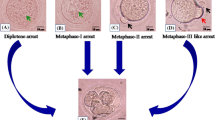
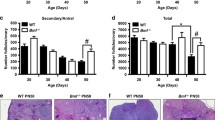
Abstract
More than 99.9% of the ovarian follicles present at birth never reach ovulation. The most common fate of follicles is instead to undergo atresia, which has been shown to be mediated by a highly organised type of cell death called apoptosis or programmed cell death (Hsueh et al. 1994; Billig et al. 1996; Kaipia and Hsueh 1997). The final tuning of the large number of growing follicles to a single ovulatory follicle is primarily achieved by cell death of granulosa cells (Hsueh et al. 1994; Reynaud and Driancourt 2000).
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Antonsson B, Martinou JC (2000) The Bc1–2 protein family Experimental Cell Res 256 50–57
Baker TG (1963) A quantitative and cytological study of germ cells in human ovaries. Proceedings of the Royal Society of London. Biol Sci 158 417–433
Billig H, Furuta I, Hsueh AJ (1993) Estrogens inhibit and androgens enhance ovarian granulosa cell apoptosis. Endocrinology 133: 2204–2212
Billig H, Chun SY, Eisenhauer K, Hsueh AJ (1996) Gonadal cell apoptosis: hormone-regulated cell demise. Hum Reprod Update 2: 103–117
Braw RH, Tsafriri A (1980) Follicles explanted from pentobarbitone-treated rats provide a model for atresia. J Reprod Fertil 59: 259–265
Chun SY, Billig H, Tilly JL, Furuta I, Tsafriri A, Hsueh AJ (1994) Gonadotropin suppression of apoptosis in cultured preovulatory follicles: mediatory role of endogenous insulin-like growth factor I. Endocrinology 135: 1845–1853
Chun SY, Eisenhauer KM, Kubo M, Hsueh AJ (1995) Interleukin-1 beta suppresses apoptosis in rat ovarian follicles by increasing nitric oxide production. Endocrinology 136: 3120–3127
Chun SY, Eisenhauer KM, Minami S, Billig H, Perlas E, Hsueh AJ (1996) Hormonal regulation of apoptosis in early antral follicles: follicle-stimulating hormone as a major survival factor. Endocrinology 137: 1447–1456
Danilovich NA, Bartke A, Winters TA (2000) Ovarian follicle apoptosis in bovine growth hormone transgenic mice. Biol of Reprod 62: 103–107
Dong J, Albertini DF, Nishimori K, Kumar TR, Lu N, Matzuk MM (1996) Growth differentiation factor-9 is required during early ovarian folliculogenesis. Nature 383: 531–535
Driancourt MA, Reynaud K, Cortvrindt R, Smitz J (2000) Roles of KIT and KIT LIGAND in ovarian function. Rev Reprod 5: 143–152
Dunkel L, Tilly JL, Shikone T, Nishimori K, Hsueh AJ (1994) Follicle-stimulating hormone receptor expression in the rat ovary: increases during prepubertal development and regulation by the opposing actions of transforming growth factors beta and alpha. Biol Reprod 50: 940–948
Eisenhauer KM, Chun SY, Billig H, Hsueh AJ (1995) Growth hormone suppression of apoptosis in preovulatory rat follicles and partial neutralization by insulin-like growth factor binding protein. Biol Reprod 53: 13–20
Fanjul LF, Ruiz de Galarreta CM, Hsueh AJ (1983) Progestin augmentation of gonadotropin-stimulated progesterone production by cultured rat granulosa cells. Endocrinol 112: 405–407
Fujinaga H, Yamoto M, Shikone T, Nakano R (1994) FSH and LH up-regulate epidermal growth factor receptors in rat granulosa cells. J Endocrinol 140: 171–177
Gougeon A (1986) Dynamics of follicular growth in the human: a model from preliminary results. Hum Reprod 1: 81–87
Hengartner MO (2000) The biochemistry of apoptosis. Nature 407: 770–776
Hirshfield AN (1991) Development of follicles in the mammalian ovary. Int Rev Cytol 124: 43–101
Hsu SY, Hsueh AJ (2000) Tissue-specific Bel-2 protein partners in apoptosis: An ovarian paradigm. Physiol Rev 80: 593–614
Hsueh AJ, Billig H, Tsafriri A (1994) Ovarian follicle atresia: a hormonally controlled apoptotic process. Endocrine Rev 15: 707–724
Iwai T, Nanbu Y, Iwai M, Tali S, Fujii S, Mori T (1990) Immunohistochemical localization of oestrogen receptors and progesterone receptors in the human ovary throughout the menstrual cycle. Virchows Arch A Pathol Anat Histopathol 417: 369–375
Kaipia A, Hsueh AJ (1997) Regulation of ovarian follicle atresia. Ann Rev Physiol 59: 349–363
Ko C, Park-Sarge OK (2000) Progesterone receptor activation Mediates LH-induced type-I pituitary adenylate cyclase activating polypeptide receptor (PAC(1)) gene expression in rat granulosa cells. Biochem Biophys Res Commun 277: 270–279
Ko C, In YH, Park-Sarge OK (1999) Role of progesterone receptor activation in pituitary adenylate cyclase activating polypeptide gene expression in rat ovary. Endocrinology 140: 5185–5194
Lee J, Park HJ, Choi HS, Kwon HB, Arimura A, Lee BJ, Choi WS, Chun SY (1999) Gonadotropin stimulation of pituitary adenylate cyclase-activating polypeptide ( PACAP) messenger ribonucleic acid in the rat ovary and the role of PACAP as a follicle survival factor. Endocrinology 140: 818–826
Lydon JP, DeMayo FJ, Funk CR, Mani SK, Hughes AR, Montgomery CA Jr, Shyamala G, Conneely OM, O’Malley BW (1995) Mice lacking progesterone receptor exhibit pleiotropic reproductive abnormalities. Genes Dev 9: 2266–2278
Lydon JP, DeMayo FJ, Conneely OM, O’Malley BW (1996) Reproductive phenotypes of the progesterone receptor null mutant mouse. J Steroid Biochem Mol Biol 56: 67–77
Makrigiannakis A, Coukos G, Christofidou-Solomidou M, Montas S, Coutifaris C (2000) Progesterone is an autocrine/paracrine regulator of human granulosa cell survival in vitro. Ann NY Acad Sci 900: 16–25
Matikianen T, Perez GI, Zheng TS, Kluzak TS, Rueda BR, Flavell RA, Tilly JL (2001) Caspase-3 gene knockout defines cell lineage specificity for programmed cell death signaling in the ovary. Endocrinology 142: 2468–2467
McGee EA (2000) The regulation of apoptosis in preantral ovarian follicles. Biol Signals Recept 9: 81–86
McGee EA, Hsueh AJ (2000) Initial and cyclic recruitment of ovarian follicles. Endocrine Rev 21: 200–214
McGee E, Spears N, Minami S, Hsu SY, Chun SY, Billig H, Hsueh Ai (1997a) Preantral ovarian follicles in serum-free culture: suppression of apoptosis after activation of the cyclic guanosine 3’,5’-monophosphate pathway and stimulation of growth and differentiation by follicle-stimulating hormone. Endocrinology 138: 2417–2424
McGee EA, Perlas E, LaPolt PS, Tsafriri A, Hsueh AJ (1997b) Follicle-stimulating hormone enhances the development of preantral follicles in juvenile rats. Biol Reprod 57: 990–998
McGee EA, Chun SY, Lai S, He Y, Hsueh Ai (1999) Keratinocyte growth factor promotes the survival, growth, and differentiation of preantral ovarian follicles. Fertil Steril 71: 732–738
McGee EA, Smith R, Spears N, Nachtigal MW, Ingraham H, Hsueh AJ (2001) Mullerian inhibitory substance induces growth of rat preantral ovarian follicles. Biol Reprod 64: 293–298
Morita Y, Tilly JL (1999) Oocyte apoptosis: like sand through an hourglass. Develop Biol 213 1–17
Mulac-Jericevic B, Mullinax RA, DeMayo FJ, Lydon JP, Conneely OM (2000) Subgroup of reproductive functions of progesterone mediated by progesterone receptor-B isoform. Science 289: 1751–1754
Mulac-Jericevic B, Mullinax RA, Conneely OM (2001) Phenotypic analysis of mice lacking progesterone receptor B isoform. The Endocrine Society’s 83rd Annual Meeting (Abstract Pl—P67)
Murray AA, Molinek MD, Baker SJ, Kojima FN, Smith MF, Hillier SG, Spears N (2001) Role of ascorbic acid in promoting follicle integrity and survival in intact mouse ovarian follicles in vitro. Reproduction 121: 89–96
Natraj U, Richards JS (1993) Hormonal regulation, localization, and functional activity of the progesterone receptor in granulosa cells of rat preovulatory follicles. Endocrinology 133: 761–769
Park OK, Mayo KE (1991) Transient expression of progesterone receptor messenger RNA in ovarian granulosa cells after the preovulatory luteinizing hormone surge. Mol Endocrinol 5: 967–978
Peluso JJ, Pappalardo A (1998) Progesterone mediates its anti-mitogenic and anti-apoptotic actions in rat granulosa cells through a progesterone-binding protein with gamma aminobutyric acidA receptor-like features. Biol Reprod 58: 1131–1137
Press MF, Greene GL (1988) Localization of progesterone receptor with monoclonal antibodies to the human progestin receptor. Endocrinology 122: 1165–1175
Rannikki AS, Zhang FP, Huhtaniemi IT (1995) Ontogeny of follicle-stimulating hormone receptor gene expression in the rat testis and ovary. Mol Cell Endocrinol 107: 199–208
Reynaud K, Driancourt MA (2000) Oocyte attrition. Mol Cell Endocrinol 163: 101–108
Robker RL, Russell DL, Espey LL, Lydon JP, O’Malley BW, Richards JS (2000) Progesterone-regulated genes in the ovulation process: ADAMTS-1 and cathepsin L proteases. Proc Natl Acad Sci USA 97: 4689–4694
Robles R, Tao X-J, Trbovich AM, Maravei DV, Nahum R, Perez GI, Tilly KL, Tilly JL (1999) Localization, regulation and possible consequences of apoptotic protease-activating factor-1 (Apaf-1) expression in granulosa cells of the mouse ovary. Endocrinology 140: 2641–2644
Robles R, Morita Y, Mann KK, Perez GI, Yang S, Matikainen T, Sherr DH, Tilly JL (2000) The aryl hydrocarbon receptor, a basic helix-loop-helix transcription factor of the PAS gene family, is required for normal ovarian germ cell dynamics in the mouse. Endocrinology 141: 450–453
Scaffidi C, Fulda S, Srinivasan A, Friesen C, Li F, Tomaselli KJ, Debatin KM, Krammer PH, Peter ME (1998) Two CD95 (APO-1/Fas) signaling pathways. EMBO J 17: 1675–1687
Svanberg B, Billig H (1999) Isolation of differentially expressed mRNA in ovaries after estrogen withdrawal in hypophysectomized diethylstilbestrol-treated rats: increased expression during apoptosis. J Endocrinol 163 309–316
Svensson PA, Johnson MS, Ling C, Carlsson LM, Billig H, Carlsson B (1999) Scavenger receptor class B type I in the rat ovary: possible role in high density lipoprotein cholesterol uptake and in the recognition of apoptotic granulosa cells. Endocrinology 140: 2494–2500
Svensson EC, Markström E, Andersson M, Billig H (2000) Progesterone receptor-mediated inhibition of apoptosis in granulosa cells isolated from rats treated with human chorionic gonadotropin. Biol Reprod 63: 1457–1464
Svensson EC, Markstrom E, Shao R, Andersson M, Billig H (2001) Progesterone receptor antagonists Org 31710 and RU 486 increase apoptosis in human periovulatory granulosa cells. Fertil Steril 76: 1225–1231
Talbert GB, Meyer RK, McShan WH (1951) Effect of hypophysectomy at the beginning of proestrus on maturing follicles in the ovary of the rat. Endocrinology 49: 687–694
Tilly JL, Billig H, Kowalski KI, Hsueh AJ (1992) Epidermal growth factor and basic fibroblast growth factor suppress the spontaneous onset of apoptosis in cultured rat ovarian granulosa cells and follicles by a tyrosine kinase-dependent mechanism. Mol Endocrinol 6: 1942–1950
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2002 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Billig, H., Markström, E., Svensson, E.C., Shao, R., Friberg, A. (2002). Follicular Development and Apoptosis. In: Eppig, J., Hegele-Hartung, C., Lessl, M. (eds) The Future of the Oocyte. Ernst Schering Research Foundation Workshop, vol 41. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-04960-0_3
Download citation
DOI: https://doi.org/10.1007/978-3-662-04960-0_3
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-662-04962-4
Online ISBN: 978-3-662-04960-0
eBook Packages: Springer Book Archive