain abscess. AJNR Am. J. Neuroradiol. 22:1738–1742
Jellison, B.J., Field, A.S., Medow, J., Lazar, M., Salamat, M.S., and Alexander, A.L. (2004) Diffusion tensor imaging of cerebral white matter: a pictorial review of physics, fiber tract anatomy, and tumor imaging patterns. AJNR Am. J. Neuroradiol. 25:356–369
Kadota, O., Kohno, K., Ohue, S., Kumon, Y., Sakaki, S., Kikuchi, K., and Miki, H. (2001) Discrimination of brain abscess and cystic tumor by in vivo proton magnetic resonance spectroscopy. Neurol. Med. Chir. (Tokyo). 41:121–126
Katz, J.M., and Segal, A.Z. (2005) Incidence and etiology of cerebrovascular disease in patients with malignancy. Curr. Atheroscler Rep. 7:280–288
Kim, S.Y., Kim, J.S., Park, H.S., Cho, M.J., Kim, J.O., Kim, J.W., Song, C.J., Lim, S.P., and Jung, S.S. (2005) Screening of brain metastasis with limited magnetic resonance imaging (MRI): clinical implications of using limited brain MRI during initial staging for non-small cell lung cancer patients. J. Korean Med. Sci. 20:121–126
Kitis, O., Altay, H., Calli, C., Yunten, N., Akalin, T., and Yurtseven, T. (2005) Minimum apparent diffusion coefficients in the evaluation of brain tumors. Eur. J. Radiol. 55:393–400
Korinth, M.C., Delonge, C., Hutter, B.O., and Gilsbach, J.M. (2002) Prognostic factors for patients with microsurgically resected brain metastases. Onkologie 25:420–425
Krabbe, K., Gideon, P., Wagn, P., Hansen, U., Thomsen, C., and Madsen, F. (1997) MR diffusion imaging of human intracranial tumours. Neuroradiology 39:483–489
Kremer, S., Grand, S., Berger, F., Hoffmann, D., Pasquier, B., Remy, C., Benabid, A.L., and Bas, J.F. (2003) Dynamic contrast-enhanced MRI: differentiating melanoma and renal carcinoma metastases from high-grade astrocytomas and other metastases. Neuroradiology 45:44–49
Kremer, S., Grand, S., Remy, C., Pasquier, B., Benabid, A.L., Bracard, S., and Le Bas, J.F. (2004) Contribution of dynamic contrast MR imaging to the differentiation between dural metastasis and meningioma. Neuroradiology 46:642–648
Law, M. (2004) MR spectroscopy of brain tumors. Top. Magn. Reson. Imaging 15:291–313
Law, M., Cha, S., Knopp, E.A., Johnson, G., Arnett, J., and Litt, A.W. (2002) High-grade gliomas and solitary metastases: differentiation by using perfusion and proton spectroscopic MR imaging. Radiology 222:715–721
Li, K.C., Pandit, S.D., Guccione, S., and Bednarski, M.D. (2004) Molecular imaging applications in nanomedicine. Biomed. Microdevices 6:113–116
Lu, S., Ahn, D., Johnson, G., and Cha, S. (2003) Peritumoral diffusion tensor imaging of high-grade gliomas and metastatic brain tumors. AJNR Am. J. Neuroradiol. 24:937–941
Lu, S., Ahn, D., Johnson, G., Law, M., Zagzag, D., and Grossman, R.I. (2004) Diffusion-tensor MR imaging of intracranial neoplasia and associated peritumoral edema: introduction of the tumor infiltration index. Radiology 232:221–228
Luker, G.D., and Piwnica-Worms, D. (2001) Molecular imaging in vivo with PET and SPECT. Acad. Radiol. 8:4–14
Nadal Desbarats, L., Herlidou, S., de Marco, G., Gondry-Jouet, C., Le Gars, D., Deramond, H., and Idy-Peretti, I. (2003) Differential MRI diagnosis between brain abscesses and necrotic or cystic brain tumors using the apparent diffusion coefficient and normalized diffusion-weighted images. Magn. Reson. Imaging 21:645–650
Nathoo, N., Chahlavi, A., Barnett, G.H., and Toms, S.A. (2005) Pathobiology of brain metastases. J. Clin. Pathol. 58:237–342
Nguyen, L.N., Maor, M.H., and Oswald, M.J. (1998) Brain metastases as the only manifestation of an undetected primary tumor. Cancer 83:2181–2184
Nussbaum, E.S., Djalilian, H.R., Cho, K.H., and Hall, W.A. (1996) Brain metastases. Histology, multiplicity, surgery, and survival. Cancer 78:1781–1788
Oh, J., Cha, S., Aiken, A.H., Han, E.T., Crane, J.C., Stainsby, J.A., Wright, G.A., Dillon, W.P., and Nelson, S.J. (2005) Quantitative apparent diffusion coefficients and T2 relaxation times in characterizing contrast enhancing brain tumors and regions of peritumoral edema. J. Magn. Reson. Imaging 21:701–708
Paget, S. (1889) The distribution of secondary growths in cancer of the breast. Lancet i:571–573
Pierpaoli, C., Righini, A., Linfante, I., Tao-Cheng, J.H., Alger, J.R., and Di Chiro, G. (1993) Histopathologic correlates of abnormal water diffusion in cerebral ischemia: diffusion-weighted MR imaging and light and electron microscopic study. Radiology 189:439–448
Posner, J.B., and Chernik, N.L. (1978) Intracranial metastases from systemic cancer. Adv. Neurol. 19:579–592
Rosen, B.R., Belliveau, J.W., Vevea, J.M., and Brady, T.J. (1990) Perfusion imaging with NMR contrast agents. Magn. Reson. Med. 14:249–265
Schouten, L.J., Rutten, J., Huveneers, H.A., and Twijnstra, A. (2002) Incidence of brain metastases in a cohort of patients with carcinoma of the breast, colon, kidney, and lung and melanoma. Cancer 94:2698–2705
Smith, J.S., Cha, S., Mayo, M.C., McDermott, M.W., Parsa, A.T., Chang, S.M., Dillon, W.P., and Berger, M.S. (2005) Serial diffusion-weighted magnetic resonance imaging in cases of glioma: distinguishing tumor recurrence from postresection injury. J. Neurosurg. 103:428–438
Tang, Y.M., Ngai, S., and Stuckey, S. (2006) The solitary enhancing cerebral lesion: can FLAIR aid the differentiation between glioma and metastasis? AJNR Am. J. Neuroradiol. 27:609–611
Terada, H., and Kamata, N. (2003) Contribution of the combination of (201)Tl SPECT and (99m)T(c)O(4)(-) SPECT to the differential diagnosis of brain tumors and tumor-like lesions. A preliminary report. J. Neuroradiol. 30:91–94
Thng, C.H., Tay, K.H., Chan, L.L., Lim, E.H., Khoo, B.K., Huin, E.L., and Tan, K.P. (1999) Magnetic resonance imaging of brain metastases: magnetisation transfer or triple dose gadolinium? Ann. Acad. Med. Singapore 28:529–533
Weiss, L. (1992) Comments on hematogenous metastatic patterns in humans as revealed by autopsy. Clin. Exp. 10:191–199
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2011 Springer Netherlands
About this chapter
Cite this chapter
Bulakbasi, N., Kocaoglu, M. (2011). Metastatic Solitary Malignant Brain Tumor: Magnetic Resonance Imaging. In: Hayat, M. (eds) Methods of Cancer Diagnosis, Therapy, and Prognosis. Methods of Cancer Diagnosis, Therapy and Prognosis, vol 8. Springer, Dordrecht. https://doi.org/10.1007/978-90-481-8665-5_24
Download citation
DOI: https://doi.org/10.1007/978-90-481-8665-5_24
Published:
Publisher Name: Springer, Dordrecht
Print ISBN: 978-90-481-8664-8
Online ISBN: 978-90-481-8665-5
eBook Packages: MedicineMedicine (R0)