Abstract
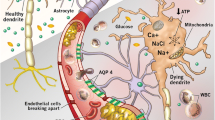
Permanent brain damage caused by ischemia and reperfusion that results from disease processes such as stroke and cardiac arrest (CA) with resuscitation has been estimated to affect approx 200,000 patients in the United States annually (1). Neuronal damage from stroke and CA occur by different mechanistic models of injury. In ischemic stroke, only a portion of the brain is at risk, and the ischemia is only complete in the center of the vulnerable area. This central area of dense ischemia is surrounded by a penumbral zone in which blood flow is diminished but not completely lost. As opposed to CA with resuscitation, flow ceases altogether and the entire brain is at risk for a transient period of complete ischemia followed by reperfusion (2).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
White BC, Grossman LI, O’Neil BJ, et al. Global brain ischemia and reperfusion. Ann Emerg Med 1996; 27:588–594.
Singh NC, Kochanek PM, Schiding JK, Melick JA, Nemoto EM: Uncoupled cerebral blood flow and metabolism after severe global ischemia in rats. J Cereb Blood Flow Metab 1992; 12:802–808.
Safar P. Brain resuscitation. Special symposium issue. Crit Care Med 1978; 6:199–214.
Abramson NS, Sutton-Tyrell K, Safar P. A randomized clinical study of a calcium-entry blocker (lidoflazine) in the treatment of comatose survivors of cardiac arrest. Brain Resuscitation Clinical Trial II Study Group. N Engl J Med 1991; 324:1225–1231.
Eisenberg MS, Horwood BT, Cummins RO, Reynolds-Haertle TR. Cardiac arrest and resuscitation: a tale of 29 cities. Ann Emerg Med 1990; 19:179–186.
Lucking SE, Pollack MM, Fields AI. Shock following generalized hypoxic-ischemic injury in previously healthy infants and children. J Pediatr 1986; 108:359–364.
Kern KB, Hilwig RW, Berg RA, et al. Postresuscitaion left ventricular dysfunction systolic and diastolic dysfunction: treatment with dobutamine. Circulation 1997; 95:2610–2613.
ACCP-SCCM Consensus Conference Definitions for sepsis and organ failure and guidelines for the use of innovative therapies in sepsis. Chest 1992; 101:1644–1655.
Oppert M, Gleiter CH, Müller C, et al. Kinetics and characteristics of an acute phase response following cardiac arrest. Intensive Care Med 1999; 25:1386–1394.
Shapiro HM: Intracranial hypertension, therapeutic and anesthetic considerations. Anesthesiology 1975; 43:445–471.
Cantu RC, Ames A, DiGancinto G, et al: Hypotension: a major factor limiting recovery from cerebral ischemia. J Surg Res 1969; 9:525–529.
Enna B, Wenzel V, Schocke M, et al. Excellent coronary perfusion pressure during cardiopulmonary resuscitation is not good enough to ensure long-term survival with good neurological outcome: a porcine case report. Resuscitation 2000; 47:41–49.
Homer-Vanniasinkam S, Crinnion JN, Gough MJ. Post-ischemic organ dysfunction: a review. Eur J Vasc Endovasc Surg 1997; 14:195–203.
Nielsen VG, Tan S, Baird MS, McCammon AT, Parks DA. Gastric intramucosal pH and multiple organ injury: impact of ischemia-reperfusion and xanthine oxidase. Crit Care Med 1996; 24:1339–1344.
Safar P. Cerebral resuscitation after cardiac arrest: A review. Circulation 1986; 74(Suppl):IV138–IV153.
Charlat MI, O’Neill PG, Hartley CJ, Roberts R, Bolli R. Prolonged abnormalities of the left ventricular diastolic wall thinning in the “stunned” myocardium in conscious dogs: time course and relation to systolic function. J Am Coll Cardiol 1989; 13:185–194.
Eisenberg MS, Bergner L, Hallstrom AP. Out-of-hospital cardiac arrest: improved survival with paramedics services. Lancet 1980; 1:812–815.
AHA medical/scientific statement: improving survival from sudden cardiac arrest: the “chain of survival” concept. Circulation 1991; 83:1832–1847.
Lee SK, Vaagenes P, Safar P, Stezoski SW, Scanlon M. Effect of cardiac arrest time on cortical cerebral blood flow during subsequent standard external cardiopulmonary resuscitation in rabbits. Resuscitation 1989; 17:105–117.
Bircher N, Safar P, Stewart R. A comparison of standard, “MAST”-augmented and open chest CPR in dogs. A preliminary investigation. Crit Care Med 1980; 8:147–152.
Safar P. Cerebral resuscitation after cardiac arrest: research initiatives and future direction. Ann Emerg Med 1993; 22:324–349.
Negovsky VA. Postresuscitation disease. Crit Care Med 1988; 16:942–946.
Beckman JS, Minor RL Jr, White CW, Repine JE, Rosen GM, Freeman BA. Superoxide dismutase and catalase conjugated to polyethylene glycol increases endothelial enzyme activity and oxidant resistance. J Biol Chem 1988; 263:6884–6892.
Safar P, Stezoski W, Nemoto EM. Amelioration of brain damage after 12 minutes of cardiac arrest in dogs. Arch Neurol 1976; 33:91–95.
Siesjö BK. Mechanisms of ischemic brain damage. Crit Care Med 1988; 16:954–963.
Safar P. Effects of the postresuscitation syndrome on cerebral recovery from cardiac arrest. Crit Care Med 1985; 13:932–935.
White BC, Sullivan JM, DeGarcia DJ, et al. Brain ischemia and reperfusion: molecular mechanism of neuronal injury. J Neurol Sci 2000; 179:1–33.
Kumar K, Goosmann M, Krause GS, et al. Ultrastructural and ionic changes in global ischemic dog brain. Acta Neuropathol 1987; 73:393–399.
Sato M, Hashimoto H, Kosaka F. Histological changes of neuronal damage in vegetative dogs induced by 18 minutes of complete global brain ischemia: two-phase damage of Purkinje cells and hippocampal CA1 pyramidal cells. Acta Neuropathol 1990; 80:527–534.
Garcia JH, Lossinsky AS, Kauffman FC, Conger KA. Neuronal ischemic injury: light microscopy, ultrastructure and biochemistry. Acta Neuropathol 1978; 43:85–95.
Martin LJ. Neuronal cell death in nervous system development, disease, and injury. Int J Mol Med 2001; 7:455–778.
Pulsinelli W, Brierley JB, Plum F. Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann Neurol 1982; 11:491–498.
Lind B, Snyder J, Safar P. Total brain ischemia in dogs: cerebral physiologic and metabolic changes after 15 minutes of circulatory arrest. Resuscitation 1975; 4:97–113.
Morimoto Y, Yamamura T, Kemmotsu O. Influence of hypoxic and hypercapnic acidosis on brain water content after forebrain ischemic in the rat. Crit Care Med 1993; 21:907–913.
Chan PH. Role of oxidants in ischemic brain damage. Stroke 1996; 27:1124–1129.
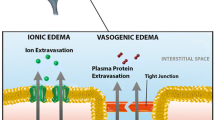
Rosenberg GA. Ischemia brain edema. Prog Cardiovasc Dis 1999; 42:209–216.
Taniguchi M, Yamashita T, Kumura E, et al. Induction of aquaporin-4 water channel mRNA after focal cerebral ischemia in rat. Brain Res Mol Brain Res 2000; 78:131–137.
Fujimura M, Gasche Y, Morita-Fujimura Y, Massengale J, Kawase M, Chan PH. Early appearance of activated matrix metalloproteinase-9 and blood-brain barrier disruption in mice after focal cerebral ischemia and reperfusion. Brain Res 1999; 842:92–100.
Heo JH, Lucero J, Abumiya T, Koziol JA, et al. Matrix metalloproteinases increase very early during experimental focal cerebral ischemia. J Cereb Blood Flow Metab 1999; 19:624–633.
Ereci¬Ωska M, Silver IA: ATP and brain function. J Cereb Blood Flow Metab 1989; 9:2–19.
Koehler RC, Backofen JE, McPherson RW, Jones MD Jr, Rogers MC, Traystman RJ. Cerebral blood flow and evoked potentials during Cushing response in sheep. Am J Physiol 1989; 256:H779–H788.
Symon L. Flow thresholds in brain ischemia and the effects of drugs. Br J Anesth 1985; 57:34–43.
Siesjö BK: Cell damage in the brain: A speculative synthesis. J Cereb Blood Flow Metab 1981; 1:155–185.
Cerchiari EL, Hoel TM, Safar P, Sclabassi RJ. Protective effects of combined superoxide dismutase and deferoxamine on recovery of cerebral blood flow and function after cardiac arrest in dogs. Stroke 1987; 18:869–878.
Sundgreen C, Larsen FS, Herzog TM, Knudsen GM, Boesgaard S, Aldershvile J. Autoregulation of cerebral blood flow in patients resuscitated from cardiac arrest. Stroke 2001; 32:128–132.
Shaffner DH, Eleff SM, Brambrink AM, et al. Effect of arrest time and cerebral perfusion pressure during cardiopulmonary resuscitation on cerebral blood flow, metabolism, adenosine triphosphate recovery, and pH in dogs. Crit Care Med 1999; 27:1335–1342.
Hossman KA, Ophoff BG. Recovery of monkey brain after prolonged ischemia. I. Electrophysiology and brain electrolytes. J Cereb Blood Flow Metab 1986; 6:15–21.
Opie LH. Effects of regional ischemia on metabolism of glucose and fatty acids. Cir Res 1978; 38(Suppl): 52–74.
Cole SL, Corday E. Four-minute limit for cardiac resuscitation. JAMA 1956; 161:1454–1458.
Eleff SM, Schleien CL, Koehler RC, et al. Brain bioenergetics during cardiopulmonary resuscitation in dogs. Anesthesiology 1992; 76:77–84.
Kompala SD, Babbs CF, Blaho KE. Effect of deferoxamine on late deaths following CPR in rats. Ann Emerg Med 1986; 15:405–407.
Fleischer JE, Lanier WL, Milde JH, Michenfelder JD. Failure of deferoxamine, an iron chelator, to improve neurological outcome following complete ischemia in dogs. Stroke 1987; 18:124–127.
Schanne FA, Kane AB, Young EE, Farber JL. Calcium dependence of toxic cell death: a final common pathway. Science 1979; 206:700–702.
Rehncrona S, Rosen I, Siesjö BK. Excessive cellular acidosis: an important mechanism of neuronal damage in the brain? Acta Physiol Scand 1980; 110:435–437.
Benveniste H. The excitotoxin hypothesis in relation to cerebral ischemia. Cerebrovasc Brain Metab Rev 1991; 3:213–245.
Petito CK, Feldmann E, Pulsinelli WA, Plum F. Delayed hippocampal damage in humans following cardiorespiratory arrest. Neurology 1987; 37:1281–1286.
Leonov Y, Sterz F, Safar P, Johnson DW, Tisherman SA, Oku K. Hypertension with hemodilution prevents multifocal cerebral hypoperfusion after cardiac arrest in dogs. Stroke 1992; 23:45–53.
Cohan SL, Mun SK, Petite J, et al. Cerebral blood flow in humans following resuscitation from cardiac arrest. Stroke 1989; 20:761–765.
Todd NV, Picozzi P, Crockard HA, Russell RR. Reperfusion after cerebral ischemia: influence of duration of ischemia. Stroke 1986; 17:460–466.
Nishijima MK, Koehler RC, Hurn PD, et al. Postischemic recovery rate of cerebral ATP, phosphocreatine, pH, and evoked potentials. Am J Physiol 1989; 257:H1860–H1870.
Buchan AM, Li H, Cho S, Pulsinelli WA. Blockade of the AMPA receptor prevents CA1 hippocampal injury following severe but transient forebrain ischemia in adult rats. Neurosci Letter 1991; 132:255–258.
Wolfson SK Jr, Safar P, Reich H, et al. Dynamic heterogenicity of cerebral hypoperfusion after prolonged cardiac arrest in dogs measured by the stable xenon/CT technique: a preliminary study. Resuscitation 1992; 23:1–20.
Sterz F, Leonov Y, Safar P, et al. Multifocal Cerbral blood flow Xe-CT and global cerebral metabolism after prolonged cardiac arrest in dogs. Reperfusion with open-chest CPR or cardiopulmonary bypasss. Resuscitation 1992; 24:27–47.
Barone FC, Globus MY, Price WJ, et al. Endothelin levels increase in rat focal and global ischemia. J Cereb Blood Flow Metab 1994; 14:337–342.
Kågström E, Smith ML, Siesjö BK. Cerebral circulatory responses to hypercapnia and hypoxia in the recovery period following complete and incomplete cerebral ischemia in the rat. Acta Physiol Scand 1983; 118:281–291.
Obrenovitch TP, Hallenbeck JM. Platelet accumulation in regions of low blood flow during the postischemic period. Stroke 1985; 16:224–234.
Artru AA, Michenfelder JD. Anoxic cerebral potassium accumulation reduced by phenytoin: Mechanism of cerebral protection? Anesth Analg 1981; 60:41.
Hossmann KA, Kleihues P. Reversibility of ischemic brain damage. Arch Neurol 1973; 29:375–384.
Hossmann KA. Treatment of experimental cerebral ischemia. J Cereb Blood Flow Metab 1982; 2: 275–297.
Kleber AG. Resting membrane potential, extracellular potassium activity, and intracellular sodium activity during acute global ischemia in isolated perfused guinea pig hearts. Cir Res 1983; 52:442–450.
Siesjö BK. Pathophysiology and treatment of focal cerebral ischemia. I. Pathophysiology. J Neurosurgery 1992; 77:169–184.
Wahl P, Schousboe A, Honoré T, Drejer J. Glutamate-induced increase in intracellular Ca2+ in cerebral cortex neurons is transient in immature cells but permanent in mature cells. J Neurochem 1989; 53:1316–1319.
Eisner DA, Lederer WJ. Na-Ca exchange: stoichiometry and electrogenecity. Am J Physiol 1985; 248:C189–C202.
Fagg GE. L-glutamate, excitatory amino acid receptors and brain function. Trends Neuro Sciences 1985; 8:207–210.
Olson JE, Evers JA. Correlations between energy metabolism, ion transport, and water content in astrocytes. Can J Physiol Pharmacol 1992; 70:S350–S355.
Kleber AG. Resting membrane potential, extracellular potassium activity, and intracellular sodium activity during acute global ischemia in isolated perfused guinea pig hearts. Cir Res 1983; 52:442–450.
Krause GS, White BC, Aust SD, Nayini NR, Kumar K. Brain cell death following ischemia and reperfusion: a proposed biochemical sequance. Crit Care Med 1988; 16:714–726.
Tosaki A, Hellegouarch A, Braquet P. Cicletanine and reperfusion injury: is there any correlation between arrhythmias, 6-keto-PGF1alpha, thromboxane B2, and myocardial ion shifts (Na+, K+, Ca2+, and Mg2+) induced by ischemia/reperfusion in isolated rat hearts. J Cardiovasc Pharmacol 1991; 17:551–559.
Blaustein MP, Goldman WF, Fontana G, et al. Physiological roles of the sodium-calcium exchanger in nerve and muscle. Ann N Y Acad Sci 1991; 639:254–274.
Benveniste H, Jørgensen MB, Diemer NH, Hansen AJ. Calcium accumulation by glutamate receptor activation is involved in hippocampal cell damage after ischemia. Acta Neurol Scand 1988; 78: 529–536.
Mayer ML, Westbrook GL. Cellular mechanisms underlying excitotoxicity. Trends Neurosci 1987; 10:59–61.
Chan PH, Kerlan R, Fishman RA. Reductions of gamma-aminobutyric acid and glutamate and (Na++K+)-ATPase activity in brain slices and synaptosomes by arachidonic acid. J Neurochem 1983; 40:309–315.
Drejer J, Benveniste H, Diemer NH, Schousboe A. Cellular origin of ischemic-induced glutamate release from brain tissue in vivo and in vitro. J Neurochem 1985; 45:145–150.
Pin JP, Duvoisin R. Neurotransmitter receptors I: The metabotropic glutamate receptors: Structure and functions. Neuropharmacol 1995; 34:1–26.
Samoilov MO, Semenov DG, Tulkova EI, Lazarewicz JW. Early postanoxic changes of polyphosphoinositides and bound Ca2+ content in relation to neuronal activity in brain cortex. Resuscitation 1992; 23:33–43.
Yasuda H, Kishiro K, Izumi N, Nakanishi M. Biphasic liberation of arachidonic and stearic acids during cerebral ischemia. J Neurochem 1985; 45:168–172.
Irvine RF. How is the level of free arachidonic acid controlled in mammalian cells? Biochem J 1982; 204:3–16.
Umemura A. Regional differences in free fatty acid release and the action of phospholipase during ischemia in rat brain. No To Shinkei 1990; 42:979–986.
McCord JM, Oxygen-derived free radicals in postischemic tissue injury. N Engl J Med 1985; 312: 159–163.
Pourcyrous M, Leffler CW, Bada HS, Korones SB, Busiji DW. Brain superoxide anion generation in asphyxiated piglets and effect of indomethacin at therapeutic dose. Pediatr Res 1993; 34:366–369.
Farber JL. The role of calcium in cell death. Life Sci 1981; 29:1289–1295.
Siesjö BK, Bengtsson F. Calcium fluxes, calcium antagonists, and calcium-related pathology in brain ischemia, hypoglycemia, and spreading depression: a unifying hypothesis. J Cereb Blood Flow Metab 1989; 9:127–140.
Erecinska M, Silver IA. Relationship between ions and energy metabolism: cerebral calcium movements during ischaemia and subsequent recovery. Can J Physiol Pharmacol 1992; 70:S190–S193.
Boening JA, Kass IS, Cottrell JE, Chambers G. The effect of blocking sodium influx on anoxic damage in the rat hippocampal slice. Neuroscience 1989; 33:263–268.
Berger JR, Busto R, Ginsberg MD. Verapamil: failure of metabolic amelioration following global forebrain ischemia in the rat. Stroke 1984; 15:1029–1032.
Taguchi J, Graf R, Rosner G, Heiss WD. Prolonged transient ischemia results in impaired CBF recovery and secondary glutamate accumulation in cats. J Cereb Blood Flow Metab 1996; 16:271–279.
Cheung JY, Bonventre JV, Malis CD, Leaf A. Calcium and ischemic injury. N Engl J Med 1986; 314: 1670–1676.
Yoshida S, Ikeda M, Busto R, Santiso M, Martinez E, Ginsberg MD. Cerebral phosphoinositide, triacylglycerol, and energy metabolism in reversible ischemia: origin and fate of free fatty acids. J Neurochem 1986; 47:744.
Ernster L. Oxygen as an environmental poison. Chemica Scripta 1986; 26:525–527.
Bulkley GB. The role of oxygen free radicals in human disease processes. Surgery 1983; 94:407–411.
Krause GS, Nayini NR, White BC, et al. Natural course of iron delocalization and lipid peroxidation following a 15 minute cardiac arrest in dogs. Ann Emerg Med 1987; 16:1200–1205.
Halliwell B, Gutteridge JM. Oxygen toxicity, oxygen radicals, transition metals and disease. Biochem J 1984; 219:1–14.
Okada D. Two pathways of cyclic GMP production through glutamate receptor-mediated nitric oxide synthesis. J Neurochem 1992; 59:1203–1210.
Fridovich I. Superoxide radical: an endogenous toxicant. Ann Rev Pharmacol Toxicol 1983; 23: 239–257.
White BC, Aust SD, Arfros KE, Aronson LD. Brain injury by ischemic anoxia-hypothesis. A tale of two ions? Ann Emerg Med 1984; 13:862–867.
Samdani AF, Dawson TM, Dawson VL. Nitric oxide synthase in models of focal ischemia. Stroke 1997; 28:1283–1288.
Ambrosio G, Weisfeldt ML, Jacobus WE, Flaherty JT. Evidence for a reversible oxygen radical-mediated component of reperfusion injury: reduction by recombinant human superoxide dismutase administered at the time of reflow. Circulation 1987; 75:282–291.
Hall ED, Yonkers PA. Attenuation of postischemic cerebral hypoperfusion by the 21-aminosteroid U74006F. Stroke 1988; 10:340–344.
Hillered L, Ernster L. Respiratory activity of isolated rat brain mitochondria following in vitro exposure to oxygen radicals. J Cereb Blood Flow Metab 1983; 3:207–214.
Weissmann G, Smolen JE, Korchak HM. Release of inflammatory mediators from stimulated neutrophils. N Engl J Med 1980; 303:27–34.
Au AM, Chan PH, Fishman RA. Stimulation of phospholipase A2 activity by oxygen-derived free radicals in isolated brain capillaries. J Cell Biochem 1985; 27:449–453.
Zaleska MM, Wilson DF. Lipid hydroperoxides inhibit reacylation of phospholipids in neuronal membranes. J Neurochem 1989; 52:255–260.
Beckman JS. The double-edged role of nitric oxide in brain function and superoxide-mediated injury. J Vevelopmental Physiol 1991; 15:53–59.
Stamler JS, Singel DJ, Loscalzo J. Biochemistry of nitric oxide and its redox-activated forms. Science 1992; 258:1898–1902.
Bodsch W, Takahashi K, Barbier A, Ophoff B, Hossmann KA. Cerebral protein synthesis and ischemia. Prog Brain Res 1985; 63:197–210.
Nowak TS Jr, Fried RL, Lust D, Passonneau JV. Changes in brain energy metabolism and protein synthesis following transient bilateral ischemia in the gerbil. J Neurochem 1985; 44:487–494.
Dienel GA, Pulsinelli WA, Duffy TE. Regional protein synthesis in rat brain following acute hemispheric ischemia. J Neurochem 1980; 35:1216–1226.
Morimoto K, Yanagihara T. Cerebral ischemia in gerbils: polyribosomal function during progression and recovery. Stroke 1981; 12:105–110.
DeGarcia DJ, O’Neil BJ, Frisch C, et al. Studies of the protein synthesis system in the brain cortex during global ischemia and reperfusion. Resuscitation 1993; 21:161–170.
White BC, Grossman LI, Krause GS. Brain injury by global ischemia and reperfusion: a theoretical perspective on membrane damage and repair. Neurology 1993; 43:1656–1665.
de Haro C, Manne V, de Herreros AG, Ochoa S. Heat-stable inhibitor of translation in reticulocyte lysates. Proc Natl Acad Sci USA 1982; 79:3134–3137.
Merrick WC: Mechanism and regulation of eukaryotic protein synthesis. Microbiol Rev 1992; 56: 291–315.
Burda J, Martín ME, García A, Alcázar A, Fando JL, Salinas M. Phosphorylation of the “subunit of initiation factor 2 correlates with the inhibition of translation following transient cerebral ischaemia in the rat. Biochem J 1994; 302:335–338.
DeGarcia DJ, Neumar RW, White BC, Krause GS. Global brain ischemia and reperfusion: modifications in eukaryotic initiation factors associated with inhibition of translation initiation. J Neurochem 1996; 67:2005–2012.
Rotman EI, Brostrom MA, Brostrom CO. Inhibition of protein synthesis in intact mammalian cells by arachidonic acid. Biochem J 1992; 282:487–494.
Gaitero F, Limas GG, Mendez E, de Haro C. Purification of a novel heat-stable translational inhibitor from rabbit reticulocyte lysates. FEBS Lett 1988; 236:479–483.
Kleihues P, Hossmann KA, Pegg AE, Kobayashi K, Zimmermann V. Resuscitation of the monkey brain after one hour complete ischemia. III. Indications of metabolic recovery. Brain Res 1975; 95:61–73.
Bodsch W, Barbier A, Oehmichen M, Grosse Ophoff BG, Hossmann KA. Recovery of monkey brain after prolonged ischemia. II. Protein synthesis and morphologic alterations. J Cereb Blood Flow Metab 1986; 6:22–33.
Widmann R, Kuroiwa T, Bonnekoh P, Hossman KA. [14C] Leucine incorporation into brain proteins in gerbils after transient ischemia: relationship to selective vulnerability of hippocampus. J Neurochem 1991; 56:789–796.
Dienel GA, Cruz NF, Rosenfeld SJ. Temporal profiles of proteins responsive to transient ischemia. J Neurochem 1985; 44:600–610.
Cattaneo E, McKay R. Proliferation and differentiation of neuronal stem cells regulated by nerve growth factor. Nature 1990; 347:762–765.
Werther GA, Hogg A, Oldfield BJ, et al. Localization and characterization of insulin receptors in rat brain and pituitary gland in vitro autoradiography and computerized densitometry. Endocrinology 1987; 121:1562–1570.
Wanaka A, Jonhson EM Jr, Milbrandt J. Localization of FGF receptor mRNA in the adult rat central nervous system by in situ hybridization. Neuron 1990; 5:267–281.
Koh S, Oyler GA, Higgins GA. Localization of nerve growth factor receptor messenger RNA and protein in the adult rat brain. Exp Neurol 1989; 106:209–221.
Lauterio TJ. Regulation and physiological function of insulin-like growth factors in the central nervous system. Adv Exp Med Biol 1991; 293:419–430.
Plata-Salaman CR. Epidermal growth factor and the nervous system. Peptides 1991; 12:653–663.
DeGarcia DJ, O’Neil BJ, White BC, et al. Insulin induces tyrosine phosphorylation of a 90-kDa protein during postischemic brain reperfusion. Exp Neurol 1993; 124:351–356.
Jørgensen MB, Deckert J, Wright DC, Gehlert DR. Delayed c-fos proto-oncogene expression in the rat brain following transient forebrain ischemia. Brain Res 1989; 484:393–398.
Schiaffonati L, Rappocciolo E, Tacchini L, Cairo G, Bernelli-Zazzera A. Reprogramming of gene expression in post-ischemic rat liver: Induction of protooncogenes and hsp-70 gene family. J Cell Physiol 1990; 143:79–87.
Beckmann RP, Mizzen LE, Welch WJ. Interaction of HSP-70 with newly synthesized proteins: implications for protein folding and assembly. Science 1990; 248:850–854.
Nowak TS Jr. Synthesis of a stress protein following transient ischemia in the gerbil. J Neurochem 1985; 45:1635–1641.
Kanduc D, Mittleman A, Serpico R, et al. Cell death: apoptosis versus necrosis. Int J Oncol 2002; 21: 165–170.
Davis JN, Antonawich FJ. Role of apoptotic proteins in ischemic hippocampal damage. Ann N Y Acad Sci 1997; 835:309–320.
Bredesen DE. Keeping neurons alive: the molecular control of apoptosis. Neuroscientist 1996; 2: 211–216.
Yuan J, Yankner BA. Apoptosis in the nervous system. Nature 2000; 407:802–809.
Nitatori T, Sato N, Waguri S, et al. Delayed neuronal death in the CA1 pyramidal cell layer of the gerbil hippocampus following transient ischemia is apoptosis. J Neurosci 1995; 15:1001–1011.
Vexler ZS, Roberts TP, Bollen AW, Derugin N, Arieff AI. Transient cerebral ischemia. Association of apoptosis induction with hypoperfusion. J Clin Invest 1997; 99:1453–1459.
Wyllie AH, Beattie GJ, Hargreaves AD. Chromatin changes in apoptosis. Histochem J 1981; 13: 681–692.
Wyllie AH. Glucocorticoid-induced thymocyte apoptosis is associated with endogenous endonuclease activation. Nature 1980; 284:555,556.
Hengartner MO. The biochemistry of apoptosis. Nature 2000; 407:770–776.
MacManus JP, Buchan AM, Hill IE, Rasquinha I, Prrston E. Global ischemia can cause DNA fragmentation indicative of apoptosis in rat brain. Neurosci Lett 1993; 164:89–92.
Schutz JB, Weller M, Moskowitz MA. Caspases as treatment targets in stroke and neurodegenerative diseases. Ann Neurol 1999; 45:421–429.
Bredesen DE. Neuronal apoptosis. Ann Neurol 1995; 38:839–851.
Miuai M, Zhu H, Rotello R, Hartwieg EA, Yuan J. Induction of apoptosis in fibroblasts by IL-1 beta-converting enzyme, a mammalian homolg of the C. elegans cell death gene ced-3. Cell 1993; 75: 653–660.
Chen J, Nagayama T, Jin K, et al. Induction of caspase-3-like protease may mediate delayed neuronal death in the hippocampus after transient cerebral ischemia. J Neurosci 1998; 18:4914–4928.
Strasser A, O’Connor L, Dixit VM. Apoptosis signaling. Annu Rev Biochem 2000; 69:217–245.
Hu WH, Johnson H, Shu HB. Tumor necrosis factor-related apoptosis-inducing ligand receptors signal NF-κB and JNK activation and apoptosis through distinct pathways. J Biol Chem 1999; 274: 30,603–30,610.
Ahmad M, Srinivasula SM, Wang L, et al. CRADD, a novel human apoptotic adaptor molecule for caspase-2 and FasL/tumor necrosis factor receptor-interacting protein RIP. Cancer Res 1997; 57: 615–619.
Antonsson B: Bax and other pro-apoptotic Bcl-2 family “killer-proteins” and their victim the mitochondrion. Cell Tissue Res 2001; 306:347–361.
Chopp M, Li Y. Apoptosis in focal cerebral ischemia. Acta Neurochir Suppl 1996; 66:21–26.
Hara H, Friedlander RM, Gagliardini V, et al. Inhibition of interleukin 1 beta converting enxyme family proteaes reduces ischemic and excitotoxic neuronal damage. Proc Natl Acad Sci USA 1997; 94:2007–2012.
Satoh MS, Lindahl T. Role of poly (ADP-ribose) formation in DNA repair. Nature 1992; 356:356–358.
Lindahl T, Satoh MS, Poirier GG, Klungland. Post-translational modification of ploy(ADP-ribose) polymerase induced by DNA strand breaks. Trends Biochem Sci 1995; 20:405–411.
Nicholson DW, Ali A, Thornberry NA, Vaillancourt JP, Dang CK, Gallant M, Gareau Y, Griffin PR, Labelle M, Lazebnik YA. Identification and inhibition of the ICE/CED-3 protease necessary for mammalian apoptosis. Nature 1995; 376:37–43.
Patel T, Gores GJ, Kaufmann SH. The role of proteases during apoptosis. FASEB 1996; 10:587–597.
Eliasson MJL, Sampei K, Mandir AS, et al. Poly (ADP-ribose) polymerase gene disruption renders mice resistant to cerebral ischemia. Nat Med 1997; 3:1089–1095.
Cole KK, Perez-Polo JR. Poly (ADP-ribose) polymerase inhibition prevents both apoptotic-like delayed neuronal death and necrosis after H202 injury. J Neurochem 2002; 82; 19–29.
Enari M, Sakahira H, Yokoyama H, Okawa K, Iwanatsu A, Nagata S. A caspase-activated DNase that degrades DNA during apoptosis, and its inhibitor ICAD. Nature 1998; 391:43–50, Erratum in: Nature 1998; 28:393–396.
Daugas E, Nochy D, Ravagnan L, Loeffler M, Susin SA, Zamzami N, Kroemer G. Apoptosis-inducing factor (AIF): a ubiquitous mitochondrial oxidoreductase involved in apoptosis. FEBS Lett 2000; 476: 118–123.
Li LY, Luo X, Wang X. Endonuclease G is an apoptotic DNase when released from mitochondria. Nature 2001; 412:95–99.
Cao G, Pei W, Lan J, et al. Caspase-activated DNase/DNA fragmentation factor 40 mediates apoptotic DNA fragmentation in transient cerebral ischemia and in neuronal cultures. J Neurosci 2001; 21: 4678–4690.
Chesler M. The regulation and modulation of pH in the nervous system. Prog Neurobiol 1990; 34: 401–427.
Eleff SM, Schleien CL, Koehler RC, et al. Brain bioenergetics during cardiopulmonary resuscitation in dogs. Anesthesiology 1992; 76:77–84. Erratum in anesthesiology 1992; 76:666.
Johnson BA, Weil MH. Redefining ischemia due to circulatory failure as dual defects of oxygen deficit and of carbon dioxide excesses. Crit Care Med 1991; 19:1432–1438.
Duggal C, Weil MH, Gazmuri RJ, et al. Regional blood flow during closed-chest cardiac resuscitation in rats. J Appl Physiol 1993; 74:147–152.
von Planta M, Weil MH, Gazmuri RJ, Bisera J, Rackow EC. Myocardial acidosis associated with CO2 production during cardiac arrest and resuscitation. Circulation 1989; 80:684–692.
Sanders AB, Kern KB, Atlas M, Bragg S, Ewy GA. Importance of the duration of inadequate coronary perfusion pressure on resuscitation from cardiac arrest. J Am Coll Cardiol 1985; 6:113–118.
Hurn PD, Koehler RC, Norris SE, Blizard KK, Traystman RJ. Dependence of cerebral energy phosphate and evoked potential recovery on end-ischemic pH. Am J Physiol 1991; 260:H532–H541.
Maruki Y, Koehler RC, Eleff SM, Traystman RJ. Intracellular pH during reperfusion influences evoked potential recovery after complete cerebral ischemia. Stroke 1993; 24:697–704.
Martin GB, Nowak RM, Paradis N, et al. Characterization of cerebral energetics and brain pH by 31P spectroscopy after graded canine cardiac arrest and bypass reperfusion. J Cereb Blood Flow Metab 1990; 10:221–226.
Welsh FA, Ginsberg MD, Rieder W, Budd WW. Deleterious effect of glucose pretreatment on recovery from diffuse cerebral ischemia in the cat. II. Regional metabolic levels. Stroke 1980; 11:355–363.
Pulsinelli WA, Waldman S, Rawlinson D, Plum F. Moderate hyperglycemia augments ischemic brain damage: a neuropathologic study in the rat. Neurology 1982; 32:1239–1246.
Warner DS, Gionet TX, Todd MM, McAllister AM. Insulin-induced normoglycemia improves ischemic outcome in hyperglycemic rats. Stroke 1992; 23:1775–1781.
Tyson R, Peeling J, Sutherland G. Metabolic changes associated with altering blood glucose levels in short duration forebrain ischemia. Brain Res 1993; 608:288–298.
O’Donnell BR, Bickler PE. Influence of pH on calcium influx during hypoxia in rat cortical brain slices. Stroke 1994; 25:171–177.
Rehncrona S, Hauge HN, Siesjö BK. Enhancement of iron-catalyzed free radical formation by acidosis in brain homogenates: difference in effect by lactic acid and CO2. J Cereb Blood Flow Metab 1989; 9:65–70.
Halliwell B. Reactive oxygen species and the central nervous system. J Neurochem 1992; 59:1609–1623.
Siesjö BK, Bendek G, Koide T, Westerberg E, Weiloch T. Influence of acidosis on lipid peroxidation in brain tissues in vitro. J Cereb Blood Flow Metab 1985; 5:253–258.
Hossmann KA, Hossmann V. Coagulopathy following experimental cerebral ischemia. Stroke 1977; 8:249–253.
Hekmatpanah J. Cerebral blood flow dynamics in hypotension and cardiac arrest. Neurology 1973; 23:174–180.
Böttiger BW, Martin E. Thrombolytic therapy during cardiopulmonary resuscitation and the role of coagulation activation after cardiac arrest. Curr Opin Crit Care 2001; 7:176–183.
Böttiger BW, Motsch J, Böhrer H, et al. Activation of blood coagulation after cardiac arrest is not balanced adequately by activation of endogenous fibrinolysis. Circulation 1995; 92:2573–2578.
Fischer M, Hossmann K. No-reflow after cardiac arrest. Intensive Care Med 1995; 21:132–141.
Lin SR, O’Connor MJ, Fischer HW, King A. The effect of combined dextran and streptokinase on cerebral function and blood flow after cardiac arrest: an experimental study on the dog Invest Radiol 1978; 13:490–498.
Love S, Barber R. Expression of P-seletin and intracellular adhesion molecule-1 in human brain after focal infarction or cardiac arrest. Neuropathol Appl Neurobiol 2001; 27:465–473.
Danton GH, Dietrich WD. Inflammatory mechanisms after ischemia and stroke. J Neuropathol Exp Neurol 2003; 62:127–136.
Schott RJ, Natale JE, Ressler SW, Burney RE, D’Alect LG. Neutrophil depletion fails to improve neurological outcome after cardiac arrest in dogs. Ann Emerg Med 1989; 18:517–522.
Buchan AM, Bruederlin B, Heinicke E, Li H. Failure of the lipid peroxidation inhibitor, U7400GF, to prevent postischemic selective neuronal injury J Cereb Blood Flow Metab 1992; 12:250–256.
Hallenbeck JM, Dutka AJ, Tanishimi T, et al. Polymorphonuclear leukocyte accumulation in brain regions with low blood flow during the early postischemic period. Stroke 1986; 17:246–253.
Dutka AJ, Kochanek P, Francis TJ, Hallenbeck JM. Leukopenia ameliorates multifocal brain ischemia. Neurology [Abstract] 1987; 37(Suppl):249.
Bednar M, Smith B, Pinto A, Mullane KM. Nafazatrom-induced salvage of ischemic myocardium in anesthetized dogs is mediated through inhibition of neutrophil function. Cir Res 1985; 57:131–141.
Crawford MH, Grover FL, Kolb WP, et al. Complement and neutrophil activation in the pathogenesis of ischemic myocardial injury. Circulation 1988; 78:1449–1458.
Kirschfink M: Controlling the complement system in inflammation. Immunopharmacol 1997; 38: 51–62.
Foreman KE, Vaporciyan AA, Borish BK, et al. C5a-induced expression of P-selectin in endothelial cells. J Clin Invest 1994; 94:1147–1155.
Butcher EC. Leukocyte-endothelial cell adhesion recognition: Three (or more) steps to specificity and diversity. Cell 1991; 67:1033–1036.
Czurko A, Nishino H. Appearance of immunoglobulin G and complement factor C3 in the striatum after transient focal ischemia in the rat. Neurosci Lett 1994; 166:51–54.
Nathan C, Sporn M. Cytokines in context. J Cell Biol 1991; 113:981–986.
Kishimoto TK. A dynamic model for neutrophil localization to inflammatory sites. J NIH Res 1991; 3:75–77.
McEver RP. Selectins: novel adhesion receptors that mediate leukocyte adhesion during inflammation. Thromb Haematol 1991; 65:223–229.
Tilton RG, Berens KL. Functional role for selectins in the pathogenesis of cerebral ischemia. Drugs News Perspect 2002; 15:351–357.
Bevilacqua MP, Nelson RM. Selectins J Clin Invest 1993; 91:379–387.
Ghezzi P, Dinarello CA, Bianchi M, Rosandich ME, Repine JE, White CW. Hypoxia increases production of interleukin-1 and tumor-necrosis-factor by mononuclear cells. Cytokine 1991; 3:189–194.
Patel KD, Zimmerman GA, Prescott SM, McEver RP, McIntyre TM. Oxygen radicals induce human endothelial cells to express GMP-140 and bind neutrophils. J Cell Biol 1991; 112:749–759.
Clark WM, Lauten JD, Lessov N, Woodward W, Coull BM. Time course of ICAM-1 expression and leukocyte subset infiltration in rat forebrain ischemia. Mol Chem Neuropathol 1995; 26:213–230.
Springer T. Adhesion receptors of the immune system. Nature 1990; 346:425–434.
Howard EF, Chen Q, Cheng C, Caroll JE, Hess D. NF-Œ∫B and ICAM-1 gene expression is upregulated during reoxygentaion of human brain endothelial cells. Neurosci Lett 1998; 248:199–203.
Lindsberg PJ, Yue TL, Frerichs KU, Hallenbeck JM, Feuerstein G. Evidence for platelet-activating factor as anovel mediator in expiremental stroke in rabbits. Stroke 1990; 21:1452–1457.
Kubes P, Ibbotson G, Russell J, Wallace JL, Granger DN. Role of platelet-activating factor in ischemia/reperfusion-induced leukocyte adherence. Am J Physiol 1990; 259; G300–G305.
Susuki M, Asako H, Kubes P, Jennings S, Grisham MB, Granger DN. Neutrophil-derived oxidants promote leukocyte adherence in postcapillary venules. Microvasc Res 1991; 42:125–138.
Weiss SJ. Tissue destruction by neutrophils. N Engl J Med 1989; 320:365–376.
Ma XL, Lefer DJ, Lefer AM, Rothelein R. Coronary endothelial and cardiac protective effects of a monoclonal antibody to intercellular adhesion molecule-1 in myocardial ischemia and reperfusion. Circulation 1992; 86:937–946.
Dreyer WJ, Michael LH, West MS, DC, Entman ML. Neutrophil accumulation in ischemic myocardium: insights into time course, distribution, and mechanism of localization during early reperfusion. Circulation 1991; 84:400,411.
Lees GJ. The possible contribution of microglia and macrophages to delayed neuronal death after ischemia. J Neurol Sci 1993; 114:119–122.
Wei G, Dawson VL, Zweier JL. Role of neuronal and endothelial nitric oxide synthase in nitric oxide generation in the brain following cerebral ischemia. Biochim Biophys Acta 1999; 1455:23,34.
De Belder AJ, Radomski MW, Why HJ, et al. Nitric oxide synthase activities in human myocardium. Lancet 1993; 341:84,85.
Nozaki K, Moskowitz MA, Maynard KI, et al. Possible origins and distribution of immunoreactive nitric oxide synthase-containing nerve fibers in cerebral arteries. J Cereb Blood Flow Metab 1993; 13:70–79.
Bredt DS, Snyder SH. Isolation of nitric oxide synthase, a calmodulin-requiring enzyme. Proc Natl Acad Sci, USA 1990; 87:682–685.
Fukuyama N, Takizawa S, Ishida H, Hoshiai K, Shinohara Y, Nakazawa H. Peroxynitrite formation in focal cerebral ischemia-reperfusion in rats occurs predominantly in the peri-infarct region. J Cereb Blood Flow Metab 1996; 18:123–129.
Bolanos JP, Almeida A. Roles of nitric oxide in brain hypoxia-ischemia. Biochim Biophys Acta 1999; 1411:415–436.
Ungureanu-Longrois D, Balligand JL, Simmons WW, et al. Induction of nitric oxide synthase activity by cytokines in ventricular myocytes is necessary but not sufficient to decrease contractile responsiveness to $-adrenergic agonists. Circ Res 1995; 77:494–502.
Park SY, Lee H, Hur J, et al. Hypoxia induces nitric oxide production in mouse microglia via p38 mitogen-activated protein kinase pathway. Brain Res Mol Brain Res 2002; 107:9–16.
Tominaga T, Stao S, Ohnishi T, Ohnishi ST. Electron paramagnetic resonance (EPR) detection of nitric oxide produced during focal ischemia in the rat. J Cereb Blood Flow Metab 1994; 14:715–722.
Southam E, East SJ, Garthwaite J. Excitatory amino acid receptors coupled to the nitric oxide/cyclic GMP pathway in rat cerebrellum during development. J Neurochem 1991; 56:2072–2081.
Garthwaite J, Garthwaite G, Palmer RMJ, Moncada S. NMDA receptor activation induces nitric oxide synthesis from arginine in rat brain slices. Eur J Pharmacol 1989; 172:413–416.
Kilbourn RG, Traber DL, Szabo C. Nitric oxide and shock. Dis Mon 1997; 43:277–348.
Radi R, Beckman JS, Bush KM, Freeman BA. Peroxynitrite oxidation of sulfhydryls. J Biol Chem 1991; 266:4244–4250.
Mullie A, Verstringe P, Buylaert W, et al. Predictive value of Glascow coma score for awakening after out-of-hospital cardiac arrest. Cerebral resuscitation study group of the Belgian Society for Intensive care. Lancet 1988; 1:137–140.
Grubb NR, Elton RA, Fox KA. In-hospital mortality after out-of-hospital cardiac arrest. Lancet 1995; 346:417–421.
Teasdale G, Jennett B. Assessment of coma and impaired consciousness: a practical scale. Lancet 1974; 2:81–84.
Kelsey SF, Abramson NS, Detre KM, Monroe J. Brain Resuscitation Clinical Trail I Study Group. A randomized clinical study of cardiopulmonary-cerebral resuscitation: design, methods, and patient characteristics. Am J Emerg Med 1986; 4:72–86.
Levy DE, Caronna JJ, Singer BH, Lapinski RH, Frydman H, Plum F: Predicting outcome from hypoxic-ischemic coma. JAMA 1985; 253:1420–1426.
Berek K, Jeschow M, Aichner F. The prognostication of cerebral hypoxia after out-of-hospital cardiac arrest in adults. Eur Neurol 1997; 37:135–145.
Edgren E, Hedstrand U, Kelsey S, Sutton-Tyrrell K, Safar R. Assessment of neurological prognosis in comatose survivors of cardiac arrest. BRCT I Study Group. Lancet 1994; 343:1055–1059.
Fiser DH, Long N, Roberson PK, Hefley G, Zolten K, Brodie-Fowler M. Relationship of pediatric overall performance category and pediatric cerebral performance category scores at pediatric intensive care unit discharge with outcome measures collected at hospital discharge and 1-and 6-month follow-up assessments. Crit Care Med 2000; 28:2616–2620.
Synek VM. Value of a revised EEG coma scale for prognosis after cerebral anoxia and diffuse head injury. Clin Electroencephalogr 1990; 21:25–30.
Yamashita S, Morinaga T, Ohgo S, Sakamoto T, Kaku N, Sugimoto S, Matsukura S. Prognostic value of electroencephalogram (EEG) in anoxic encephalopathy after cardiopulmonary resuscitation: relationship among anoxic period, EEG grading and outcome. Intern Med 1995; 34:71–76.
Geocadin RG, Sherman DL, Christian Hansen H, et al. Neurological recovery by EEG bursting after resuscitation from cardiac arrest in rats. Resuscitation 2002; 55:193–200.
Jørgensen E, Malchow-Moller A. Natural history of global and critical brain ischemia. Part I: EEG and neurological signs during the first year after cardiopulmonary resuscitation in patients subsequently regaining consciousness. Resuscitation 1981; 9:133–153.
Wijdicks EF, Parisi JE, Sharborough FW. Prognostic value of myoclonus status in comatose survivors of cardiac arrest. Ann Neurol 1994; 35:239–243.
Rothstein TL, Thomas EM, Sumi SM. Predicting outcome in hypoxic-ischemic coma. A prospective clinical and electrophysiological study. Electroencephalogr Clin Neurophysiol 1991; 79:101–107.
Kjos BO, Brant-Zawadzki M, Young RG. Early CT findings of global central nervous system hypoperfusion. Am J Roentgenol 1983; 141:1227–1232.
Roine RO, Raininko R, Erkinjuntti T, Ylikoski A, Kaste M. Magnetic resonance imaging findings associated with cardiac arrest. Stroke 1993; 24:1005–1014.
Cohan SL, Mun SK, Petitie J, Correia J, Tavelra Da Silva AT, Waldhom RE. Cerebral blood flow in humans following resuscitation from cardiac arrest. Stroke 1989; 20:761–765.
Roine RO, Launes J, Nikkinen P, Lindroth L, Kaste M. Regional cerebral blood flow after human cardiac arrest. A hexamethylpropyleneamine oxime single photon emission computed tomographic study. Arch Neurol 1991; 48:625–629.
Barone FC, Clark RK, Price WJ, White RF, Feuerstein GZ, Barone FC. Neuron-specific enolase increases in cerebral and systemic circulation following focal ischaemia. Brain Res 1993; 623: 77–82.
Hachimi-Idrissi S, Van der Auwera M, Schiettecatte J, Ebinger G, Michotte Y, Hughens L. S-100 protein as early predictor of regaining consciousness after out of hospital arrest. Resuscitation 2002; 53:251–257.
Karkela J, Bock E, Kaukinen S. CSF and serum brain-specific creatine kinase isoenzyme (CK-BB), neuron-specific enolase (NSE) and neural cell adhesion molecule (NCAM) as prognostic markers for hypoxic brain injury after cardiac arrest in man. J Neurol Sci 1993; 116:100–109.
Abramson NA, Safar P, Detre KM, Kelsey SF, Monroe J, Reinmuth O, Snyder JV: Neurological recovery after cardiac arrest: effect of duration of ischemia. Crit Care Med 1985; 13:930–931.
Safar P. Resuscitation from clinical death: pathophysiologic limits and therapeutic potentials. Crit Care Med 1988; 16:923–941.
Selman WR, Spetzler RF, Roski RA. Barbiturate resuscitation from focal cerebral ischemia-A review. Resuscitation 1981; 9:189–196.
Nordstrom CH, Rehncrona S, Siesjo BK. Restitution of cerebral energy state, as well as of glycolytic metabolites, citric acid cycle intermediates and associated amino acids after 30 minutes of complete ischemia in rats anesthetized with nitrous oxide or pentobarbital. J Neurochem 1978; 30:479–486.
Todd MM, Chadwick HS, Shapiro HM, Dunlop BJ, Marshall LF, Dueck R. The neurological effects of thiopental therapy following experimental cardiac arrest in cats. Anesthesiology 1982; 57:76–86.
Safar P. Amelioration of postischemic brain damage with barbiturates. Stroke 1980; 11:34–38.
Smith AL, Hoff JT, Nielson SL, Larson CP. Barbiturate protection against cerebral infarction. Stroke 1974; 5:1–7.
Nussmeier NA, Arlund C, Slogoff S. Neuropsychiatric complications after cardiopulmonary bypass: cerebral protection by a barbiturate. Anesthesiology 1986; 64:165–170.
Ward JD, Becker DP, Miller DJ, et al. Failure of prophylactic barbiturate coma in the treatment of severe head trauma. J Neurosurg 1985; 62:383.
Bleyaert AL, Nemoto EM, Safar P, et al. Thiopental amelioration of brain damage after global ischemia in monkeys. Anesthesiology 1978; 49:390–398.
Snyder BD, Ramirez-Lessepas M, Sukhum P, Fryd D, Sung JH. Failure of thiopental to moderate global anoxic injury. Stroke 1979; 10:135.
Rogers MC, Kirsch JR. Current concepts in brain resuscitation. JAMA 261:3143–3147.
Safar P. Brain resuscitation. Special symposium issue. Critical care Med 1978; 6:199–214.
Ebmeyer U, Safar P, Radovsky A, et al. Thiopental combination treatments for cerebral resuscitation after prolonged cardiac arrest in dogs. Exploratory outcome study. Resuscitation 2000; 45:119–131.
Vaagenes P, Cantadore R, Safar P, et al. Amelioration of brain damage by lidoflazine after prolonged ventricular fibrillation cardiac arrest in dogs. Crit Care Med 1984; 12:846–855.
White BC, Winegar CD, Wilson RF, Hoehner PJ, Trombley JH Jr. Possible role of calcium blockers in cerebral resuscitation: A review of the literature and synthesis for future studies. Crit Care Med 1983; 11:202–207.
White BC, Gadzinski DS, Hoehner PJ, et al. Effect of flunarizine on canine cerebral cortical blood flow and vascular resistance post cardiac arrest. Ann Emerg Med 1982; 11:119–126.
Mohamed AA, Mendelow AD, Teasdale GM, Teasdale GM, Harper Am, McCulloch J. Effect of the calcium antagonist nimodipine on local cerebral blood flow and metabolic coupling. J Cereb Blood Flow Metab 1985; 5:26–33.
Steen PA, Gisvold SE, Milde JH, et al. Nimodipine improves outcome when given after complete cerebral ischemia in primates. Anesthesiology 1985; 62:406–414.
Fleischer JE, Lanier WL, Milde JH, Michenfelder JD. Lidoflazine does not improve neurological outcome when administered after complete cerebral ischemia in dogs. Anesthesiology 1987; 66: 304–311.
Calle PA, Paridaens K, De Ridder LI, Buylaert WA. Failure of nimodipine to prevent brain damage in a global brain ischemia model in the rat. Resuscitation 1993; 25:59–71.
Lanza RP, Cooper DK, Barnard CN. Lack of efficacy of high-dose verapamil in preventing brain damage in baboons and pigs after prolonged partial cerebral ischemia. Am J Emerg Med 1984; 2:481–485.
White BC, Winegar CD, Wilson RF, Krause GS. Calcium blockers in cerebral resuscitation. J Trauma 1983; 23:788–794.
Kelsey SF, Sutton-Tyrrell K, Abramson S, et al. A randomized clinical trial of calcium entry blocker administration to comatose survivors of cardiac arrest. Design, methods, and patient characteristics. Brain resuscitation Clinical Trial II Study Group. Control Clin trials 1991; 12:525–545.
Safar P. Cerebral resuscitation after cardiac arrest: research initiatives and future directions. Ann Emerg Med 1993; 2:324–349.
Abramson NS, Kelsey SF, Safar P, Sutton-Tyrell K. Simpson’s paradox and clinical trials: what you find is not necessarily what you prove. Ann Emerg Med 1992; 21:1480–1482.
Roine RO, Kaste M, Kinnunen A, Nikki P, Sarna S, Kajaste S. Nimodipine after resuscitation from out-of-hospital ventricular fibrillation. A placebo-controlled, double-blind randomized trial. JAMA 1990; 264:3171–3177.
Fleischer JE, Tateishi A, Drummond JC, et al. MK-801, an excitatory amino acid antagonist, does not improve neurological outcome following cardiac arrest in cats. J Cereb Blood Flow Metab 1989; 9:795–804.
Schwarcz R, Meldrum B. Excitatory aminoacid antagonists provide a therapeutic approach to neurological disorders. Lancet 1985; 2:140–143.
Muir KW, Grosset DG, Lees KR. Effects of prolonged infusions of the NMDA antagonist aptiganel hydrochloride (CNS 1102) in normal volunteers. Clin Neuropharmacol 1997; 20:311–321.
Gill R, Foster AC, Woodruff GN: Systemic administration of MK-801 protects against ischaemia-induced hippocampal neuroregeneration in the gerbil. J Neurosci 1987; 7:3343–3349.
Gerlach R, Beck M, Zeitschel U, Seifert V: MK 801 attenuates c-Fos and c-Jun expression after in vitro ischemia in rat neuronal cell cultures but not in PC 12 cells. Neurol Res 2002; 24:725–729.
Gill R, Foster A, Woodruff GN. MK-801 is neuroprotective in gerbils when administered during the post-ischemic period. Neurosci 1988; 25:847–855.
Lanier WL, Perkins WJ, Karlsson BR, et al. The effects of dizocilpine melaete (MK-801) an antagonist of the N-methyl-D-aspartate receptor, on neurological recovery and histopathology following complete cerebral ischemia in primates. J Cereb Blood Flow Metab 1990; 10:252–261.
Sterz F, Leonov Y, Safar P, et al. Effect of excitatory amino acid receptor blocker MK-801 on overall and neurological outcome after prolonged cardiac arrest in dogs. Anesthesiology 1989; 71:907–918.
Helfaer MA, Ichord RN, Martin LJ, Hurn PD, Castro A, Traystman RJ. Treatment with the competitive NMDA antagonist GPI 3000 does not improve outcome after cardiac arrest in dogs. Stroke 1998; 29: 824–829.
Nellgård B, Wieloch T. Postischemic blockade of AMPA but not NMDA receptors mitigates neuronal damage in the rat brain following transient severe cerebral ischemia. J Cereb Blood Flow Metab 1992; 12:2–11.
Menniti FS, Buchan AM, Chenard BL, et al. CP-465,022, a selective noncompetitive AMPA receptor antagonist, blocks AMPA receptors but is not neuroprotective in vivo. Stroke 2003; 34:171–176.
Cerchiari EL, Hoel TM, Safar P, Sclabassi RJ. Protective effects of combined superoxide dismutase and deferoxamine on recovery of cerebral blood flow and function after cardiac arrest in dogs. Stroke 1987; 18:869–878.
Vaagenes P, Safar P, Cantadore R, et al. Outcome trails of free radical scavengers and calcium entry blockers after cardiac arrest in two dog models [abstract]. Ann Emerg Med 1986; 15:665.
Natale JE, Schott RJ, Hall ED, Braughler JM. The 21-aminosteroid U74006F reduces systemic lipid peroxidation, improves neurological function, and reduces mortality after cardiopulmonary arrest in dogs. Prog Clin Biol Res 1989; 308:891–896.
Hall ED. Lipid antioxidants in acute central nervous system injury. Ann Emerg Med 1993; 22:1022–1027.
Perkins WJ, Milde LN, Milde JH, Michenfelder JD. Pretreatment with U74006F improves neurological outcome following complete cerebral ischaemia in dogs. Stroke 1991; 22:902–909.
Beck T, Bielenberg GW. Failure of the lipid peroxidation inhibitor U74006F to improve neurological outcome after transient forebrain ischemia in the rat. Brain Res 1990; 532:336–338.
Sterz F, Safar P, Johnson DW, Oku K, Tisherman SA. Effects of U74006F on multifocal cerebral blood flow and metabolism after cardiac arrest in dogs. Stroke 1991; 22:889–895.
Feng Y, LeBlanc MH, LeBlanc EB, et al. Desmethyl tirilazad improves neurological function after hypoxic ischemic brain injury in piglets. Crit Care Med 2000; 28:1431–1438.
Meyer FB, Sundt TM, Yanagihara T, Anderson RE. Focal cerebral ischemia: pathophysiologic mechanisms and rationale for future avenues of treatment. Mayo Clin Proc 1987; 62:35–55.
Palmer C, Roberts RL, Bero C. Deferoxamine posttreatment reduces ischemic brain injury in neonatal rats. Stroke 1994; 25:1039–1045.
Rosenthal RE, Chanderbhan R, Marshall G, Fiskum G. Prevention of post-ischemic brain lipid conjugated diene production and neurological injury by hydroxyethyl starch-conjugated deferoxamine. Free Radic Biol Med 1992; 12:29–33.
Liachenko S, Tang P, Xu Y. Deferoxamine improves early postresuscitation reperfusion after prolonged cardiac arrest in rats. J Cereb Blood Flow Metab 2003; 23:574–581.
Liu TH, Beckman JS, Freeman BA, Hogan EL, Hsu CY. Polyethylene glycol-conjugated dismutase and catalase reduce ischemic brain injury. Am J Physiol 1989; 256:H589–H593.
Itoh T, Kawakami M, Yamauchi Y, Shimizu S, Nakamura M. Effecet of allopurinol on ischemia and reperfusion-induced cerebral injury in spontaneous hypertensive rats. Stroke 1986; 17:1284–1287.
Forsman M, Fleischer JE, Milde JH, Steen PA, Michenfelder JD. Superoxide dismutase and catalase failed to improve neurological outcome after cerebral ischemia in the dog. Acta Anaesthesiol Scand 1988; 32:152–155.
Reich H, Safar P, Angelos M, Basford R, ernster L. Failure of a multifaceted anti-reoxygenation injury (RI) therapy to ameliorate brain damage after ventricular fibrillation (VF) cardiac arrest (CA) of 20 minutes in dogs [abstract]. Crit Care Med 1988; 16:387.
Cerchiari EL, Sclabassi RJ, Safar P, Hoel TM. Effects of combined superoxide dismutase and deferoxamine on recovery of brainstem auditory evoked potentials and EEG after asphyxial cardiac arrest in dogs. Resuscitation 1990,19:25–40.
Shigeno T, Yamasaki Y, Kato G, et al. Reduction of delayed neuronal death by inhibition of protein synthesis. Neurosci Lett 1990; 120:117–119.
Davies AM. The Bcl-2 family of proteins, and the regulation of neuronal survival. Trends Neurosci 1995; 18:355–358.
Chen J, Graham SH, Nakayama M, Zhu RL, Jin K, Stetler RA, Simon RP. Apoptosis repressor genes Bcl-2 and Bcl-x-long are expressed in the rat brain following global ischemia. J Cereb Blood Flow Metab 1997; 17:2–10. Erratum in: J Cereb Blood Flow Metab 1998; 18:931.
Kitagawa K, Matsumoto M, Tsujimoto Y, et al. Amelioration of hippocampal neuronal damage after global ischemia by neuronal overexpression of Bcl-2 in transgenic mice. Stroke 1998; 29:2616–2621.
Chen J, Nagayama T, Jin K, Stetler RA, Zhu RL, Graham SH, Simon RP. Induction of caspase-3-like protease may mediate delayed neuronal death in the hippocampus after transient cerebral ischemia. J Neurosci 1998; 18:4914–4928.
Vogel P, Putten H, Popp E, Krumnikl JJ, et al. Improved resuscitation after cardiac arrest in rats expressing the baculovirus caspase inhibitor protein p35 in central neurons. Anesthesiology 2003; 99:112–121.
Harkema JM, Chaudry IH. Magnesium-adenosine triphosphate in the treatment of shock, ischemia and sepsis. Crit Care Med 1992; 20:263–275.
Fukunaga AF. Intravenous administration of large doses of adenosine or adenosine triphosphate with minimal blood pressure fluctuations. Life Sci 1995; 56:PL209–PL218.
Paskitti M, Reid KH. Use of an adenosine triphosphate-based ‘cocktail’ early in reperfusion substantially improves brain protein synthesis after global ischemia in rats. Neurosci Lett 2002; 331:147–150.
Krep H, Brinker G, Schwindt W, Hossmann KA. Endothelin type A-antagonist improves long-term neurological recovery after cardiac arrest in rats. Crit Care Med 2000; 28:2873–2880.
Krep H, Brinker G, Phillekamp F, Hossman KA. Treatment with an endothelin type A receptor-antagonist after cardiac arrest and resuscitation improves cerebral hemodynamic and functional recovery in rats. Crit Care Med 2000; 28:2866–2872.
Fischer Böttiger BW, Popov-Cenic S, Hossman KA. Thrombolysis using plasminogen activator and heparin reduces cerebral no-reflow after resuscitation from cardiac arrest: an experimental study in the cat. Intensive care Med 1996; 22:1214–1223.
Safar P, Xiao F, Radovsky A, Tanigawa K, et al. Improved cerebral resuscitation from cardiac arrest in dogs with mild hypothermia plus blood flow promotion. Stroke 1996; 27:105–113.
Lin SR, O’Conner MJ, Fischer HW, King A. The effects of combined dextran and streptokinase on cerebral function and blood flow after cardiac arrest: an experimental study on the dog. Invest Radiol 1978; 13:490–498.
Böttiger BW, Bode C, Kern S, et al. Efficacy and safety of thrombolytic therapy after initially unsuccessful cardiopulmonary resuscitation: A prospective clinical trial. Lancet 2001; 357:1583–1585.
Cullen JP, Aldrete JA, Janovsky L, Romo-Salas F. Protective action of phenytoin in cerebral ischemia. Anesth Analg 1979; 58:165–169.
Aldrete JA, Romo-Salas F, Mazzia VD, Tan SL. Phenytoin for brain resuscitation after cardiac arrest: An uncontrolled clinical trial. Crit Care Med 1981; 9:474.
Ebmeyer U, Safar P, Radovsky A, et al. Thiopental combination treatments for cerebral resuscitation after prolonged cardiac arrest in dogs. Exploratory outcome study. Resuscitation 2000; 45:119–131.
Taft WC, Clifton GL, Blair RE, DeLorenzo RJ. Phenytoin protects against ischemia-produced neuronal cell death. Brain Res 1989; 483:143–148.
Kennedy C, Grave GD, Jehle JW. The effect of diphenylhydantoin on local cerebral blood flow. Neurology 1972; 22:451.
Varon S, Hagg T, Manthorpe M. Nerve growth factor in CNS repair and regeneration. Adv Exp Med Biol 1991; 296:267–276.
Mattson MP, Murrain M, Gurthrie PB, Kater SB. Fibroblast growth factor and glutamate: opposing roles in the generation and degeneration of hippocampal neuroarchitecture. J Neurosci 1989; 9: 3728–3740.
Shigeno T, Mima T, Takakura K, Graham DI, Kato G, Hashimoto y, Furukawa S. Amelioration of delayed neuronal death in the hippocampus by nerve growth factor. J Neurosci 1991; 11:2914–2919.
Voll CL, Aver RN. Insulin attenuates ischemic brain damage independent of its hypoglycemic effect: J Cereb Blood Flow Metab 1991; 11:1006–1014.
LeMay DR, Gehua L, Zelenock GB, D’Alecy G. Insulin administration protects neurological function in cerebral ischemia in rats. Stroke 1988; 19:1411–1419.
Katz LM, Wang Y, Ebmeyer U, Radovsky A, Safar P. Glucose plus insulin improves cerebral outcome after asphyxial cardiac arrest. Neuroreport 1998; 9:3363–3367.
Yarden Y. Growth factor receptor tyrosine kinases. Annu Rev Biochem 1988; 57:443–478.
Pillion DJ, Kim SJ, Kim H, Meezan E. Insulin signal transduction: the role of protein phosphorylation. Am J Med 1992; 303:40–52.
Sacks DB, Fujita-Yamaguchi Y, Gale RD, McDonald JM. Tyrosine-specific phosphorylation of calmodulin by the insulin receptor kinase purified from human placenta. Biochem J 1989; 263:803–812.
Moss AM, Unger JW, Moxley RT, Livingston JN. Location of phosphotyrosine-containing proteins by immunocytochemistry in the rat forebrain corresponds to the distribution of the insulin receptor. Proc Natl Acad Sci USA 1990; 87:4453–4457.
Ting LP, Tu CL, Chou CK. Insulin-induced expression of human heat shock protein gene. J Biol Chem 1989; 264:3403–3408.
Parrizas M, Saltiel AR, LeRoith D. Insulin-like growth factor 1 inhibits apoptosis using the phophatidylinositol 3′-kinase and mitogen-activated protein kinase pathways. J Biol Chem 1997; 272:154–161.
Drews G, Debuyser A, Nenquin M, Henquin JC. Galanin and epinephrine act on distinct receptors to inhibit insulin release by the same mechanisms, including an increase in K+ permeability of the $-cell membrane. Endocrinology 1990; 126:1646–1653.
Cryer PE. Physiology and pathophysiology of the human sympathoadrenal neuroendocrine system. N Engl J Med 1980; 303:436–444.
Jansson L. Influence of adrenaline on blood perfusion and vascular conductance of the whole pancreas and the islets of Langerhans in the rat. Arch Int Pharmacodyn 1991; 313:90–97.
Rorsman P, Bokvist K, Ammala C, et al. Activation by adrenaline of a low-conductance G protein-dependent K+ channel in mouse pancreatic B cells. Nature 1991; 349:44–79.
Yu KT, Pessin JE, Czech MP. Regulation of insulin receptor kinase by multisite phosphorylation. Biochime 1985; 67:1081–1090.
Sotomatsu A, Nakano M, Hirai S. Phospholipid peroxidation induced by the catechol-Fe3+ (Cu2+) complex: a possible mechanism of nigrostriatal cell damage. Arch Biochem Biophys 1990; 283: 334–341.
Callaham M, Madsen CD, Barton CW, Saunders CE, Pointer J. A randomized clinical trial of high-dose epinephrine and norepinephrine in prehospital cardiac arrest. JAMA 1992; 268:2667–2672.
Markov AK, Oglethorpe N, Grillis M, Neely WA, Hellems HK. Therapeutic action of fructose-1,6-diphosphate in traumatic shock. World Surg 1983; 7:430–406.
Vexler ZS, Wong A, Francisco C, et al. Fructose-1, 6-biphosphate preserves intracellular glutathione and protects cortical neurons against oxidative stress. Brain Res 2003; 960:90–98.
Woodhall B, Kramer RS, Currie WD, Sanders AP. brain energetics and neurosurgery. A review of recent studies done at Duke University. J Neurosurgery 1971; 34:3–14.
Furuichi Y, Katsuta K, Maeda M, et al. Neuroprotective action of tracolimus (FK506) in focal and global cerebral ischemnia in rodents: dose dependency, therapeutic time window and long-term efficacy. Brain Res 2003; 965:137–145.
Katsura K, Kurihara J, Hiraide T, Takahashi K, Kato H, Katayama Y. Effects of FK506 on the translocation of protein kinase C and CaM kinase II in the gerbil hippocampal CA1 neurons. Neurol Res 2003; 25:522–527.
Kirsmer AC, Linder KH, Wenzel V, Rainer B, Muller G, Lingnau W. Inhibition of nitric oxide improves coronary perfusion pressure and return of spontaneous circulation in a porcine cardiopulmonary resuscitation model. Crit Care Med 2001; 29:482–486.
Sasaki T, Hamada J, Shibata M, Araki N, Fukuuchi Y. Inhibition of nitric oxide production during global ischemia ameliorates ischemic damage of pyramidal neurons in the hippocampus. Keio J Med 2001; 50:182–187.
Ditchey RV, Winkler JV, Rhodes CA. Relative lack of coronary blood flow during closed-chest resuscitation in dogs. Circulation 1982; 66:297–302.
Paradis NA, Martin GB, Rivers EP, et al. Coronary perfusion pressure and the return of spontaneous circulation in human cardiopulmonary resuscitation. JAMA 1990; 263; 1106–1113.
Sanders AB, Kern KB, Berg RA, Hilwig RW, Heidenrich J, Ewy GA. Survival and neurological outcome after cardiopulmonary resuscitation with four different chest compression-ventilation ratios. Ann Emerg Med 2002; 40:553–562.
Ralston SH, Babbs CF, Niebauer MJ. Cardiopulmonary resuscitation with interposed abdominal compression in dogs. Anesth Analg 1982; 61:645–651.
Walker JW, Bruestle JC, White BC, Evans AT, Indreri R, Bialek H. Perfusion of the cerebral cortex by use of abdominal counterpulsation during cardiopulmonary resuscitation. Am J Emerg Med 1984; 2:391–393.
Howard M, Carruba C, Foss F, Janiak B, Hogan B, Guinness M. Interposed abdominal compression-CPR: its effects on parameters of coronary perfusion in human subjects. Ann Emerg Med 1987; 16: 253–259.
Berryman CR, Phillips GM. Interposed abdominal compression-CPR in human subjects. Ann Emerg Med 1984; 13:226–229.
Mateer J, Steuven HA, Thompson BM, Aprahamian C, Darin JC. Pre-hospital IAC-CPR versus standard CPR: Paramedic resuscitation of cardiac arrests. Am J Emerg Med 1985; 3:143–146.
Sack JB, Kesselbrenner MB, Bregman D. Survival from in-hospital cardiac arrest with interposed abdominal counter pulsation during cardiopulmonary resuscitation JAMA 1992; 267:379–385.
Sack JB, Kesselbrenner MB, Jarrad A. Interposed abdominal compression-cardiopulmonary resuscitation and resuscitation outcome during asystole and electromechanical dissociation. Circulation 1992; 86:1692–1700.
Mateer JR, Stueven HA, Thompson BM., et al. Interposed abdominal compression CPR versus standard CPR in prehospital cardiopulomnary arrest: preliminary results. Ann Emerg Med 1984; 13:764–766.
Weiser FM, Adler LN, Kuhn LA. Hemodynamic effects of closed and open chest cardiac resuscitation in normal dogs, and those with acute myocardial infarction. Am J Cardiol 1962; 10:555–561.
Del Guercio LR, Feins NR, Cohn JD, Coomaraswamy RP, Wollman SB, State D. Comparison of blood flow during external and internal cardiac massage in man. Circulation 1965; 31/32(Suppl I):I-171–I-180.
Sanders AB, Kern K, Ewy GA, Atlas M, Bailey L. Improved resuscitation from cardiac arrest with open-chest massage. Ann Emerg Med 1984; 13:672.
Fleisher G, Sagy M, Swedlow DB, Belani K. Open-versus closed-chest cardiac compression in a canine model of pediatric cardiopulmonary resuscitation. Am J Emerg Med 1985; 3:305–310.
Kern KB, Sanders AB, Badylak SF, et al. Long-term survival with open-chest cardiac massage after ineffective closed-chest compression in a canine preparation. Circulation 1987; 75:498–503.
Arai T, Dote K, Tsukahara I, Nitta K, Nagaro T. Cerebral blood flow during conventional, new and open-chest cardio-pulmonary resuscitation in dogs. Resuscitation 1984; 12:147–154.
Geehr EC, Lewis, Auerbach PS. Failure of open heart massage to improve survival after pre-hospital non-traumatic cardiac arrest. N Engl J Med 1986; 314:1189,1190.
Halperin HR, Guerci AD, Chandra N, et al. Vest inflation without simultaneous ventilation during cardiac arrest in dogs: improved survival from prolonged cardiopulmonary resuscitation. Circulation 1986; 74:1407–1415.
Halperin HR, Tsitlik JE, Gelfand M, et al. A preliminary study of cardiopulmonary resuscitation by circumferential compression of the chest with use of a pneumatic vest. N Engl J Med 1993; 329:762–768.
Lurie KG. Active compression-decompression CPR: a progress report. Resuscitation 1994; 28: 115–122.
Lindner KH, Pfenninger EG, Lurie KG, Schürmann W, Lindner IM, Ahnefeld FW. Effects of active compression-decompression resuscitation on myocardial and cerebral blood flow in pigs. Circulation 1993; 88:1254–1263.
Lurie K, Zielinski T, McKnite S, Sukhum P. Improving the efficiency of cardiopulmonary resuscitation with an inspiratory impedance threshold valve. Crit Care Med 2000; 28:N207–N209.
Voelckel WG, Lurie KG, Zielinski T, et al. The effects of positive end-expiratory pressure during active compression decompression cardiopulmonary resuscitation with the inspiratory threshold valve. Anesth Analg 2001; 92:967–974.
Lurie KG, Coffeen P, Shultz J, McKnite S, et al. Improving active compression-decompression cardiopulmonary resuscitation with an inspiratory impedance valve. Circulation 1995; 91:1629–1632.
Lurie K, Zielinski T, McKnite S, Aufderheide T, Voelckel W. Use of an inspiratory impedance valve improves neurologically intact survival in a porcine model of ventricular fibrillation. Circulation 2002; 105:124–129.
Plaisance P, Lurie KG, Payen D. Inspiratory impedance during active compression-decompression cardiopulmonary resuscitation: a randomized evaluation in patients in cardiac arrest. Circulation 2000; 101:989–994.
Wolcke BB, Mauer DK, Schoefmann MF, et al. Standard CPR versus active compression-decompression CPR with an impedance threshold valve in patients with out of hospital cardiac arrest [Abstract]. Resuscitation 2002; 55:115.
Cohen TJ, Goldner BG, Maccaro PC, et al. Comparison of active compression-decompression cardiopulmonary resuscitation with standard cardiopulmonary resuscitation for cardiac arrests occurring in the hospital. N Engl J Med 1993; 329:1918–1921.
Lurie KG, Shultz JJ, Callaham ML, et al. Evaluation of active compression-decompression CPR in victims of out-of-hospital cardiac arrest. JAMA 1994; 271:1405–1411.
Schwab TM, Callaham ML, Madsen CD, Utecht TA. A randomized clinical trial of active compression-decompression CPR vs standard CPR in out-of-hospital cardiac arrest in two cities. JAMA 1995; 273:1261–1268.
Stiell IG, Hébert PC, Wells GA, et al. The Ontario trial of active compression-decompression cardiopulmonary resuscitation for in-hospital and prehospital cardiac arrest. JAMA 1996; 275:1417–1423.
Plaisance P, Lurie K, Vicaut E, et al. Comparison of standard cardiopulmonary resuscitation and active compression-decompression for out-of-hospital cardiac arrest. N Engl J Med 1999:341:569–575.
Mauer DK, Nolan J, Plaisance P, et al. Effect of active compression-decompression resuscitation (ACD-CPR) on survival: a combined analysis using individual patient data. Resuscitation 1999; 41:249–256.
Babbs CF, Weaver JC, Ralston S, Geddes LA. Cardiac, thoracic, and abdominal pump mechanisms in cardiopulmonary resuscitation: studies in an electrical model of the circulation. Am J Emerg Med 1984; 2:299–308.
Tang W, Weil MH, Schock RB, et al. Phased chest and abdominal compression-decompression. A new option for cardiopulmonary resuscitation. Circulation 1997; 95:1335–1340.
Sterz FBW, Berzanovich A. Active compression-decompression of thorax and abdomen (Lifestick CPR) in patients with cardiac arrest [Abstract]. Circulation 1996; 94:19.
Arntz H, Agrawal R, Richter A, et al. Phased chest and abdominal compression-decompression versus conventional cardiopulmonary resuscitation in out-of-hospital cardiac arrest. Circulation 2001; 104: 768–772.
Adams JA, Bassuk J, Wu D, Kurlansky P. Survival and normal neurological outcome after CPR with periodic GZ acceleration and vasopressin. Resuscitation 2003; 56:215–221.
Adams JA, Mangino MJ, Bassuk J, Kurlansky P, Sackner MA. Novel CPR with periodic Gz acceleration. Resuscitation 2001; 51:55–62.
Reich H, Angelos M, Safar P, Sterz F, Leonov Y. Cardiac resuscitability with cardiopulmonary bypass after increasing ventricular fibrillation times in dogs. Ann Emerg Med 1990; 19:887–890.
Angelos MG, Gaddis ML, Gaddis GM, Leasure JE. Improved survival and reduced myocardial necrosis with cardiopulmonary bypass reperfusion in a canine model of coronary occlusion and cardiac arrest. Ann Emerg Med 1990; 19:1122–1128.
Angelos MG, Ward KR, Hobson J, Beckley PD. Organ blood flow following cardiac arrest in a swine low flow cardiopulmonary bypass model. Resuscitation 1994; 27:245–254.
Gazmuri RJ, Weil MH, von Planta M, Gazmuri RR, Shah DM, Rackow EC. Cardiac resuscitation by extracorporeal circulation after failure of convention CPR. J Lab Clin Med 1991; 118:65–73.
Safar P, Abramson NS, Angelos M, et al. Emergency cardiopulmonary bypass for resuscitation from prolonged cardiac arrest. Am J Emerg Med 1990; 8:55–67.
Levine R, Gorayeb M, Safar P, Abramson N, Stezoski W, Kelsey S. Cardiopulmonary bypass after cardiac arrest in prolonged closed-chest CPR in dogs. Ann Emerg Med 1987; 16:620–627.
Martin GB, Nowak RM, Carden DL, Eisiminger RA, Tomlanovich MC. Cardiopulmonary bypass versus CPR as treatment for prolonged canine cardiopulmonary arrest. Ann Emerg Med 1987; 16:628–636.
Safar P, Abramson NS, Angelos M, et al. Emergency cardiopulmonary bypass for resuscitation from prolonged cardiac arrest. Am J Emerg Med 1990; 8:55–67.
Reichman RT, Joyo CI, Dembitsky WP, et al. Improved patient survival after cardiac arrest using a cardiopulmonary support system. Ann Thorac Surg 1990; 49; 101–105.
Phillips SJ, Zeff RH, Kongtahworn C, et al. Percutaneous cardiopulmonary bypass: application and indication for use. Ann Thorac Surg 1989; 47:121–123.
Martin GB, Rivers EP, Paradis NA, Goetting MG, Morris DC, Nowak RM. Emergency department cardiopulmonary bypass in the treatment of human cardiac arrest. Chest 1998; 113:743–751.
Iijima T, Bauer R, Hossmann KA. Brain resuscitation by extracorporeal circulation after prolonged cardiac arrest in cats. Intensive Care Med 1993; 19:82–88.
del Nido PJ, Dalton HJ, Thompson AE, Siewers RD. Extracorporeal membrane oxygenator rescue in children during cardiac arrest after cardiac surgery. Circulation 1992; 86(5 Suppl 2):II300–II304.
Walcott GP, Booker RG, Ideker RE. Defibrillation with a minimally invasive direct cardiac device. Resuscitation 2002; 55:301–307.
Buckman RF Jr, Badellino MM, Eynon AC, et al. Open-chest cardiac massage without major thoracotomy: metabolic indicators of coronary and cerebral perfusion. Resuscitation 1997; 34:247–253.
Paiva EF, Kern KB, Hilwig RW, Scalabrini A, Ewy GA. Minimally invasive direct cardiac massage versus closed-chest cardiopulmonary resuscitation in a porcine model of prolonged ventricular fibrillation cardiac arrest. Resuscitation 2000; 47: 287–299.
Tang W, Weil MH, Noc M, Sun S, Gazmuri RJ, Bisera J. Augmented efficacy of external CPR by intermittent occlusion of the ascending aorta. Circulation 1993; 88: 1916–1921.
Nozari A, Rubertsson S, Wiklund L. Improved cerebral blood supply and oxygenation by aortic balloon occlusion combined with intra-aortic vasopressin administration during experimental cardiopulmonary resuscitation. Acta Anaesthesiol Scand 2000; 44:1209–1219.
Liu XL, Nozari S, Basu G, Ronquist S, Rubertsson S, Wiklund L. Neurological outcome after experimental cardiopulmonary resuscitation: a result of delayed and potentially treatable neuronal injury? Acta Aneasthesiol Scand 2002; 46:537–546.
Nozari A, Rubertsson S, Gedeborg R, Nordgren A, Wiklund L. Maximisation of cerebral blood flow during experimental cardiopulmonary resuscitation does not ameliorate post-resuscitation hypoperfusion. Resuscitation 1999; 40:27–35.
Reed RL, Johnston TD, Chen Y, Fischer RP. Hypertonic saline alters plasma clotting times and platelet aggregation. J Trauma 1991; 31:8–14.
Nolte D, Bayer M, Lehr HA, et al. Attenuation of postischeamic microvascular disturbances in striated muscle by hyperosmolar saline dextran. Am J Physiol 1992; 263:1411–1416.
Steinbauer M, Harris A, Hoffman T, Messmer K. Pharmacologic effects of dextrans on the postischemic leukocyte-endothelial interaction. Prog Appl Microcirc 1996; 22:114–125.
Otto CW, Yakaitis RW, Blitt CD. Mechanism of action of epinephrine and resuscitation from asphyxial arrest. Crit Care Med 1981; 9:364,365.
Brown CG, Taylor RB, Werman HA, Luu T, Ashton J, Hamlin RL. Myocardial oxygen delivery/consumption during cardiopulmonary resuscitation: a comparison of epinephrine and phenylephrine. Ann Emerg Med 1988; 17:302–308.
Ditchey RV, Lindenfeld J. Failure of epinephrine to improve the balance between myocardial oxygen supply and demand during closed-chest resuscitation in dogs. Circulation 1988; 78:382–389.
Ditchey RV, Rubio-Perez A, Slinker BK. Beta-adrenergic blockade reduces myocardial injury during experimental cardiopulmonary resuscitation. J Am Coll Cardiol 1994; 24:804–812.
Midei MG, Sugiura S, Maughan, L Sagawa K, Weisfeldt ML, Guerci AD. Preservation of ventricular function by treatment of ventricular fibrillation with phenylephrine. J Am Coll Cardiol 1990; 16: 489–494.
Michael JR, Guerci AD, Koehler RC, et al. Mechanism by which epinephrine augments cerebral and myocardial perfusion during cardiopulmonary resuscitation in dogs. Circulation 1984; 69:822–835.
Koide T, Wieloch TE, Siesjö BK. Circulating catecholamines modulate ischemic brain damage. J Cereb Blood Flow Metab 1986; 6:559–565.
Chase PB, Kern KB, Sanders AB, Otto CW, Ewy GA. Effects of greater doses of epinephrine on both non-invasive and invasive measures of myocardial perfusion in blood flow during cardiopulmonary resuscitation. Crit Care Med 1993; 21:413–419.
Brown CG, Werman HA, Davis EA, Hamlin R, et al. Comparative effect of graded doses of epinephrine on regional brain blood flow during CPR in a swine model. Ann Emerg Med 1986; 15:1138–1144.
Menegazzi JJ, Davis EA, Yealy DM, et al. An experimental algorithm versus standard advanced cardiac life support in a swine model of out-of-hospital cardiac arrest. Ann Emerg Med 1993; 22:235–239.
Berkowitz ID, Gervais H, Schleien CL, Koehler RC, Dean JM, Traystman RJ. Epinephrine dosage effects on cerebral and myocardial blood flow in an infant swine model of cardiopulmonary resuscitation. Anesthesiol 1991; 75:1041–1050.
Steill IG, Hebert MD, Weitzman BN, et al. High dose epinephrine in adult cardiac arrest. N Engl J Med 1992; 327:1045–1050.
Brown CG, Martin DR, Pepe PE, et al. A comparison of standard-dose and high-dose epinephrine in cardiac arrest outside the hospital. The multicenter high-dose epinephrine study group. N Engl J Med 1992; 327:1051–1055.
Vandycke C, Martens P. High dose versus standard dose epinephrine in a cardiac arrest-a meta-analysis. Resuscitation 2000; 45:161–166.
Gedeborg R, Silander HC, Ronne-Engstrom E, Rubertsson S, Wiklund L. Adverse effects of high-dose epinephrine on cerebral blood flow during experimental cardiopulmonary resuscitation. Crit Care Med 2000; 28:1423–1430.
Roberts D, Landolfo K, Dobson K, Light RB. The effects of methoxamine and epinephrine on survival and regional distribution of cardiac output in dogs with prolonged ventricular fibrillation. Chest 1990; 98:999–1005.
Klouche K, Weil MH, Sun S, Tang W, Zhao DH. A comparison of “-methylnorepinephrine, vasopressin and epinephrine for cardiac resuscitation. Resuscitation 2003; 57:93–100.
Weil MH, Bisera J, Trevino RP, Rackow EC. Cardiac output and end-tidal carbon-dioxide. Crit Care Med 1985; 13:907–909.
Wenzel V, Lindner KH, Krismer AC, et al. Survival with full neurological recovery and no cerebral pathology after prolonged cardiopulmonary resuscitation with vasopressin in pigs. J Am Coll Cardiol 2000; 35:527–533.
Prengel AW, Lindner KH, Keller A. Cerebral oxygenation during cardiopulmonary resuscitation with epinephrine and vasopressin in pigs. Stroke 1996; 27:1241–1248.
Voelckel WG, Lurie KG, McKnite S, et al. Comparison of epinephrine and vasopressin in a pediatric porcine model of asphyxial cardiac arrest. Crit Care Med 2000; 28:3777–3783.
Voelckel WG, Lurie KG, McKnite S, et al. Effects of epinephrine and vasopressin in a piglet model of prolonged ventricular fibrillation and cardiopulmonary resuscitation. Crit Care Med 2002; 30:957–962.
Lurie KG, Voelckel WG, Iskos DN, et al. Combination drug therapy with vasopressin, adrenaline (epinephrine) and nitroglycerin improves vital organ blood flow in a porcine model of ventricular fibrillation. Resuscitation 2002; 54:187–194.
Stadlbauer KH, Wagner-Berger HG, Wenzel V, et al. Survival with full neurological recovery after prolonged cardiopulmonary resuscitation with a combination of vasopressin and epinephrine in pigs. Anesth Analg 2003; 96:1743–1749.
Stiell IG, Hebert PC, Wells GA, et al. Vasopressin versus epinephrine for inhospital cardiac arrest: a randomised controlled trial. Lancet 2001; 358:105–109.
Koehler RC, Michael JR. Cardiopulmonary resuscitation, brain blood flow, and neurological recovery. Crit Care Clin 1985; 1:205–222.
Rivers EP, Rady MY, Martin GB, e H, Appelton T, Nowak RM. Venous hyperoxia after cardiac arrest: characterization of a defect in systemic oxygen utilization. Chest 1992; 102:1787–1793.
Kuisma M, Suominen P, Korpela R. Paediatric out-of-hospital cardiac arrests-epidemiology and outcome. Resuscitation 1995; 30:141–150.
Liberthson RR. Sudden death from cardiac causes in children and young adults. N Engl J Med 1996; 334:1039–1044.
Takeda T, Tanigawa K, Tanaka H, Hayashi Y, Goto E, Tanaka K. The assessment of three methods to verify tracheal tube placement in the emergency setting. Resuscitation 2003; 56:153–157.
Ornato JP, Shipley JB, Racht EM, et al. Multicenter study of a portable, hand-size, colorimetric endtidal carbon dioxide detection device. Ann Emerg Med 1992; 21:518–523.
Spaulding CM, Joly LM, Rosenberg A, et al. Immediate coronary angioraphy in survivors of out-of-hospital cardiac arrest N Engl J Med 1997; 336:1629–1633.
Tenaglia AN, Califf RM, Candela RJ, et al. Thrombolytic therapy in patients requiring cardiopulmonary resuscitation. Am J Cardiol 1991; 68:1015–1019.
Wijdicks EF, Parisi JE, Sharborough FW. Prognostic value of myoclonus status in comatose survivors of cardiac arrest. Ann Neurol 1994; 35:239–243.
Buyleart WA, Calle PA, Houbrechts HN. Serum electrolyte disturbances in the post-resuscitation period. The Cerebral resuscitation Study Group. Resuscitation 1989; 17(Suppl):S189–S206.
Vukmir RB, Bircher NG, Radovsky A, Safar P. Sodium bicarbonate may improve outcome in dogs with brief or prolonged cardiac arrest. Crit care Med 1995; 23; 515–522.
Emerman CL, Pinchak AC Hagen JF, Hancock D. A comparison of venous blood gases during cardiac arrest. Am J Emerg Med 1988; 6:580–583.
Tucker KJ, Idris AH, Wenzek V, Orban DJ. Changes in arterial and mixed venous blood gases during untreated ventricular fibrillation and cardiopulmonary resuscitation. Resuscitation 1994; 28:137–141.
Idris AH, Staples ED, O’Brian D, et al. Effect of ventilation on acid-base balance and oxygenation during low blood flow states. Crit Care Med 1994; 22:1827–1834.
Weil MH, Rackow EC, Trevino R, Grundler W, Falk JL, Griffel MI. Difference in acid-base state between venous and arterial blood during cardiopulmonary resuscitation. N Engl J Med 1986; 315:153–156.
Leong EC, Bendall JC, Boyd AC, Einstein R. Sodium bicarbonate improves the chance of resuscitation after 10 minutes of cardiac arrest in dogs. Resuscitation 2001; 51:309–315.
Filley G, Kindig N: Carbicarb, an alkalizing ion generating agent of possible clinical usefulness. Trans Am Clin Climatol Assoc 1984; 96:141–153.
Shapiro J, Whalen M, Kucera R, Kindig N, Filley GF, Chan L. Brain pH responses to sodium bicarbonate during systemic acidosis. Am J Physiol 1989; 256:H1316–H1321.
Katz LM, Wang Y, Rockoff S, Bouldin TW. Low-dose Carbicarb improves cerebral outcome after asphyxial cardiac arrest in rats. Ann Emerg Med 2002; 39:359–365.
Liu X, Nozari A, Rubertsson S, Wiklund L. Buffer administration during CPR promotes cerebral reperfusion after return of spontaneous circulation and mitigates post-resuscitation cerebral acidosis. Resuscitation 2002; 55:45–55.
Wiklund L, Ronquist G, Stjernström H, Waldenström A. Effects of alkaline buffer administration on survival and myocardial energy metabolism in pigs subjected to ventricular fibrillation and closed chest CPR. Acta Anaesthesiol Scand 1990; 34:430–439.
von Planta M, Gudipati CV, Weil MH, Kraus LJ, Rackow EC. Effects of tromethamine and sodium bicarbonate buffers during cardiac resuscitation. J Clin Pharmacol 1988; 28:594–599.
De Feo P, Perriello G, De Cosmos S, et al. Comparison of glucose counterregulation during short-term and prolonged hypoglycemia in normal humans. Diabetes 1986; 35:563–569.
Siesjö BK, Ingvar M, Pelligrino D. Regional differences in vascular autoregulation in the rat brain in severe insulin-induced hypoglycemia. J Cereb Blood Flow Metab 1983; 3:478–485.
Ginsberg MD, Welsh FA, Budd WW. Deleterious effect of glucose pretreatment on recovery from diffuse cerebral ischemia in the cat. Stroke 1980; 11:347–354.
Nakakimura K, Fleischer JE, Drummond JC, et al. Glucose administration before cardiac arrest worsens neurological outcome in cats. Anesthesiology 1990; 72: 1005–1011.
Katz LM, Wang Y, Ebmeyer U, Radovsky A, Safar P. Glucose plus insulin infusion improves cerebral outcome after asphyxial cardiac arrest. Neuroreport 1998; 9:3363–3367.
Müllner M, Sterz F, Binder M, Schreiber W, Deimel A, Laggner AN. Blood glucose concentration after cardiopulmonary resuscitation influences functional neurological recovery in human cardiac arrest survivors. J Cereb Blood Flow Metab 1997; 17:430–436.
Longstreth WT Jr, Diehr P, Cobb LA, Hanson RW, Blair AD. Neurological outcome and blood glucose levels during out-of-hospital cardiopulmonary resuscitation. Neurology 1986; 36:1186–1191.
Schultz CH, Rivers EP, Feldkamp CS, et al. A characterization of hypothalamic-pituitary-adrenal axis function during and after human cardiac arrest. Crit Care Med 1993; 21:1339–1347.
Katsura K, Kurihara J, Siesjö BK, Wieloch T. Acidosis enhances translocation of protein kinase C but not Ca (2+)/calmodulin-dependent protein kinase II to cell membranes during complete cerebral ischemia. Brain Res 1999; 849:119–127.
Martin GB, O’Brien JF, Best R, Goldman J, Tomlanovich MC, Nowak RM. Insulin and glucose levels during CPR in the canine model. Ann Emerg Med 1985; 14:293–297.
Budihardjo I, Oliver H, Lutter M, Luo X, Wang X. Biochemical pathways of caspase activation during apoptosis. Annu Rev Cell Dev Biol 1999; 15:269–290.
Rehncrona S, Rosen I, Siesjö BK. Excessive cellular acidosis: an important mechanism of neuronal damage in the brain? Acta Physiol Scand 1980; 110:435–437.
Rehncrona S, Rosen I, Siesjö BK. Brain lactic acidosis and ischemic cell damage: 1. Biochemistry and neurophysiology. J Cereb Blood Flow Metab 1981; 1:297–311.
Rehncrona S, Hauge HN, Siesjö BK. Enhancement of iron-catalyzed free radical formation by acidosis in brain homogenates: Difference in effect by lactic acid and CO2. J Cereb Blood Flow Metab 1989; 9:65–70.
Dietrich D, Alonso O, Busto R. Moderate hyperglycemia worsens acute blood-brain barrier injury after forebrain ischemia in rats. Stroke 1993; 24:111–115.
Schurr A, Payne RS, Tseng MT, Miller JJ, Rigor BM. The Glucose paradox in cerebral ischemia. New insights. Ann NY Acad Sci 1999; 386:386–390.
Schurr A, Payne RS, Miller JJ, Tseng MT. Preischemic hyperglycemia-aggravated damage: evidence that lactate utilization is beneficial and glucose-induced corticosterone is detrimental. J Neurosci Res 2001; 66:782–789.
Rayne RS, Teseng MT, Schurr A. The glucose paradox of cerebral ischemia: evidence for corticosterone involvement. Brain Res 2003; 971:9–17.
Rello J, Valles J, Jubert P, et al. Lower respiratory tract infections following cardiac arrest and cardiopulmonary resuscitation. Clin Infect Dis 1995; 21:310–314.
Dohi S. Postcardiopulmonary resuscitation pulmonary edema. Crit Care Med 1983; 11:434–437.
Liu Y, Rosenthal RE, Haywood Y, Miljkovic-Lolic M, Vanderhoek JY, Fiskum G. Normoxic ventilation after cardiac arrest reduces oxidation of brain lipids and improves neurological outcome. Stroke 1998; 29:1679–1686.
Zwemer CF, Whitesall SE, D’Alecy LG. Hypoxic cardiopulmonary-cerebral resuscitation fails to improve neurological outcome following cardiac arrest in dogs. Resuscitation 1995; 29:225–236.
Fercakova A, Marsala M, Marsala J. Influence of graded postischemic reoxygenation on reperfusion alterations in rabbit dorsal root ganglion neurons. J Hisrnforsch 1994; 35:295–302.
Rootwelt T, Loberg EM, Moen A, Oyasaeter S, Saugstad OD. Hypoxemia and reoxygentaion with 21% or 100% oxygen in newborn pigs: changes in blood pressure, base deficit, and hypoxanthine and brain morphology. Pediatr Res 1992; 32:107–113.
Klaus S, Heringlake M, Gliemroth J, Pagel H, Staubach K, Bahlmann L. Biochemical tissue monitoring during hypoxia and reoxygenation. Resuscitation 2003; 56:299–305.
Ernster L: Biochemistry of reoxygenation injury. Crit care Med 1988; 16:947–953.
Douzinas EE, Patsouris E, Kypriades EM, et al. Hypoxaemic reperfusion ameliorates the histopathological changes in the pig brain after a severe global cerebral ischaemic insult. Intensive Care Med 2001; 27:905–910.
Kutzsche S, Ilves P, Kirkeby OJ, Saugstad OD. Hydrogen peroxide production in leukocytes during cerebral hypoxia and reoxygenation with 100% or 21% oxygen in newborn piglets. Pediatr Res 2001; 49:834–842.
Zwemer CF, Whitesall SE, D’Alecy LG. Cardiopulmonary-cerebral resuscitation with 100% oxygen exacerbates neurological dysfunction following nine minutes of normothermic cardiac arrest in dogs. Resuscitation 1994; 27:159–170.
Lipinski CA, Hicks SD, Callaway CW. Normoxic ventilation during resuscitation and outcome from asphyxial cardiac arrest in rats. Resuscitation 1999; 42:221–229.
Dietrich WD. Morphological manifestations of reperfusion injury in brain. Ann N Y Acad Sci 1994; 723:15–24.
Feet BA, Gilland E, Groenendaal F, et al. Cerebral excitatory amino acids and Na+, K+-ATPase activity during resuscitation of severely hypoxic newborn piglets. Acta Paediatr 1998; 87:889–895.
Vanicky I, Marsala M, Murar J, Marsala J. Prolonged postischemic hyperventilation reduces acute neuronal damage after 15 min of cardiac arrest in the dog. Neurosci Lett 1992; 135:167–170.
Muizelaar JP, Marmarou A, Ward JD, Kontos HA, Choi SC, Becker DP, Gruemer H, Young HF: Adverse effects of prolonged hyperventilation in patients with severe head injury: a randomized clinical trial. J Neurosurg 1991; 75:731–739.
Safar P, Xiao F, Radovsky A, et al. Improved cerebral resuscitation from cardiac arrest in dogs with mild hypothermia plus blood flow promotion. Stroke 1996; 27:105–113.
Rosner MJ, Daughton S. Cerebral perfusion pressure management in head injury. J Trauma 1990; 30:933–941.
Ligas JR, Mosleshi F, Epstein MA. Occult positive end-expiratory pressure with different types of mechanical ventilators. J Crit Care 1990; 52:95–100.
Krakovsky M, Rogatsky G, Zarchin N, Mayevsky A. Effect of hyperbaric oxygen therapy on survival after global cerebral ischemia in rats. Surg Neurol 1998; 49:412–416.
Kapp JP, Phillips M, Markov A, Smith RR. Hyperbaric oxygen after circulatory arrest: modification of postischemic encephalopathy. Neurosurg 1982; 11:496–499.
Iwatsuki N, Takahashi M, Ono K, Tajima T. Hyperbaric oxygen combined with nicardipine administration accelerate neurological recovery after cerebral ischemia in a canine model. Crit Care Med 1992; 20:858–863.
Miljkovic-Lolic M, Silbergleit R, Fiskum G, Rosenthal RE. Neuroprotective effects of hyperbaric oxygen treatment in experimental focal cerebral ischemia are associated with reduced brain leukocyte myeloperoxidase activity. Brain Res 2003; 971:90–94.
Thom SR. Functional inhibition of leukocyte B2 integrins by hyperbaric oxygen in carbon monoxide-mediated brain injury in rats. Toxicol Appl Pharmacol 1993; 123:248–256.
Mink RB, Dutka AJ. Hyperbaric oxygen after cerebral ischemia in rabbits reduces brain vascular permeability and blood flow. Stroke 1995; 26:2307–2312.
Wada K, Miyazawa LJ. Inflammatory cell adhesion molecules in ischemic cerebrovascular disease. Stroke 2002; 173:168–181.
Yin W, Badr AE, Mychaskiw G, Zhang JH. Down regulation of COX-2 is involved in hyperbaric oxygen treatment in a rat transient focal cerebral ischemia model. Brain Res 2002; 926:165–171.
Gunther A, Manaenko A, Franke H, Dickel T, Berrouschot J, Wagner A, Illes P, Reinhardt R. Early biochemical and histological changes during hyperbaric or normobaric reoxygenation after in vitro ischaemia in primary corticoencephalic cell cultures of rats. Brain Res 2002; 946:130–138.
Rusyniak DE, Kirk MA, May JD, et al. Hyperbaric oxygen therapy in acute ischemic stroke: results of the Hyperbaric Oxygen in Acute Ischemic Stroke Trial Pilot Study. Stroke 2003; 34:571–574.
Rosenthal RE, Silbergleit R, Hof PR, Haywood Y, Fiskum G. Hyperbaric oxygen reduces neuronal death and improves neurological outcome after canine cardiac arrest. Stroke 2003; 34:1311–1316.
McKinley BA, Morris WP, Parmley CL, Butler BD. Brain parenchyma PO2, PCO2, and pH during and after hypoxic, ischemic brain insult in dogs. Crit Care Med 1996; 24:1858–1868.
Bogaert YE, Sheu KF, Hof PR, et al. Neuronal subclass-selective loss of pyruvate dehydrogenase immunoreactivity following canine cardiac arrest and resuscitation. Exp Neurol 2000; 161:115–126.
ACC/AHA Task Force Report. Guidelines for early management of patients with acute myocardial infarction. J Am Coll Cardiol 1990; 16:249–292.
Rivers EP, Wortsman J, Rady M, Blake HC, McGeorge FT, Buderer NM. The effect of the total cumulative epinephrine dose administered during human CPR on hemodynamic, oxygen transport, and utilization variables in the postresuscitation period. Chest 1994; 106:1499–1507.
Tang W, Weil MH, Sun SJ, Gazmuri RJ, Bisera J. Progressive myocardial dysfunction after cardiac resuscitation. Crit Care Med 1993; 21:1046–1050.
Charlat ML, O’Neill PG, Hartley CJ, Roberts Bolli R. Prolonged abnormalities of the left ventricular diastolic wall thinning in the “stunned” myocardium in conscious dogs: time course and relation to systolic function. J Am Coll Cardiol 1989; 13:185–194.
Rady MY, Rivers EP, Martin GB, Smithline H, Appelton T, Nowak RM. Continuous central venous oximetry and shock index in the emergency department: use in the evaluation of clinic shock. Am J Emerg Med 1992; 10:538–541.
Stoddard MF, Longaker RA. The safety of transesophageal echocardiography in the elderly. Am Heart J 1993; 125:1358–1362.
Rivers EP, McGeorge FT, Boczar ME. A hemodynamic comparison of mechanical, standard, and active compression-decompression CPR in human cardiac arrest [Abstract]. Clin Intensive Care 1994; 5:S30.
Rivers EP, Rady MY, Martin BG, et al. Venous hyperoxia after cardiac arrest. Chest 1992; 102: 1787–1793.
Rivers EP, Wortsman J, Rady MY, Blake HC, McGeorge FT, Buderer NM. The effects of the total cumulative epinephrine dose administered during human CPR on hemodynamics, oxygen transport, and utilization in the postresuscitation period. Chest 1994; 106:1499–1507.
Bernard GR, Sopko G, Cerra F, et al. Pulmonary artery catheterization and clinical outcomes: National Heart, Lung, and Blood Institute and Food and Drug Administration Workshop Report. Consensus Statement. JAMA 2000; 283:2568–2572.
Meyer RJ, Kern KB, Berg RA, Hilwig RW, Ewy GA. Post-resuscitation right ventricular dysfunction: delineation and treatment with dobutamine. Resuscitation 2002; 55:187–191.
Voelckel WG, Lindner KH, Wenzel V, et al. Effect of small-dose dopamine on mesenteric blood flow and renal function in a pig model of cardiopulmonary resuscitation with vasopressin. Anesth Analg 1999; 89:1430–1436.
Leir CV. Regional blood flow response to vasodilators and inotropes in congestive heart failure. Am J Cardiol 1988; 25:75–83.
Figulla HR. Circulatory support devices in clinical cardiology. Current concepts. Cardiology 1994; 84:149–155.
Jaffe AS. The use of antiarrhythmics in advanced cardiac life support. Ann Emerg Med 1993; 22:307–316.
DiMarco JP, Miles W, Akhtar M, et al. Adenosine for paraxosmal supraventricular tachycardia: dose ranging and comparison with verapamil: assessment in placebo-controlled, multicenter trials: the Adenosine for PSVT Study Group. Ann Intern Med 1990; 113:104–110.
Hashimi-Idrissi S, Corne L, Huyghens L. The effect of mild hypothermia and induced hypertension on long term survival rate and neurological outcome after asphyxial cardiac arrest in rats. Resuscitation 2001; 49:73–82.
Sterz F, Leonov Y, Safar P, Radovsky A, Tisherman SA, Oku K. Hypertension with or without hemodilution after cardiac arrest in dogs. Stroke 1990; 21:1178–1184.
Spivey WH, Abramson NS, Safar P. Correlation of blood pressure with mortality and neurological recovery in comatose postresuscitation patients [abstract]. Ann Emerg Med 1991; 20:453.
Ames A 3rd, Wright RL, Kowada M, Thurston JM, Majno G. Cerebral ischemia. The no-reflow phenomenon. Am J Pathol 1968; 52:437–453.
Smrcka M, Horky M, Otevrel F, Kuchtickova S, Kotola V, Muzik J. The onset of apoptosis of neurons induced by ischemia-reperfusion injury is delayed by transient period of hypertension in rats. Physiol Res 2003; 52:117–122.
Hossman KA. Resuscitation potentials after proilonged global ischemia in cats. Crit Care Med 1988; 16:964–971.
Hashimi-Idrissi S, Corne L, Huyghens L. The effect of mild hypothermia and induced hypertension on long term survival rate and neurological outcome after asphyxial cardiac arrest in rats. Resuscitation 2001; 49:73–82.
Eriksson M, Saldeen T. Effect of dextran on plasma tissue plasminogen activator (t-PA) and plasminogen activator inhibitor-1 (PAI-1) during surgery. Acta Anaesthesiol Scand 1995; 39:163–166.
Krieter H, Denz C, Janke C, et al. Hypertonic-hyperoncotic solutions reduce the release of cardiac troponin I and s-100 after successful cardiopulmonary resuscitation in pigs. Anesth Analg 2002; 95:1031–1036.
Safar P, Sterz F, Leonov Y, Radovsky A, Tisherman S, Oku K. Systematic development of cerebral resuscitation after cardiac arrest. Three promising treatments: cardiopulmonary bypass, hypertensive hemodilution, and mild hypothermia. Acta Neurochir Suppl 1993; 57:110–121.
Klatzo I. Brain edema following brain ischaemia and the influence of therapy. Br J Anaesth 1985; 57:18–22.
Bleyaert AL, Sands PA, Safar P, et al. Augmentation of postischemic brain damage by severe intermittent hypertension. Crit Care Med 1980; 8:41–47.
Mullner M, Sterz F, Binder M, Hellwagner K, Meron G, Herkner H, Laggner AN. Arterial blood pressure after human cardiac arrest and neurological recovery. Stroke 1996; 27:59–62.
Sasse HC, Safar P, Kelsey SF. Arterial hypertension after cardiac arrest is associated with good cerebral outcome in patients [Abstract]. Crit Care Med 1999; 27:A29.
Hickey RW, Kochanek PM, Ferimer H, Graham SH, Safar P. Hypothermia and hyperthermia in children after resuscitation from cardiac arrest. Pediatrics 2000; 106:118–122.
Hickey RW, Kochanek PM, Ferimer H, Alexander HL, Garman RH, Graham SH. Induced hyperthermia exacerbates neuronal histologic damage after asphyxial cardiac arrest in rats. Crit Care Med 2003; 31:531–555.
Zeiner A, Holzer M, Sterz F, Schorkhuber W, Eisenberger P, Uray T, Behringer W. Hyperthermia after cardiac arrest is associated with an unfavorable neurological outcome. Arch Intern Med 2001; 161:2007–2012.
Morris MC, Nadkarni VM. Temperature regulation after cardiac arrest: Timing is everything! Crit Care Med 2003; 31:654,655.
Gaussorgues P, Gueugniaud PY, Vedrinne JM, Salord F, Mercatello A, Robert D. Bacteremia following cardiac arrest and cardiopulmonary resuscitation. Intensive Care Med 1988; 14:575–577.
Sweeney MS, Cooley DA, Reul GJ, Ott DA, Duncan JM. Hypothermic circulatory arrest for cardiovascular lesions: technical considerations and results. Ann Thorac Surg 1985; 40:498–503.
Weiss SJ, Muñiz AE, Ernst AA, Lippton HL, Nick TG. The Effect of Prior Hypothermia on the Physiological Response to Norepinephrine. Resuscitation 2000; 45:201–207.
Steen PA, Soule EH, Michenfelder JD. Detrimental effect of prolonged hypothermia in rats and monkeys with and without regional cerebral ischemia. Stroke 1979; 10:522–529.
Rohrer MJ, Natale AM. Effect of hypothermia on the coagulation cascade. Crit Care Med 1992; 20:1402–1405.
Ji Y, Lui J. Numerical studies on the effect of lowering temperature on the oxygen transport during brain hypothermia resuscitation. Comput Biol Med 2002; 32:495–514.
Rosomoff HL. Protective effects of hypothermia against pathological processes of the nervous system. Ann N Y Acad Sci 1959; 80:475–486.
Benson DW, Williams GR, Spencer FC. The use of hypothermia after cardiac arrest. Anesth Analg 1958; 38:213–245.
Weinrauch V, Safar P, Tisherman S, Kuboyama K, Radovsky A. Beneficial effect of mild hypothermia and detrimental effect of deep hypothermia after cardiac arrest in dogs. Stroke 1992; 23: 1454–1462.
Holzer M, Behringer W, Schorkhuber W, et al. Hypothermia for Cardiac Arrest (HACA) Study Group. Mild hypothermia and outcome after CPR. Acta Anaesthsiol Scand Suppl 1997; 111:55–58.
Leonov Y, Sterz F, Safar P, Radovsky A, Oku K, Tisherman S, Stezoski SW. Mild cerebral hypothermia during and after cardiac arrest improves neurological outcome in dogs. J Cereb Blood Flow Metab 1990; 10:57–70.
Sterz F, Safar P, Tisherman S, Radovsky A, Kuboyama K, Oku K. Mild hypothermic cardiopulmonary resuscitation improves outcome after prolonged arrest in dogs [see comments]. Crit Care Med 1991; 19:379–389.
Busto R, Dietrich WD, Globus MY, Valdes I, Scheinberg P, Ginsberg MD. Small differences in intraischemic brain temperature critically determine the extent of ischemic neuronal injury. J Cereb Blood Flow Metab 1987; 7:729–738.
Xiao F, Safar P, Radovsky A. Mild protective and resuscitative hypothermia for asphyxial cardiac arrest in rats. Am J Emerg Med 1998; 16:17–25.
Wass CT, Lanier WL, Hofer RE, Scheithauer BW, Andrews AG. temperature changes of > 1 C alter functional neurological outcome and histopathology in a canine model of complete cerebral ischemia. Anesthesiology 1995; 83:325–335.
Rosomoff HL, Holaday DA. Cerebral blood flow and cerebral oxygen consumption during hypothermia. Am J Physiol 1954; 179:85–88.
Kuboyama K, Safar P, Oku KL, et al. Mild hypothermia after cardiac arrest in dogs does not affect postarrest cerebral oxygen uptake/delivery mismatching. Resuscitation 1994; 27:231–244.
Oku K, Sterz F, Safar P, et al. Mild hypothermia after cardiac arrest in dogs does not affect postarrest multifocal cerebral hypoperfusion. Stroke 1993; 24:1590–1597.
Busto R, Globus MY, Dietrich D, Martinez E, Valdes I, Ginsberg MD. Effect of mild hypothermia on ischemic-induced release of neurotransmitters and fatty acids in rat brain. Stroke 1989; 20: 904–910.
Kumar K, Wu X, Evans AT, Marcoux F. The effects of hypothermia on induction of heat shock proteins (HSP)-72 in ischemic brain. Metab Brain Dis 1995; 10:283–291.
Kamme F, Campbell K, Wieloch T. Bipahsic expression of the fos and jun families of transcription factors following transient forebrain ischemia in the rat. Effect of hypothermia. Eur J neurosci 1995; 7:2007–2016.
Winfree CJ, Baker CJ, Connolly ES Jr, Fiore AJ, Solomon RA. Mild hypothermia reduces penumbal glutamate levels in the rat permanent focal cerebral ischemia model. Neurosurg 1996; 38:1216–1222.
Kristian T, Katsura K, Siesjö BK. The influence of moderate hypothermia on cellular calcium uptake in complete ischemia: implications for the excitotoxic hypothesis. Acta Physiol Scand 1992; 146: 531,532.
Kil HY, Zhang J, Piantadosi CA. Brain temperature alters hydroxyl radical production during cerebral ischemia/reperfusion in rats. J Cereb Blood Flow Metab 1996; 16:100–106.
Taft WC, Yang K, Dixon CE, Clifton GL, Hayes RL. Hypothermia attenuates the loss of hippocampal microtubule-associated protein 2 (MAP2) following traumatic brain injury. J Cereb Blood Flow Metab 1993; 13:796–802.
Hicks SD, DeFranco DB, Callaway CW. Hypothermia during reperfusion after asphyxial cardiac arrest improves functional recovery and selectively alters stress-induced protein expression. J Cereb Blood Flow Metab 2000; 20:520–530.
Dempsey RJ, Combs DJ, Maley ME, Cowen DE, Roy MW, Donaldson DL. Moderate hypothermia reduces postischemic edema development and leukotriene production. Surgery 1987; 21:177–181.
Ginsburg MD, Busto R, Castella Y. The protective effect of moderate intraischemia brain hypothermia is associated with improved postischemic glucose utilization [abstract]. J Cereb Blood Flow Metab 1989; 9:S380.
D’Cruz BJ, Fertig KC, Filiano A, Hicks SD, DeFranco DB. Hypothermic reperfusion after cardiac arrest augments brain-derived neurotrophic factor activation. J Cereb Blood Flow Metab 2002; 22: 843–851.
Shibano T, Morimoto Y, Kemmotsu O, Shikama H, Hisano K, Hua Y: Effects of mild and moderate hypothermia on apoptosis in neuronal PC12 cells. Br J Anaesth 2002; 89: 301–305.
Marion DW, Leonov Y, Ginsberg M, et al. Resuscitative hypothermia. Crit Care Med 1996; 24: S81–S89.
Bernard SA, Jones BM, Horne MK: Clinical trial of induced hypothermia in comatose survivors of out-of-hospital cardiac arrest. Ann Emerg Med 1997; 30:146–153.
Bernard SA, Gray TW, Buist MD, Jones BM, Silvester W, Gutteridge G, Smith K. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med 2002; 346:557–563.
The Hypothermia After Cardiac Arrest Study Group. Mild therapeutic hypothermia to improve the neurological outcome after cardiac arrest. N Engl J Med 2002; 346:549–556.
Schwab S, Schwarz S, Spranger M, Keller E, Bertram M, Hacke W. Moderate hypothermia in the treatment of patients with severe middle cerebral artery infarction. Stroke 1998; 29:2461–2466.
Hachimi-Idrissi S, Corne L, Ebinger G, Michotte Y, Huyghens L. Mild hypothermia induced by a helmet device: a clinical feasibility study. Resuscitation 2001; 51:275–281.
Zeiner A, Holzer M, Sterz, F, et al. Mild resuscitative hypothermia to improve neurological outcome after cardiac arrest. A clinical feasibility trial. Hypothermia After cardiac Arrest (HACA) Study Group. Stroke 2000; 31:86–94.
Felberg RA, Krieger DW, Chuang R, et al. Hypothermia after cardiac arrest: feasibility and safety of an external cooling protocol. Circulation 2001; 104:1799–1804.
Gunn AJ, Gluckman PD, Gunn TR. Selective head cooling in newborn infants after perinatal asphyxia: a safety study. Pediatrics 1998; 102:885–892.
Rajek A, Greif R, Sessler DL, Baumgardner J, Laciny S, Bastanmehr H. Core cooling by central venous infucion of ice-cold (4EC and 20EC) fluid: isolation of core and peripheral thermal compartments. Anesthesiology 2000; 93:629–637.
Brenard S, Buist M, Monteiro O, Smith K. Induced hypothermia using large volume, ice-cold intravenous fluid in comatose survivors of out-of-hospital cardiac arrest: A preliminary report. Resuscitation 2000; 56:9–13.
Xiao F, Safar P, Alexander H. Peritoneal cooling for mild cerebral hypothermia after cardiac arrest in dogs. Resuscitation 1995; 30:51–59.
Ao H, Moon JK, Tanimoto H, Sakanashi Y, Terasaki H. Jugular vein temperature reflects brain temperature during hypothermia. Resuscitation 2000; 45:111–118.
Henker RA, Brown SD, Marion DW. Comparison of brain temperature with bladder and rectal temperatures in adults in adults with severe head injury. Neurosurgey 1998; 42:1071–1075.
Ao H, Tanimoto H, Yoshitake A, Moon JK, Terasaki H. Long-term mild hypothermia with extracorporeal lung and heart assist improves survival from prolonged cardiac arrest in dogs. Resuscitation 2001; 48:163–174.
Dietrich WD, Busto R, Alonso O, Globus MY, Ginsberg MD. Intraischemic but not postischemic brain hypothermia protects chronically following global forebrain ischemia in rats. J Cereb Blood Flow Metab 1993; 13:541–549.
Colubourne F, Li H, Buchan AM. Indefatigable CA1 sector neuroprotection with mild hypothermia induced 6 hours after severe forebrain ischemia in rats. J Cereb Blood Flow Metab 1999; 19:742–749.
Marion DW, Penrod LE, Kelsey SF, et al. Treatment of traumatic brain injury with moderate hypothermia. N Engl J Med 1997; 336:540–546.
Pomeranz S, Safar P, Radovsky A, Tisherman SA, Alexander H, Stezoski W. The effect of resuscitative moderate hypothermia following epidural brain compression on cerebral damage in a canine outcome model. J Neurosurg 1993; 79:241–251.
Ebmeyer U, Safar P, Radovsky A, Obrist W, Alexander H, Pomeranz S. Moderate hypothermia for 48 hours after temporary epidural brain compression injury in a canine outcome model. J Neurotrauma 1998; 15:323–336.
Nolan JP, Morley PT, Vanden Hoek TL, et al. ALS Task Force: Therapeutic hypothermia after cardiac arrest. An advisory statement by the Advanced Life Support Task Force of the International Liaison Committee on Resuscitation. Circulation 3002; 108:118–122.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2005 Humana Press Inc., Totowa, NJ
About this chapter
Cite this chapter
Muñiz, A.E. (2005). Postresuscitation Cerebral Dysfunction. In: Ornato, J.P., Peberdy, M.A. (eds) Cardiopulmonary Resuscitation. Contemporary Cardiology. Humana Press. https://doi.org/10.1385/1-59259-814-5:523
Download citation
DOI: https://doi.org/10.1385/1-59259-814-5:523
Publisher Name: Humana Press
Print ISBN: 978-1-58829-283-4
Online ISBN: 978-1-59259-814-4
eBook Packages: MedicineMedicine (R0)