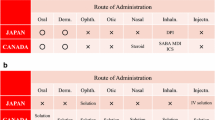
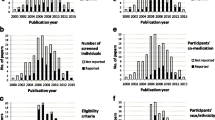
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Homma M (1992). Requirements for assessment of clinical safety regulatory perspectives. In: D’Arcy PF and Harron DWG (eds) Proceedings of The First International Conference on Harmonisation, Brussels, 1991. The Queen’s University of Belfast, pp. 390–399.
Pocock SJ (1983). Clinical Trials. A Practical Approach. John Wiley & Sons, Chichester, p. 75.
Yamamoto K (1991). Utility of drugs and its assessments in clinical trials. Clin Eval 19:205–212 (in Japanese).
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1994 Springer Science+Business Media Dordrecht
About this chapter
Cite this chapter
Naito, C. (1994). Evaluation methods for clinical trials of drugs in Japan which may affect ethnic differences. In: Walker, S., Lumley, C., McAuslane, N. (eds) The Relevance of Ethnic Factors in the Clinical Evaluation of Medicines. CMR Workshop Series. Springer, Dordrecht. https://doi.org/10.1007/978-94-011-1420-2_4
Download citation
DOI: https://doi.org/10.1007/978-94-011-1420-2_4
Publisher Name: Springer, Dordrecht
Print ISBN: 978-94-010-4621-3
Online ISBN: 978-94-011-1420-2
eBook Packages: Springer Book Archive