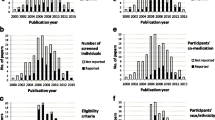
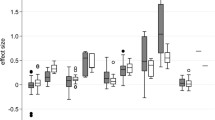
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Ueie K, Kanai T, Hashimoto Y et al. (1992). The general situation of mortality rate in 1990. Kosei no Shihyo, 39(5):22–33 (in Japanese).
Office of Population Censuses and Surveys. (1991). Review of the Registrar General on deaths by cause, sex and age, in England and Wales, 1990. In: Mortality Statistics,Cause. HMSO, London.
British Hospital Index. (1991). International Medical Statistics, Ltd., Middlesex.
British Pharmaceutical Index. (1991). International Medical Statistics, Ltd., Middlesex.
Japan Pharmaceutical Market. (1991). IMS Japan, Ltd., Tokyo.
Japan Medical Data Index. (1991). IMS Japan, Ltd., Tokyo.
Matsumoto K, Saito R, Nagahama F et al. (1981). A comparative clinical study of cefaclor and cephalexin in bacterial bronchitis. Chemotherapy (Tokyo) 29:653–97 (in Japanese).
Ishigami J, Mita T, Ohno S et al. (1981). Clinical evaluation of cefaclor in acute simple cystitis. A double-blind comparative study of cefaclor. Chemotherapy (Tokyo), 29:250–66 (in Japanese).
Arata J, Yamamoto Y, Nohara N et al. (1981). A double-blind comparison between cefaclor and cephalexin in the treatment of acute bacterial skin infections. Chemotherapy (Tokyo), 29:267–79 (in Japanese).
Horii M, Morinaga T, Takeuchi T et al. (1984). A double-blind comparison between cefaclor and cephalexin in the treatment of dental infections. Jpn J Antibiotics, 37:152–75 (in Japanese).
Konno K, Saitoh A, Ohizumi K et al. (1986). Comparison of cefixime and cefaclor in bacterial bronchitis. Chemotherapy (Tokyo), 34:1150–83 (in Japanese).
Konno K, Saitoh A, Ohizumi K et al. (1986). Comparative test of the efficacy of cefixime and amoxicillin on pneumonia by double-blind method. Chemotherapy (Tokyo), 34:1184–218 (in Japanese).
Arakawa S, Fujii A, Kamidono S et al. (1986). A double-blind study to compare cefixime and L-cephalexin for the treatment of complicated urinary tract infections. Nishinippon Hinyoukika, 48:645–74 (in Japanese).
Kawada Y, Kumamoto E, Nishimoto T et al. (1986). Comparative study of T-2588 and cefadroxil in complicated urinary tract infections. Chemotherapy (Tokyo), 34:908–29 (in Japanese).
Kobayashi H, Kawai S, Saitoh A et al. (1986). Comparative clinical study of T-2588 and Bacampicillin for bacterial pneumonia by double-blind method. Kansensyougaku Zasshi, 60:1078–106 (in Japanese).
Kobayashi H, Kawai S, Saitoh A et al. (1986). Comparative clinical study of T-2588 and cefaclor for chronic respiratory tract infections by a double-blind method. Kansensyougaku Zasshi, 60:1052–77 (in Japanese).
Shiba K, Saitoh A, Shimada J et al. (1988). Comparative clinical study of CS-807 and cefaclor for bacterial pneumonia by a double-blind method. Kansensyougaku Zasshi, 62:973–1001 (in Japanese).
Shiba K, Saitoh A, Shimada J et al. (1988). Comparative clinical study of CS-807 and cefaclor for chronic respiratory tract infections by a double-blind method. Kansensyougaku Zasshi, 62:1166–91 (in Japanese).
Ogata N, Kumazawa J, Kushimoto T et al. (1988). A double-blind study to compare CS-807 and L-cephalexin for the treatment of complicated urinary tract infections. Nishinippon Hinyoukika, 50:2077–98 (in Japanese).
Kobayashi H, Takamura K, Kono K et al. (1984). Comparison of DL-8280 and amoxicillin in the treatment of respiratory tract infection. Kansensyougaku Zasshi, 58:525–54 (in Japanese).
Fujimori I, Kobayashi Y, Obana M et al. (1984).Comparative clinical study of ofloxacin and cefaclor in bacterial bronchitis. Kansensyougaku Zasshi, 58:832–61 (in Japanese).
Fujita K, Nakano M, Nonami E et al. (1984). Comparative clinical study of DL-8280 and cefaclor for suppurative skin and soft tissue infections by a double-blind method. Kansensyougaku Zasshi, 58:793–819 (in Japanese).
Saito M, Seo T, Matsubara Y et al. (1984). Comparison of clinical efficacy of ofloxacin (OFLX: DL8280) and pipemidic acid (PPA) in acute infectious diarrhoea by a double-blind method. Kansensyougaku Zasshi, 58:965–81 (in Japanese).
Kishi H, Nito H, Saitoh I et al. (1984). Comparative studies of DL-8280 and pipemidic acid in complicated urinary tract infections by double-blind method. Hinyoukiyou, 30:307–55 (in Japanese).
Ishigami J, Kamidono S, Harada M et al. (1984). A double-blind controlled study of DL-8280 in comparison with pipemidic acid in the treatment of acute simple cystitis. Nishinippon Hinyoukika, 46:967–88 (in Japanese).
Kawamura S, Fukimaki Y, Iwasawa T et al. (1984). A comparative double-blind study of DL-8280 and pipemidic acid in suppurative otitis media. Otologia Fukuoka, 30:642–70 (in Japanese).
Sasaki T, Unno T, Tomiyama T et al. (1984). Evaluation of clinical effectiveness and safety of DL8280 in acute lacunar tonsillitis; in comparison with amoxicillin by double-blind method. Otologia Fukuoka, 30:484–513 (in Japanese).
Takase Z, Komoto K, Katayama M et al. (1984). Comparative clinical study of ofloxacin (OFLX) and amoxicillin (AMPC) on the infectious disease in the field of obstetrics and gynaecology. Chemotherapy (Tokyo), 34:31–63 (in Japanese).
Ohizumi K, Saitoh A, Nagahama F et al. (1987). Comparative double-blind study of the efficacy of 6315-S (Flomoxef) and Latamoxef on chronic respiratory tract infections. Chemotherapy (Tokyo), 35 (Suppl 1):780–809 (in Japanese).
Kumazawa J, Matsumoto T, Kumamoto E et al. (1987). 6315-S (Flomoxefl in complicated urinary tract infections: a double-blind controlled study using LMOX. Chemotherapy (Tokyo), 35 (Suppl 1):1 138–63 (in Japanese).
Soejima R, Matsushima T, Kawane H et al. (1986). Comparative study of MK-0787/MK-0791 and piperacillin in respiratory tract infections. Kansensyougaku Zasshi, 60:345–77 (in Japanese).
Kawada Y, Nishiura T, Kumamoto E et al. (1986).Comparative study of MK-0787 /MK-0791 and cefoperazone in complicated urinary tract infections. Chemotherapy (Tokyo), 34:536–60 (in Japanese).
Yura J, Shinagawa N, Ishikawa S et al. (1986). Comparative clinical study of imipenem/cilastatin sodium and ceftizoxime in the treatment of puruient peritonitis. Chemotherapy (Tokyo), 34:713–38 (in Japanese).
Matsumoto K, Uzuka Y, Shishido H et al. (1979). Clinical evaluation of cefotiam (SCE-963) in pulmonary infections: a comparative study with cefazolin by a randomized double-blind technique. Chemotherapy (Tokyo), 27 (Suppl 3):399–419 (in Japanese).
Ishigami J, Ohno S, Tomioka O et al. (1979). Clinical evaluation of cefotiam (SCE-963) in complicated urinary tract infections: a comparative study with cefazolin by a randomized double blind technique. Chemotherapy (Tokyo), 27 (Suppl 3):629–48 (in Japanese).
Baba S, Iwasawa T, Kawamura S et al. (1983). Clinical evaluation of cefotiam (SCE-963) in suppurative otitis media: a comparative study with cefazolin by a randomized double-blind technique. Jitenn,Suppl 5:451–68 (in Japanese).
Ohtomo E, Araki G, Hasegawa K et al. (1985). Clinical efficacy of CV-2619 (Idebenone) in the treatment of cerebrovascular disorders. Multi-centre double-blind study in comparison with Ca-hopantenate. Igaku no Ayumi, 134:220–42 (in Japanese).
Ohtomo E, Tohgi H, Hirai S et al. (1986). Clinical effectiveness of YM-08054 (Indeloxazine) in the treatment of cerebrovascular disorders. Multi-centre double-blind study in comparison with Ca-hopantenate. Igaku no Ayumi, 136:535–55 (in Japanese).
Ohtomo E, Hirai S, Hasegawa K et al. (1986). Clinical evaluation of TA-079 (Nicergoline) in the treatment of cerebrovascular disorders. Multi-centre double-blind study in comparison with Ca-hopantenate. Clin Eual, 14:575–602 (in Japanese).
Tasaki Y, Kutsuzawa T, Tohgi H et al. (1986). Clinical efficacy of E-0687 (Bifemelane) in the treatment of cerebrovascular disorders. Multi-centre double-blind study in comparison with Ca-hopantenate. Igaku no Ayumi, 137:647–70 (in Japanese).
Atarashi J, Araki G, Itoh E et al. (1983). Clinical efficacy of TCV-3B (Vinpocetin) in the treatment of cerebrovascular disorders. Multi-centre double-blind study in comparison with Ifenprodil tartarate. Igaku no Ayumt, 1983;124:66–90 (in Japanese).
Serizawa S, Shirakawa K, Sakita T et al. (1983). Clinical efficacy of E-0671 (Tetraprenylacetone) in the treatment of gastric ulcer. Multi-centre double-blind study with proglumide. Prog Med, 3 (Suppl):1 169–91 (in Japanese).
Ichita F, Tashiro N, Murayama H et al. (1976). Clinical efficacy of Marzulene-S granule in the treatment of gastric ulcer. Double-blind study with L-glutamine or azulene. Shinyaku to Rinsho, 25:167–75 (in Japanese).
Kobayashi S, Sekiguchi T, Takei A et al. (1981). Clinical study of Marzulene-S granule in the treatment of duodenal ulcer. Double-blind study with L-glutamine. Shtnyaku to Rinsho, 30:1855–73 (in Japanese).
Ishii K, Takebe T, Hirayama N et al. (1980). Clinical evaluation of FOY-305 in pancreatitis. Multi-centre double-blind study. Gendat Iryou, 12:261–78 (in Japanese).
Ishii K, Takebe T, Hirayama N et al. (1984). Clinical evaluation of FOY-305 in chronic pancreatitis. Multi-centre double-blind study. Gendai Iryou, 16:844–54 (in Japanese).
Itami Y, Inoue T, Takahashi H et al. (1982). Clinical evaluation of elcatonin in the treatment of lumbodorsal pain complicated in osteoporosis. Multi-centre double-blind study. Igaku no Ayumi, 120:1180–95 (in Japanese).
Itami Y, Fujita T, Inoue T et al. (1982). Clinical efficacy of alfacalcidol (1a-OH-D3) in the treatment of osteoporosis. Comparative study by multi-centre double-blind method. Igaku no Ayumi, 123:958–73 (in Japanese).
Shichikawa K, Igarashi M, Sugawara S and Iwasaki Y. (1983). Clinical evaluation of high molecular weight sodium hyaluronate (SPH) on osteoarthritis of the knee. Multi-centre well controlled comparative study. Jpn J Clin Pharmacol Ther, 14:545–58 (in Japanese).
Hirohata K, Matsuda T, Watanabe Y et al. (1985). Clinical evaluation of CS-600 (loxoprofen sodium) on lumbago. Comparative study with ibprofen by multi-centre double-blind method. Prog Med, 5:1487–505 (in Japanese).
Aoki T, Sugawara S, Hoshino T et al. (1986). Clinical evaluation of CS-600 (loxoprofen sodium) on osteoarthritis. Comparative study with diclofenac sodium by multi-centre double-blind method. Igaku no Ayumi, 136:983–1001 (in Japanese).
Hirasawa Y, Hirashima K, Arakawa M et al. (1989). Clinical evaluation of recombinant human erythropoietin (EPOCH) on renal anaemia: A double-blind, three doses comparative study. Kidney and Dialysis, 27:157–77 (in Japanese).
Agishi Y, Asanuma Y, Shimizu S et al. (1986). Clinical evaluation of lipo PGE, on occupational vibration syndrome using double-blind comparative method. Rinsho Iyaku, 2:1269–89 (in Japanese).
Katumura T, Uemichi T, Ohshiro T et al. (1986). Clinical evaluation of lipo PGE1 in the treatment of ischemic ulcer of the extremities. A multi-centre double-blind comparison with inositol niacinate. Junkankika, 20:331–50 (in Japanese).
Kumagai A, Takahashi S, Shida T et al. (1982). Clinical evaluation of HC 20–511 (Ketotifen), a new oral anti-anaphylactic compound, in bronchial asthma. II - Multi-centre double-blind study in comparison with disodium cromoglycate. Clin Eval, 10:737–85 (in Japanese).
Kumagai A, Takahashi S, Shida T et al. (1980). Clinical evaluation of HC 20–511 (Ketotifen), a new orally anti-anaphylactic compound, in bronchial asthma. I - Multi-centre double-blind study in comparison with placebo. Clin Eval, 8:353–96 (in Japanese).
Yasugi T, Goto Y, Yoshida S et al. (1988). Clinical evaluation of CS-514, (Pravastatin) on hyperlipidemia. Double-blind study with clinofibrate. Clin Eval, 16:211–49 (in Japanese).
Goto Y, Yamamoto A, Matsuzawa Y et al. (1988). Clinical evaluation of pravastatin (CS-514) on hyperlipidaemia. Double-blind study with probucol. Igaku no Ayumi, 146:927–55 (in Japanese).
Saito Y, Goto Y, Nakaya N et al. (1988). Dose-dependent hypolipidaemic effect of an inhibitor of HMG-CoA reductase, pravastatin (CS-514), in hypercholesterolaemic subjects. Atherosclerosis, 72:205–11.
Katsumura T, Mishima Y, Kamiya K, Sakaguchi S, Tanabe T, Sakuma A. (1986). Therapeutic effect of ticlopidine, a new inhibitor of platelet aggregation, on chronic arterial occlusive disease. A double blind study with inactive placebo. Junkankika, 7:396–406 (in Japanese).
Murakami M, Toyokura Y, Omae T et al. (1983). Therapeutic effect of ticlopidine or aspirin on transient ischemic attacks (TIA) - Comparative double blind study for 12 months. Igaku no Ayumi, 127:950–71.
British National Formulary No 22. (1991). British Medical Association and Royal Pharmaceutical Society of Great Britain.
British National Formulary No 24. (1992). British Medical Association and Royal Pharmaceutical Society of Great Britain.
Handbook of Medicines (1990). A Society for Pharmaceuticals edn. Yakuji Jihou-sya Ltd., Tokyo (in Japanese).
Dept. of Social Statistics, Ministry of Health and Welfare. (1992). Summary of medical expenditure in Japan. Kosei no Shihyo, 39 (3):32–38. (in Japanese).
The Association of the British Pharmaceutical Industries. (1993). The Health Manager’s Guide to the Pharmaceutical Industry. 1993 edition. ABPI, London.
Ohkoshi M, Isigami J, Kamidono S et al. (1986). The criteria proposed by the UTI committee in Japan. Chemotherapy (Tokyo), 34:408–41 (in Japanese).
Sakita T, Fukutomi H. (1971). Diagnosis of gastric ulcer. In: Yoshitoshi Y (ed.) Gastroduodenal Ulcer, Nankodo, Tokyo, pp. 197–208. (in Japanese).
The Official Book Society of Japan edn. (1992). Guidelines on Clinical Evaluation of Medicines. Yakuji Nippo-sya Ltd., Tokyo, 1992. (in Japanese).
Matsumoto K, Saitoh A, Yokoyama K et al. (1977). An assessment method of clinical states in bacterial pneumonia. Jpn J Clin Pharmacol Ther, 8:155–68 (in Japanese).
Ohtomo E, Kutsuzawa N, Hasegawa K et al. (1982). Clinical evaluation of HOPA (HOPATE) in the treatment of cerebrovascular disorders. - Multi-centre double-blind study in comparison with Ifenprodil tar-tarate and placebo. Clin Eval, 9:673–710 (in Japanese).
FX-505 project for Phase III study. (1976). Double blind study of FX-505 (Ifenprodil) on cerebrovascular diseases. Clin Eval, 4:419–58 (in Japanese).
Kawal K, Kohli Y, Takeda S et al. (1978). A multi-clinical double-blind controlled study on the clinical efficacy of proglumide in gastric ulcers. Clin Eval, 6:69–84 (in Japanese).
New Drugs Division, Japanese Ministry of Health and Welfare edn. (1990). Handbook on Good Clinical Practice. Yakuji Jiho-sya Ltd., Tokyo (in Japanese).
New Drugs Division, Japanese Ministry of Health and Welfare. (1992). Guidelines on statistical analyses in clinical trials. Clin Eval, 20:205–219 (in Japanese).
New Drugs Division, Japanese Ministry of Health and Welfare. (1992). General guidelines on clinical evaluation of new drugs. Jpn J Clin Pharmacol Ther, 23:609–21 (in Japanese).
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1994 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Hirokawa, K., Dollery, C.T. (1994). The top 50 drugs in the UK and Japan: why are they so different?. In: Walker, S., Lumley, C., McAuslane, N. (eds) The Relevance of Ethnic Factors in the Clinical Evaluation of Medicines. CMR Workshop Series. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-1420-2_5
Download citation
DOI: https://doi.org/10.1007/978-94-011-1420-2_5
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-4621-3
Online ISBN: 978-94-011-1420-2
eBook Packages: Springer Book Archive