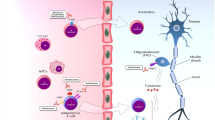
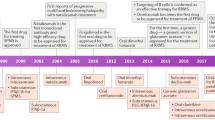
Abstract
Multiple sclerosis (MS) is a disease leading to a significant disability in the vast majority of patients [1, 2]. The available immunomodulatory treatments are not a cure for MS, but there is clear evidence from class I clinical trials that they significantly reduce the disease activity and delay the increase of disability in relapsing-remitting patients [3-10]. However, the positive effects are less clear in secondary progressive patients [11, 12]. The different effects of immunomodulatory treatments according to the disease course is probably explained by the complex pathogenesis of MS. Indications on the use of available therapies for MS have substantially changed in a few years, passing from a conservative [13, 14] to a more-aggressive attitude [15]. It is interesting to note that the consensus statement of the Canadian MS Clinic Network, recently published [15], on the use of disease modifying agents in MS, requires evidence of ongoing disease activity, which can be based on clinical or magnetic resonance imaging (MRI) data. The previous consensus of treatment [13, 14, 16, 17] required two or more relapses in the last 2 years in order to start treatment. These changes are probably explained by the results of the new trials testing the efficacy and safety of inter-ferons and glatiramer acetate [5-7, 10], by the experience acquired during these years, and by the recent knowledge of the pathophysiology of the disease. The demonstration of early irreversible axonal damage is a strong argument in favor of early treatment, an option that is beginning to be favored by many neurologists [18, 19].
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Confavreux C, Aimard G, Devic M (1980) Course and prognosis of multiple sclerosis assessed by the computerized data processing of 349 patients. Brain 103:281–300
Weinshenker BG, Bass B, Rice GPA, Noseworthy J, Carriere W, Baskerville J, Ebers GC (1989) The natural history of multiple sclerosis: a geographically based study. Clinical course and disability. Brain 112:133–146
The IFNB MS Study Group (A) (1993) Interferon Beta-lb is effective in relapsing-remitting multiple sclerosis. I. Clinical results of a multicenter, randomized, double-blind, placebo-controlled trial. Neurology 43:655–661
Jacobs LD, Cookfair DL, Rudick RA et al (1996) Intramuscular interferon beta-la for disease progression in relapsing multiple sclerosis. Ann Neurol 39:285–294
Rudick RA, Goodkin D, Jacobs L et al (1997) Impact of interferon beta-la on neurologic disability in relapsing multiple sclerosis. Neurology 49:358–363
PRISMS (Prevention of Relapses and Disability by Interferon B-la Subcutaneously in Multiple Sclerosis) Study Group (1998) Randomised double-blind placebo-controlled study of interferon β-la in relapsing/remitting multiple sclerosis. Lancet 352:1498–1504
The OWIMS Study. The Once Weekly Interferon for MS study Group (OWIMS) (1999) Evidence of interferon (3-la dose response in relapsing-remitting MS. Neurology 53:679–686
Johnson KP, Brooks RB, Cohen JA, The Copolymer 1 Multiple Sclerosis Research Group (1995) Copolymer 1 reduces relapse rate and improves disability in relapsing-remitting multiple sclerosis: results of the phase III multicenter, double-blind, placebo-controlled trial. Neurology 45:1268–1276
Johnson KP, Brooks RB, Cohen JA, The Copolymer 1 Multiple Sclerosis Research Group (1998) Extended use of glatiramer acetate (Copaxone) is well tolerated and maintains its clinical effect on multiple sclerosis relapse rate and degree of disability. Neurology 50:701–708
Comi G, Filippi M, Copaxone MRI Study Group (1999) The effect of glatiramer acetate (Copaxone) on disease activity as measured by cerebral MRI in patients with relapsing-remitting multiple sclerosis (RRMS) Neurology 52:A289
European Study Group on Interferon β- lb in Secondary Progressive MS (1998) Placebo multicentre randomised trial of interferon β-lb in treatment of secondary progressive multiple sclerosis. Lancet 352:1491–1497
Paty DW on behalf of the SPECTRIMS Study Group (1999) Results of the 3-year, double blind, placebo controlled study of interferon beta-la (Rebif) in secondary progressive MS. J Neurol 246 [suppl 11:I15
Report of the Quality Standards Subcommittee of the American Academy of Neurology (1994) Practice advisory on selection of patients with multiple sclerosis for treatment with Betaseron. Neurology 44:1537–1540
Lublin FD, Whitaker JN, Eidelman BH, Miller AE, Arnason BGW, Burks JS (1996) Management of patients receiving interferon beta-lb for multiple sclerosis. Neurology 46:12–18
Oger J, Freedman M (1999) Consensus Statement of the Canadian MS Clinical Network on: The Use of Disease Modyfing Agents in Multiple Sclerosis. Can J Neurol Sci 26:274–275
Polman CH, Miller DH, Mcdonald WI, Thompson AJ (1999) Treatment recommendations for interferon 13 in multiple sclerosis. J Neurol Neurosurg Psychiatry 67:561–566
Rieckman P, Toyka KV, and the Austrian-German-Swiss Multiple Sclerosis Therapy Consensus Group (MSTCG) (1999) Escalating immunotherapy of multiple sclerosis. Eur Neurol 42:121–127
Comi G, Martinelli V, Martino G (1998) Early treatment of multiple sclerosis. Eur J Neurol 5 [suppl 2J:S19–S21
Rudick AR (1999) Disease-modifying drugs for relapsing remitting multiple sclerosis and future directions for multiple sclerosis therapeutics. Arch Neurol 56:1079–1084
Filippi M, Horsfield MA, Morissey SP, MacManus DG, Rudge P, McDonald WI, Miller DH (1994) Quantitative brain MRI lesion load predicts the course of clinically isolated syndromes suggestive of multiple sclerosis. Neurology 44:635–641
O’Riordan JI, Thompson AJ, Kingsley PE, MacManus DG, Kendall BE, Rudge P, McDonald WI, Miller DH (1998) The prognostic value of brain MRI in clinically isolated syndromes of the CNS. A 10-year follow-up. Brain 121:495–503
Brex PA, O’Riordan JI, Miszkiel KA, Moseley IF, Thompson AJ, Plant GT, Miller DH (1999) Multisequence MRI in clinically isolated syndromes and the early development of MS. Neurology 53:1184–1190
Comi G, Filippi M, Barkhof F and the ETOMS Study Group (2001) Effects of early interferon treatment on conversion to definite multiple sclerosis: a randomised study. Lancet 357:1576–1582
Lucchinetti C et al (2000) Heterogeneity of multiple sclerosis lesions: implications for the pathogenesis of demyelination. Ann Neurol 47:707–717
Soldestrom M, Jin Ya-Ping, Hillert J, Link H (1998) Optic neuritis: prognosis for multiple sclerosis from MRI, CSF, and HLA findings. Neurology 50:708–714
Sciacca FL, Ferri C, Vandenbroeck K, Veglia F, Gobbi C, Martinelli F, Franciotta D, Zaffaroni M, Marrosu M, Martino G, Martinelli V, Comi G, Canal N, Grimaldi LMG (1999) Relevance of interleukin 1 receptor antagonist intron 2 polymorphism in Italian MS. Neurology 52:1896–1898
Weinshenker BG (1994) Natural history of multiple sclerosis. Ann Neurol 36 [Suppl]: 56–511
Runmarker B, Andersen O (1993) Prognostic factors in a multiple sclerosis incidence cohort with twenty-five years of follow-up. Brain 116:117–134
Yu BM, Johnson MJ, Tuohy VK (1996) A predictable sequential determinant spreading cascade invariably accompanies progression of experimental autoimmune encephalomyelitis: a basis for peptide-specific therapy after onset of clinical disease. J Exp Med 183:1777–1788
Thuoy VK, Weinstock-Guttman B, Kinkel RP (1997) Diversity and plasticity of self recognition during the development of multiple sclerosis. J Clin Invest 99:1682–1690
Balashov KE, Smith DR, Khoury SJ, Hafler DA, Weiner HL (1997) Increased interleukin 12 production in progressive multiple sclerosis: induction by activated CD4+ T cells via CD40 ligand. Proc Natl Acad Sci U S A 94:599–603
Nicoletti F et al (1996) Elevated serum levels of interleukin-12 in chronic progressive multiple sclerosis. Neuroimmunology 70:87–90
Lucchinetti CF, Bruck W, Rodriguez M, Lassmann H (1996) Distinct patterns of multiple sclerosis pathology indicates heterogeneity on pathogenesis. Brain Pathol 6:259–274
Lassmann H (1998) Neuropathology in multiple sclerosis: new concepts. Mult Scler 4:93–98
Bo L et al (1994) Induction of nitricoxide synthase in demyeinating regions of multiple sclerosis brains. Ann Neurol 36:778–786
Charcot JM (1868) Histologie de la sclerose en plaques. Gaz Hop (Paris) 141:554–558
Putnam TJ (1936) Studies in multiple sclerosis. Arch Neurol Psychol 35:1289–1308
Greenfield J, King L (1936) Observations on the histopathology of the cerebral lesions in disseminated sclerosis. Brain 59:445–458
Prineas WJ, Barnard RO, Revesz T, Kwon EE, Sharer L, Cho ES (1993) Multiple sclerosis. Pathology of recurrent lesions. Brain 116:681–693
Rodriguez et al (1992) Central nervous system demyelination and remyelination in multiple sclerosis and viral models of disease. J Neuroimmunol 40:255–263
Bruck W, Schmied M, Suchanek G, Bruck Y, Breitschopf H, Poser S et al (1994) Oligodendrocytes in the early course of multiple sclerosis. Ann Neurol 35:65–73
Ozawa K, Suchanek G, Breitschopf H, Bruck W, Budka H, Jellinger K, Lassmann H (1994) Patterns of oligodendroglia pathology in multiple sclerosis. Brain 117:1311–1322
Linington C, Engelhardt B, Kapocs G, Lassmann H (1992) Induction of persistenly demyelinated lesions in the rat following the repeated adoptive transfer of encephalitogenic T cells and demylinating antibody. J Neuroimmunol 40:219–224
Ferguson B, Matyszak MK, Esiri MM, Perry VH (1997) Axonal damage in acute multiple sclerosis lesions. Brain 120:393–399
Trapp BD, Peterson J, Ransohoff RM, Rudick R, Mork S, Bo L (1998) Axonal transection in the lesions of multiple sclerosis. New Engl J Med 338:278–285
Raine C, Cannella B, Hauser S, Genain C (1999) Demyelination in primate autoimmune encephalomyelitis and acute multiple sclerosis lesions: a case for antigen-specific antibody mediation. Ann Neurol 46:144–160
Lovas G, Szilagyi N, Majtenyi K, Palkovits M, Komoly S (2000) Axonal changes in chronic demyelinated cervical spinal cord plaques. Brain 123:308–317
Filippi M, Campi A, Colombo B, Pereira C, Martinelli V, Baratti C, Comi G (1996) A spinal cord MRI study of benign and secondary progressive multiple sclerosis. J Neurol 243(7):502–505
Losseff NA, Webb SL, O’Riordan JI, Page R, Wang L, Barker GJ, Tofts PS, McDonald WI, Miller DH, Thompson AJ (1996) Spinal cord atrophy and disability in multiple sclerosis. A new reproducible and sensitive MRI method with potential to monitor disease progression. Brain 119:701–708
Narayana PA, Doyle TJ, Lai D, Wolinsky JSM (1998) Serial proton magnetic resonance spectroscopic imaging, contrast-enhanced magnetic resonance imaging, and quantitative lesion volumetry in multiple sclerosis. Ann Neurol 43:56–71
Tourbah A, Stievenart JL, Gout O, Fontaine B, Liblau R, Lubetzki C, Cabanis EA, Lyon-Caen O (1999) Localized proton magnetic resonance spectroscopy in relapsing remitting versus secondary progressive multiple sclerosis. Neurology 53:1091–1097
Sarchielli P, Presciutti O, Pelliccioli GP, Tarducci R, Gobbi G, Chiarini P, Alberti A, Vicinanza F, Gallai V (1999) Absolute quantification of brain metabolites by proton magnetic resonance spectroscopy in normal-appearing white matter of multiple sclerosis patients. Brain 122:513–521
Matthews PM, De Stefano N, Narayanan S, Francis GS, Wolinsky JS, Antel JP, Arnold DL (1998) Putting magnetic resonance spectroscopy studies in context: axonal damage and disability in multiple sclerosis. Semin Neurol 18:327–336
Falini A, Calabrese G, Filippi M, Origgi D, Lipari S, Colombo B, Comi G, Scotti G (1998) Benign versus secondary-progressive multiple sclerosis: the potential role of proton MR spectroscopy in defining the nature of disability. Am J Neuroradiol 19:223–229
Rocca MA, Mastronardo G, Rodegher M, Comi G, Filippi M (1999) Long-term changes of magnetization transfer-derived measures from patients with relapsing-remitting and secondary progressive multiple sclerosis. Am J Neuroradiol 20:821–827
Davie CA, Silver NC, Barker GJ, Tofts PS, Thompson AJ, McDonald WI, Miller DH (1999) Does the extent of axonal loss and demyelination from chronic lesions in multiple sclerosis correlate with the clinical subgroup? J Neurol Neurosurg Psychiatry 67:710–715
Filippi M, Rocca MA, Minicucci L, Martinelli V, Ghezzi A, Bergamaschi R, Comi G (1999) Magnetization transfer imaging of patients with definite MS and negative conventional MRI. Neurology 52:845–848
Brochet B, Dousset V (1999) Pathological correlates of magnetization transfer imaging abnormalities in animal models and humans with multiple sclerosis. Neurology 53 [Suppl3]:S127
Rudick RA, Fisher E, Lee JC, Simon J, Jacobs L (1999) Use of the brain parenchymal fraction to measure whole brain atrophy in relapsing-remitting MS. Multiple Sclerosis Collaborative Research Group. Neurology 53:1698–1704
Dastidar P, Heinonen T, Lehtimaki T, Ukkonen M, Peltola J, Erila T, Laasonen E, Elovaara I (1999) Volumes of brain atrophy and plaques correlated with neurological disability in secondary progressive multiple sclerosis. J Neurol Sci 165:36–42
van Waesberghe JH, Kampphorst W, De Groot CJ, van Walderveen MA, Castelijns JA, Ravid R, Lycklama à Nijeholt G, van der Valk P, Polman C, Thompson AJ, Barkhof F (1999) Axonal loss in multiple sclerosis lesions: magnetic resonance imaging insights into substrates of disability. Ann Neurol 46:747–754
Gass A et al (1994) Correlation of magnetization transfer ratio with disability in multiple sclerosis. Ann Neurol 36:62–67
Filippi M et al (1999) Comparison of MS clinical phenotypes using conventional and magnetization transfer MRI. Neurology 52:588–594
Filippi M et al (1995) A magnetization transfer imaging study of normal appearing white matter in multiple sclerosis. Neurology 45:478–482
Iannucci G, Tortorella C, Rovaris M, Sormani MP, Comi G, Filippi M (2000) Prognostic value of MR and magnetization transfer imaging findings in patients with clinically isolated syndromes suggestive of multiple sclerosis at presentation. Am J Neuroradiol 21:1034–1038
Brex PA et al (2000) Detection of ventricular enlargement in patients at the earliest clinical stage of MS. Neurology 54:1689–1691
Rudick RA et al (1999) Use of the brain parenchimal fraction to measure whole brain atrophy in relapsing-remitting MS. Neurology 10:1698–1704
Simon JH, Jacobs LD, Campion MK, Rudick RA, Cookfair DL, Herndon RM, Richert JR, Salazar AM, Fisher JS, Goodkin DE, Simonian N, Lajaunie M, Miller DE, Wende K, Martens-Davidson A, Kinkel RP, Munschauer III FE, Brownscheidle and The Multiple Sclerosis Collaborative Research (1999) A longitudinal study of brain atrophy in relapsing multiple sclerosis. Neurology 53:139–148
Beck RW, Cleary PA, Trobe JD et al (1993) The effect of corticosteroids for acute optic neuritis on the subsequent development of multiple sclerosis. N Engl J Med 329:1764–1769
Optic Neuritis Study Group (1997) The 5-year risk of MS after optic neuritis. Experience of the optic neuritis treatment trial. Neurology 49:1404–1413
Jacobs LD, Beck RW, Simon JH and the CHAMPS Study Group (2000) Intramuscular interferon b-la therapy initiated during a first demyelinating event in multiple sclerosis. N Engl J Med 343:898–904
McDonald WI, Compston A, Edan G et al (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the international panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2004 Springer-Verlag Italia
About this chapter
Cite this chapter
Comi, G., Moiola, L. (2004). Evidence for an Early Treatment of Multiple Sclerosis. In: Hommes, O.R., Comi, G. (eds) Early Indicators Early Treatments Neuroprotection in Multiple Sclerosis. Topics in Neuroscience. Springer, Milano. https://doi.org/10.1007/978-88-470-2117-4_1
Download citation
DOI: https://doi.org/10.1007/978-88-470-2117-4_1
Publisher Name: Springer, Milano
Print ISBN: 978-88-470-2171-6
Online ISBN: 978-88-470-2117-4
eBook Packages: Springer Book Archive