Abstract
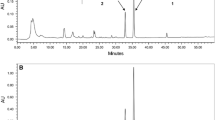
“Aescin”, the saponin mixture obtained from the seeds of Aesculus hippocastanum L. (horse chestnut), is widely used for the treatment of peripheral vascular disorders. This mixture of saponins has been reported to show antiinflammatory activity [1–3] and recently structure-requirements of escins for this activity were obtained by Matsuda et al [4] by investigating the effects of the pure saponins on acute inflammation in experimental animal models.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Vogel, G., Marek, M.L. and Stoeckert, I. (1965) Weitere Untersuchungen zum Wirkungsmechanismus des Rosskastanien-Saponins Aescin, Arzneim.Forsch 13, 59–64.
Vogel, G., Marek, M.L. and Oertner, R. (1970) Untersuchungen zum Mechanismus der therapeutischen und toxischen Wirkung des Rosskastanien-Saponins Aescin, Arzneim.Forsch 20, 699–700.
Rothkopf, M. and Vogel, G. (1976) Neue Befunde zum Wirkungsmechanismus des RosskastanienSaponins Aescin, Arzneim.Forsch 26, 225–235.
Matsuda, H., Li, Y., Murakami, T., Ninomiya, K., Araki, N., Yoshikawa, M. and Yamahara, Y. (1997) Antiinflammatory effects of escins Ia, Ib, IIa, and IIb from horse chestnut, the seeds of Aesculus hippocastanum L., Bioorganic & Medicinal Chemistry Letters 7 (13), 1611–1616.
Deutsches Arzneibuch (1997) (DAB 1997), Rosskastaniensamen, Deutscher Apotheker Verlag Stuttgart, Govi-Verlag-Pharmazeutischer Verlag GmbH, Eschbom,.
Wagner, H., Reger, H. and Bauer, R. (1985) Saponinhaltige Drogen und Fertigarzneimittel. HPLC- Analyse am Beispiel von Rosskastaniensamen, Deutsche Apotheker Zeitung 125 (30), 1513–1518.
Profumo, P., Caciglia, A.M., Gastaldo, P. and Dameri, R.M. (1991) Aescin Content in Embryogenic Callus and in Embryoids from Leaf Explains of Aesculus hippocastanum, Planta Medica 57, 50–52.
Pietta, P. Mauri, P., Facino, R.M. and Carini, M. (1989) High-performance liquid chromatographic analysis of (3-escin, J. of Chromatography 478, 259–263.
Hammerstein, F. and Kaiser, F. (1972) Quantitative Direktauswertung von Arzneipflanzen-extrakten auf Dünnschichtchromatogrammen, Planta Medica. 21, 5–15.
Vanhaelen, M. and Vanhaelen-Fastre, R. (1983) Quantitative determination of biologically active constituents in medicinal plant crude extracts by thin-layer chromatography-densitometry, J Chromatography 281, 263–271.
Uberti, E., Martinelli, E.M. and Pifferi, G. (1990) TLC-densitometric analysis of Aescin in ointments, Fitoterapia 61 (1), 57–60.
Yoshikawa, M., Harada, E., Murakami, T., Matsuda, H., Wariishi, N., Yamahara, J., Murakami, N. and Kitagawa, I. (1994) Bioactive Saponins and Glycosides. III. Horse Chestnut. (1): The Structures, Inhibitory Effects on Ethanol Absorption, and Hypoglycemic Activity of Escins Ia, Ib, IIa, IIb, and IIIa from the Seeds of Aesculus hippocastanum L, Chem. Pharm. Bull. 42, 1357–1359.
Sindambiwe, J.B., Calomme, M., Geerts, S., Pieters, L., Vlietinck, A. and Vanden Berghe, D. (1998) Evaluation of biological activities of triterpenoid saponins from Maesa lanceolata, J. Nat. Prod. 61, 585–590.
Foriers, A., Hemeryck, A., Vander Heyden, Y. and Vercruysse A. (1996) 44th Annual Congress of the Society for Medicinal Plant Research, Prague (Czech Republic) abstract P258.
Vydac, HPLC - Columns and Separation Materials, Vydac/The Separation Group, Hesperia (C.A.), U.S.A., (1994–1995).
Vydac, The Handbook of Analysis and Purification of Peptides and Proteins by Reversed Phase HPLC, Vydac/The Separation Group, Hesperia (C.A.), U.S.A., 1994–1995.
Beutler J.A. (1997) Separation of high molecular weight saponins of Archidendron ellipticum by hydrophilic interaction chromatography, J. Liq. Chrom. & Rel. Technol. 20, 2415–2426.
Regnier F.E. (1987) HPLC of Biological Macromolecules: The First Decade, Chromatogr. 24, 24 1251.
Lewis, R.V., Fallon, A., Stein, S., Gibson, K.D. and Volenfriend, S. (1980) Supports for Reverse-Phase High-Performance Liquid Chromatography of Large Proteins, Analytical Biochemistry 104, 153–159.
Brandt, A., Mann, G. and Arlt W. (1997) Temperature gradients in preparative high-performance liquid chromatography columns, J. Chromatogr. A 769 109–117.
Welsch, T., Schmid, M., Kutter, J. and KâIman, A. (1996) Temperature of the eluent: a neglected tool in high-performance liquid chromatography, J. Chromatogr. A 728, 299–306.
Waters, Symmetry Shield TM Application Notebook, Waters Corporation, Massachusetts, U.S.A., 1998.
Foriers, A. and Vercruysse A., (1997) 45th Annual Congress of the Society for Medicinal Plant Research, Regensburg (Germany) abstract E03.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2000 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Apers, S., Pieters, L., Vlietinck, A., Vercruysse, A., Foriers, A. (2000). Single Run HPLC Separation of Escins Ia and IIa by Means of a Waters Symmetry Shield C8 Column. In: Oleszek, W., Marston, A. (eds) Saponins in Food, Feedstuffs and Medicinal Plants. Proceedings of the Phythochemical Society of Europe, vol 45. Springer, Dordrecht. https://doi.org/10.1007/978-94-015-9339-7_6
Download citation
DOI: https://doi.org/10.1007/978-94-015-9339-7_6
Publisher Name: Springer, Dordrecht
Print ISBN: 978-90-481-5341-1
Online ISBN: 978-94-015-9339-7
eBook Packages: Springer Book Archive