Abstract
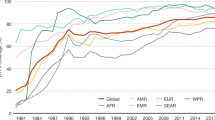
In both industrialized and developing countries, childhood immunization has become one of the most important and cost-effective public health interventions. National immunization programs have prevented millions of deaths since WHO initiated the ‘Expanded Program on Immunization’ in 1974. Smallpox was eradicated in 1979, poliomyelitis is on the verge of eradication, and two thirds of developing countries have eliminated neonatal tetanus. Global immunization coverage was at 78% in 2005. Through their impact on childhood morbidity and mortality, immunization programs are contributing to reaching the ‘Millennium Development Goal 4’ — a two-thirds reduction of under-five mortality by 2015. However, the failure to reach more than 20% of the world’s children with existing vaccines was responsible for at least 2.5 million of an estimated 10.5 million deaths of children under 5 years, mainly in developing countries. Of these deaths, 1.4 million could have been prevented by vaccines currently recommended by WHO. Rapid progress in our understanding of the pathogenesis of infectious diseases, immunology, and biotechnology has increased the number of candidate vaccine antigens available. Pressures are growing on public health decision makers to establish evidence-based ways to decide which new vaccines should be introduced on a large scale into national immunization programs. The gap in access to new vaccines between the developing and industrialized worlds is still wide, and wealthy countries are still the first to introduce and use new vaccines. Interest from countries and partner agencies in vaccination, as one of the most cost-effective public health interventions, continues to be strong, also due to rapid progress in biotechnology and vaccine development and the emergence of global infectious disease threats, including HIV/AIDS, SARS, and influenza. The establishment of the Global Alliance for Vaccines and Immunization has focused global activities to support vaccination programs through raising considerable funds, and to assist especially poorer countries in improving and expanding their vaccination programs. Global efforts concentrate on further reducing the gap in the access to all existing vaccines between industrialized and developing countries.
R. Tangermann and R. Eggers are staff members of the World Health Organization. The authors alone are responsible for the views expressed in this publication and they do not necessarily represent the decisions, policy or views of the World Health Organization.
Chapter PDF
Similar content being viewed by others
Keywords
These keywords were added by machine and not by the authors. This process is experimental and the keywords may be updated as the learning algorithm improves.
References
Miller MA, Hinman AR (2004) Economic analysis of vaccine policies. In: SA Plotkin, WA Orenstein (eds): Vaccines, 4th edn. Elsevier Inc., Philadelphia, PA, 1463–1490
Bloom DE, Canning D, Weston M (2005) The value of vaccination. World Economics 6: 15–39
Keja K, Chan C, Hayden G, Henderson RH (1988) Expanded programme on immunization. World Health Stat Q 41: 59–63
Fenner F, Henderson DA, Arita I, Jezek A, Ladnyi ID (1988) Smallpox and its eradication. World Health Organization, Geneva
Expanded Programme on Immunisation (2006) Progress towards the global eradication of poliomyelitis, 2005. Wkly Epidemiol Rec 81: 164–172
World Health Organization (2005) WHO vaccine-preventable diseases: monitoring system 2005 summary. WHO IVB/2005, Geneva
World Health Organization and United Nations Children’s Fund (2003) State of the World’s Vaccines and Immunization. World Health Organization, Geneva
World Health Organization and United National Children’s Fund. Global Immunization Vision and Strategy, 2006–2015 (2005) Geneva, Switzerland: World Health Organization and United National Children’s Fund. (WHO/IVB/05.05). Accessed on June 30 at http//:www.who.int/vaccines/GIVS/English/GIVS_final_17Oct05.pdf
Murray CJL, Lopez AD, Mathers CD, Stein C (2001) The Global Burden of Disease 2000 Project: Aims, Methods, and Data Sources (Global Programme on Evidence for Health Policy Discussion Paper No 36). World Health Organization, Geneva
World Health Organization (2006) Challenges in global immunization and the global Immunization Vision and Strategy, 2006–2015. Wkly Epidemiol Rec 81: 190–195
UNICEF. A world fit for children: Millennium Development Goals (2003) Special session on children documents: the convention on the rights of the child. July 2002. Accessed on June 30, 2006 at www.unicef.org/publications/index_4445.html
Feudtner C, Marcuse EK (2001) Ethics and immunization policy: promoting dialogue to sustain consensus. Pediatrics 107: 1158–1164
Global Alliance for Vaccines and Immunization (2000) Second GAVI Board Meeting, January 2000 (GAVI/00.01). World Health Organization, Geneva
Declaration of Alma-Ata (1978) International Conference on Primary Health Care, Alma-Ata, USSR, 6–12 September, 1978
World Health Organization (2003) Vaccines, Immunization & Biologicals. Case Information for the Development of Immunization Policy (WHO/V&B/02.28). World Health Organization, Geneva
Campbell JD, Burgess M (2004) Heterogeneity of pediatric immunization schedules in industrialized countries. In: MM Levine, JB Kaper, R Rappuoli, MA Liu, MF Good (eds): New Generation Vaccines, 3rd edn. Marcel Dekker, New York
Hadler S, Dietz V, Okwo-Bele JM, Cutts FT (2004) Vaccination programs in developing countries. In: SA Plotkin, WA Orenstein (eds): Vaccines, 4th edn. Elsevier, Philadelphia, PA
World Health Organization (2005) WHO vaccine preventable diseases monitoring system:2005 Global summary. Accessed June 30, 2006 at http//:www.who. int/vaccines-documents/globalsummary/globalsummary.pdf
Expanded Programme on Immunization (1987) Issues in Neonatal Tetanus Control. World Health Organization, Geneva WHO/EPI/GAG/87/WP.11
World Health Organization (2006) Tetanus vaccine. WHO position paper. Wkly Epidemiol Rec 81: 198–208
World Health Organization (2000) Sustainable outreach services (SOS): A strategy for reaching the unreached with immunization and other services. WHO/V&B/00.37. World Health Organization, Geneva
Global Tuberculosis Programme and Global Programme on Vaccines (2004) BCG vaccine — WHO position paper. Wkly Epidemiol Rec 79: 27–38
Yih WK, Lett SM, des Vignes FN (2000) The increasing incidence of pertussis in Massachusetts’ adolescents and adults, 1989–1998. J Infect Dis 182: 1409–1416
World Health Organization (2000) Strategies for reducing global measles mortality. Wkly Epidemiol Rec 75: 409–416
Expanded Programme on Immunization (2006) Progress in reducing global measles deaths, 1999–2004. Wkly Epidemiol Rec 81: 90–94
Ching P, Birmingham M, Goodman T (2000) Childhood mortality impact and costs of implementing vitamin A supplementation in immunization campaigns. Am J Public Health 90: 1526–1529
Centers for Disease Control and Prevention (2005) Distribution of insecticide-treated bednets during an integrated nationwide immunization campaign — Togo, West Africa, December 2004. MMWR 54: 994–996
Taylor C, Cutts F, Taylor ME (1997) Ethical dilemmas in current planning for polio eradication. Am J Public Health 87: 922–925
Taylor Commission (1995) The Impact of the Expanded Program on Immunization and the Polio Eradication Initiative on Health Systems in the Americas. Pan American Health Organization, Washington, DC
Loevinsohn B, Aylward B, Steinglass R, Ogden E, Goodman T, Melgaard B (2002) Impact of targeted programs on health systems: A case study of the Polio Eradication Initiative. Am J Public Health 92: 19–23
World Health Organization Expanded Programme on Immunization and United Nations Children’s Fund (2000) Product Information Sheets, 2000. World Health Organization, Geneva. WHO document WHO/V&B/000.13.
WHO and UNICEF (1999) Quality for the Cold Chain — WHO/UNICEF policy statement on the use of vaccine vial monitors in immunization practice. World Health Organization, Geneva, WHO/V&B99.18
Nelson CM, Wibisono H, Purwanto H, Mansyur I, Moniaga V, Widjaya A (2004) Hepatitis B vaccine freezing in the Indonesian cold chain: evidence and solutions. Bull World Health Organ 82: 99–105
World Health Organization (2006) Global Advisory Committee on Vaccine Safety, 1–2 December 2005. Wkly Epidemiol Rec 81:15–19
Expanded Programme on Immunisation (1993) Surveillance of Adverse Events Following Immunization: Field Guide for Managers of Immunization Programmes. World Health Organization, Geneva (WHO/EPI/TRAM/93.2)
Folb PI, Bernatowska E, Chen R, Clemens J, Dodoo ANO, Ellenberg SS, Farrington P, John TJ, Lambert PH, MacDonald NE et al (2004) A global perspective on vaccine safety and public health: the Global Advisory Committee on Vaccine Safety. Am J Public Health 94:1926–1931
Simonsen L, Kane A, Lloyd J, Zaffran M, Kane M (1999) Unsafe injections in the developing and the transmission of bloodborne pathogens: a review. Bull World Health Organ 77: 789–798
Anonymous (1999) Safety of injections. WHO-UNICEF-UNFPA joint statement on the use of auto-disable syringes in immunization services. World Health Organization, Geneva, WHO/V&B/99.25
Duclos P, Delo A, Aguado T, Bilous J, Birmingham M, Kieny MP, Milstien J, Wood D, Tarantola D (2003) Immunization safety priority project at the World Health Organization. Semin Pediatr Infect Dis 14: 233–239
Henderson R, Keja J (1989) Global control of vaccine-preventable diseases: how progress can be evaluated. Rev Infect Dis 11(Suppl): S649–S654
World Health Organization (2005) Vaccines, Immunization & Biologicals. WHO/UNICEF Joint Reporting Form on Vaccine Preventable Diseases, 2005. Accessed on the internet June 30, 2006 at www.who.int/immunization_monitoring/Joint_reporting/en/index.html
Henderson RH, Sundaresan T (1982) Cluster sampling to assess immunization coverage: a review of experience with a simplified sampling method. Bull World Health Organ 60: 253–260
de Quadros, CA, Hersh BS., Olive JM (1997) Eradication of wild poliovirus from the Americas: acute flaccid paralysis surveillance, 1988–1995. J Infect Dis 175(Suppl 1): S37–S42
WHO and UNICEF (2005) WHO/UNICEF Joint statement: global plan for reducing measles mortality 2006–2010. Geneva (WHO/IVB/05.11)
World Health Organization (2005) World Health Report 2005. Statistical Annex Tables 3 + 4, 191–192
World Health Organization (2005) Laboratory surveillance for wild and vaccine-derived polioviruses, January 2004–June 2005. Wkly Epidemiol Rec 80: 333–340
Bresee JS, Glass RI, Fang ZY(2004) First report from the Asian Rotavirus Surveillance Network. Emerg Infect Dis J 10: 988–995
World Health Organization (2004) Acute flaccid paralysis surveillance: a global platform for detecting and responding to priority infectious diseases. Wkly Epidemiol Rec 79: 425–433
World Health Organization (2006) Challenges in global immunization and the Global Immunization Vision and Strategy 2006–2015. Wkly Epidemiol Rec 81: 190–195
Hardy I, Dittman S, Sutter R (1996) Current situation and control strategies for resurgence of diphtheria in newly independent states of the former Soviet Union. Lancet 347: 1739–1744
Tangermann RH, Hull HF, Jafari H, Nkowane B, Everts H, Aylward RB (2000) Eradication of poliomyelitis in countries affected by conflict. Bull World Health Organ 78: 330–338
Gwatkin D, Davidson A, Yazbeck A, Wagstaff A (eds) (2005) Reaching the Poor with Health, Nutrition and Population Services: What Works, What Doesn’t, and Why. World Bank, Washington, D.C.
Technical consultation on imbalances in the health workforce (2002) WHO/EIP/OSD/02.3. World Health Organization, Geneva
World Health Organization (2000) Key elements for improving supplementary immunization activities for polio eradication. World Health Organization, Geneva WHO/V&B/00.22
World Health Assembly (1988) Global eradication of poliomyelitis by the year 2000. World Health Organization, Geneva (Resolution WHA41.28)
Dowdle WR, Cochi SL (2002) Global eradication of poliovirus: history and rationale. In: BL Semler, E Wimmer (eds): Molecular biology of picornaviruses. ASM Press, Washington, DC
World Health Organization (2001) Transmission of wild poliovirus type 2: apparent global interruption. Wkly Epidemiol Rec 76: 95–97
Kew OM, Sutter RW, de Gourville EM, Dowdle WR, Pallansch MA (2005) Vaccine-derived polioviruses and the endgame strategy for global polio eradication. Annu Rev Microbiol 59: 587–635
Dowdle WR, Hopkins DR (1998) The Eradication of Infectious Diseases: Dahlem Workshop Report. John Wiley & Sons, Chichester
De Quadros CA, Olive JM, Hersh BS (1996) Measles elimination in the Americas: evolving strategies. JAMA 275: 224–229
Expanded Programme on Immunisation (2006) Progress in reducing global measles deaths, 1999–2004. Wkly Epidemiol Rec 81: 90–94
Wolfson LJ, Strebel PM, Gacic-Dobo M, Hoekstra EJ, McFarland JW, Hersh BS (2007) Has the 2005 measles mortality reduction goal been achieved? A natural history modelling study. Lancet 369: 165–166
WHO and UNICEF (2005) WHO/UNICEF Joint Statement: Global plan for reducing measles mortality 2006–2010. Geneva (WHO/IVB/05.11)
PAHO (2004) XVI Meeting of the Technical Advisory Group on Vaccine Preventable Diseases, Mexico City. 3–5 November 2004. Final Report. Accessed June 30 at http://www.paho.org/English/AD/FCH/IM/TAG16_FinalReport_2004.pdf
World Health Organization (1999) Progress towards the global elimination of neonatal tetanus, 1990–1998. Wkly Epidemiol Rec 74: 73–80
Vandelaer J, Birmingham M, Gasse F, Kurian M, Shaw C, Garnier S (2003) Tetanus in developing countries: an update on the maternal and neonatal tetanus elimination initiative. Vaccine 21: 3442–3445
Adegbola RA, Secka O, Lahai G (2005) Elimination of Haemophilus influenzae type b (Hib) disease from the Gambia after the introduction of routine immunization with a Hib conjugate vaccine: a prospective study. Lancet 366: 144–150
Steinhoff MC (1997) Haemophilus influenzae type b infections are preventable everywhere. Lancet 349: 1186–1187
de Andrade AL, de Andrade JG, Martelli CM, e Silva SA, de Oliveira RM, Costa MS, Laval CB, Ribeiro LH, Di Fabio JL (2004) Effectiveness of Haemophilus influenzae b conjugate vaccine on childhood pneumonia: a casecontrol study in Brazil. Int J Epidemiol 33: 173–178
Gessner BD, Sutanto A, Linehan M (2005). Incidences of vaccine-preventable Haemophilus influenzae type b pneumonia and meningitis in Indonesian children: hamlet-randomized vaccine-probe trial. Lancet 365: 43–52
World Health Organization (2006) Conclusions and recommendations from the Strategic Advisory Group of Experts to the Department of Immunization, Vaccines and Biologicals. Wkly Epidemiol Rec 81: 2–11
Kane MA, Clements J, Hu D (1993) Hepatitis B. In: DT Jamison, WH Mosley, AR Measham, J Bobadilla (eds): Disease Control Priorities in Developing Countries. Oxford University Press, New York, 321–330
Van Damme P, Kane M, Meheus A (1997) Integration of hepatitis B vaccination into national immunization programmes. BMJ 314: 1033–1037
Chang MH, Chen CJ, Lai MS, Hsu HM, Wu TC, Kong MS, Liang DC, Shau WY, Chen DS (1997) Universal hepatitis B vaccination in Taiwan and the incidence of hepatocellular carcinoma in children. N Engl J Med 336:1855–1859
Margolis HS, Alter MJ, Hadler SC (1991) Hepatitis B: evolving epidemiology and implications for control. Semin Liver Dis 11: 84–92
Chen HL, Chang MH, Ni YH, Hsu HY, Lee PI, Lee CY, Chen DS (1996) Seroepidemiology of hepatitis B virus infection in children: Ten years of mass vaccination in Taiwan. JAMA 276: 906–908
World Health Organization (2006) Progress in the control of yellow fever in Africa. WHO Wkly Epidemiol Rec 80: 50–54
World Bank (1993) World development report 1993: investing in health. Oxford University Press, New York
Monath TP, Nasidi A (1993) Should YF vaccine be included in the expanded program of immunization in Africa? A cost-effectiveness analysis for Nigeria, Am J Trop Med Hyg 48: 274–299
Williams BG, Gouws E, Boschi-Pinto C, Bryce J, Dye C (2002) Estimates of world-wide distribution of child deaths from acute respiratory infections. Lancet Infect Dis 2: 25–32
Madhi SA, Peterson K, Madhi A, Wasas A, Klugman KP (2000) Impact of human immunodeficiency virus type 1 on the disease spectrum of Streptococcus pneumoniae in South African children. Pediatr Infect Dis J 19: 1141–1147
Fedson DS, Musher DM (2004) Pneumococcal polysaccharide vaccine. In: S Plotkin, W Orenstein (eds): Vaccines, 4th edn. Saunders, Philadelphia, 529–588
Whitney CG, Farley MM, Hadler J (2003) Decline in invasive pneumococcal disease after the introduction of protein-polysaccharide conjugate vaccine. N Engl J Med 348: 1737–1746
Centers for Disease Control and Prevention (CDC) (2005) Direct and indirect effects of routine vaccination of children with 7-valent pneumococcal conjugate vaccine on incidence of invasive pneumococcal disease: United States, 1998–2003. MMWR Morb Mortal Wkly Rep 54: 893–897
Ray GT, Whitney CG, Fireman BH, Ciuryla V, Black SB (2006) Cost-effectiveness of pneumococcal conjugate vaccine: evidence from the first 5 years of use in the United States incorporating herd effects. Pediatr Infect Dis J 25: 494–501
Salo H, Sintonen H, Kilpi T, Linna M, Nohynek H, Verho J, Nuorti PJ (2005) Economic evaluation of pneumococcal conjugate vaccination in Finland. Scand Infect Dis J 37: 821–832
Dagan R (2004) The potential of pneumococcal conjugate vaccines to reduce antibiotic resistance. Adv Expe Med Biol 549: 211–219
Cutts FT, Zaman SM, Enwere G (2005) Efficacy of nine-valent pneumococcal conjugate vaccine against pneumonia and invasive pneumococcal disease in The Gambia: randomised, double-blind, placebo-controlled trial. Lancet 365: 1139–1146
Ray GT, Whitney CG, Fireman BH, Ciuryla V, Black SB (2006) Pneumococcal vaccination in developing countries. Lancet 367: 1880–1882
Schmitt HJ, Booy R, Weil-Olivier C, Van Damme P, Cohen R, Peltola H (2003) Child vaccination policies in Europe: a report from the Summits of Independent European Vaccination Experts. Lancet Infect Dis 3: 103–108
Baltimore RS (2006) Recent trends in meningococcal epidemiology and current vaccine recommendations. Curr Opin Pediatr 18: 58–63
Centers for Disease Control and Prevention (CDC) (2005) Prevention and control of meningococcal disease. Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR Recomm Rep 54(RR-7): 1–21
World Health Organization (2005) World Health Report 2004. WHO, Geneva
Baseman JG, Koutsky LA (2005) The epidemiology of human papillomavirus infections. J Clin Virol 32S: S16–S24
Parikh S, Brennan P, Boffetta P (2003) Meta-analysis of social inequality and the risk of cervical cancer. Int J Cancer 105: 687–691
Jansen KU, Shaw AR (2004) Human papillomavirus vaccines and prevention of cervical cancer. Annu Rev Med 55: 19–31
Fine PE (1995) Variation in protection by BCG: implications of and for heterologous immunity. Lancet 346: 1339–1345
Kaufmann SH (2006) Envisioning future strategies for vaccination against tuberculosis. Nat Rev Immunol 6: 699–704
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Birkhäuser Verlag Basel/Switzerland
About this chapter
Cite this chapter
Tangermann, R.H., Nohynek, H., Eggers, R. (2007). Global control of infectious diseases by vaccination programs. In: Schroten, H., Wirth, S. (eds) Pediatric Infectious Diseases Revisited. Birkhäuser Advances in Infectious Diseases. Birkhäuser Basel. https://doi.org/10.1007/978-3-7643-8099-1_1
Download citation
DOI: https://doi.org/10.1007/978-3-7643-8099-1_1
Publisher Name: Birkhäuser Basel
Print ISBN: 978-3-7643-7997-1
Online ISBN: 978-3-7643-8099-1
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)