Summary
The glial reaction is generally considered to be a consequence of neuronal death in neurodegenerative diseases such as Alzheimer’s disease, Hunting-ton’s disease and Parkinson’s disease.
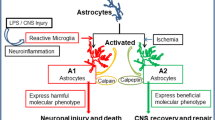
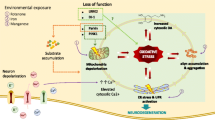
In Parkinson’s disease, postmortem examination reveals that the loss of dopaminergic neruons in the substantia nigra is associated with massive astroglisois and the presence of activated nucriglial cells. Recent evidence suggests that the disease may progress even when the initial cause of neuronal degeneration has diasppeared, implying that toxic subdtances released by glial cells may be involved in neuronal dageneration. Glial cells can release various compounds, including pro-inflammatory cytokines. These substances may act on specific receptors, located on the dopaminergic neurons, that contain intracytoplasmic death domains and are involved in apoptosis. Alternatively, since cytokines are known to induce the expression of nitric oxide via the induction and activation of the low affinity IgE receptor CD23, the gradual release of nitric oxide from glial cells may account for the increased oxidative stress, protein nitration, altered iron homeostasis and blood vessel alterations reported in the disease. In turn, such cellular alterations may provoke the degeneration of dopaminergic neurons. The exact cascade of events leading to neuronal degeneration in Parkinson’s disease is not known but may involve activation of proteases such as caspase-3, which are known effectors of the cascade of events leading to nerve cell death.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
Agid Y (1991) Parkinson’s disease: pathophysiology. Lancet (Ed. Française) 337:1311–1324
Agid Y, Ruberg M, Javoy-Agid F, Hirsch E, Raisman-Vozari R, Vyas S, Faucheux B, Michel P, Kastner A, Blanchard V, Damier P, Villares J, Ping Zhang (1993) Are dopaminergic neurons selectively vulnerable to Parkinson’s disease? Adv Neurol 60: 148–164
Aloe L, Fiore M (1997) TNF-a expressed in the brain of transgenic mice lowers central tyrosine hydroxylase immunoreactiviy and alters grooming behavior. Neurosci Lett 238: 65–68
Anglade P, Vyas S, Javoy-Agid F, Herrero MT, Michel PP, Marquez J, Mouatt-Prigent A, Ruberg M, Hirsch EC, Agid Y (1997) Apoptosis and autophagy in nigral neurons of patients with Parkinson’s disease. Histol Histopathol 12: 25–31
Arock M, Le Goff L, Bécherel PA, Dugas B, Debré P, Mossalayi MD (1994) Involvement of FcepsillonRII/CD23 and L-arginine dependent pathway in IgE-mediated activation of human eosinophils. Biochem Biophys Res Commun 203: 265–271
Baker SJ, Reddy EP (1998) Modulation of life and death by the TNF receptor superfamily. Oncogene 17: 3261–3270
Boka G, Anglade P, Wallach D, Javoy-Agid F, Agid Y, Hirsch EC (1994) Immunocytochemical analysis of tumor necrosis factor and its receptors in Parkinson’s disease. Neurosci Lett 172: 151154
Bronstein DM, Perez-Otano I, Sun V, Mullis Sawin SB, Chan J, Wu GC, Hudson PM, Kong LY, Hong JS, McMillian MK (1995) Glia-dependent neurotoxicity and neuroprotection in mesencephalic cultures. Brain Res 704: 112–116
Damier P, Hirsch EC, Zhang P, Agid Y, Javoy-Agid F (1993) Glutathione peroxidase, glial cells and Parkinson’s disease. Neuroscience 52: 1–6
Damier P, Hirsch EC, Agid Y, Graybiel AM (1999a) The substantia nigra of the human brain: I. Nigrosomes and the nigral matrix, a compartmental organization based on calbindin D-28K immunohistochemistry. Brain 122: 1421–1436
Damier P, Hirsch EC, Agid Y, Graybiel AM (1999b) The substantia nigra of the human brain: II. Patterns of loss of dopamine-containing neurons in Parkinson’s disease. Brain 122: 1437–1448
Dehmer T, Lindenau J, Haid S, Dichgans J, Schulz JB (2000) Deficiency of inducible nitric oxide synthase protects against MPTP toxicity in vivo. J Neurochem 74: 2213–2216
Delespesse G, Sutter U, Mossalayi MD, Bettler B, Sarfati M, Hofstetter H, Kilchherr E, Debré P, Dalloul AH (1991) Expression, structure, and function of the CD23 antigen. Adv Immunol 49: 149–191
Dugas B, Mossalayi MD, Damais C, Kolb J-P (1995) Nitric oxide production by human monocytes: evidence for a role of CD23. Immunol Today 16: 574–580
Good PF, Hsu A, Werner P, Perl DP, Olanow CW (1998) Protein nitration in Parkinson’s disease. J Neuropathol Exp Neurol 57: 338–342
Hartmann A, Hunot S, Michel PP, Muriel M-P, Vyas S, Faucheux BA, Mouatt-Prigent A, Turmel E, Evan GI, Agid Y, Hirsch EC (2000) Caspase-3: a vulnerability factor and final effector in apoptotic death of dopaminergic neurons in Parkinson’s disease. Proc Natl Acad Sci 97: 2875–2880
Hartmann A, Hirsch EC (2001) Parkinson’s disease: the apoptosis hypothesis revisited. Adv Neurol 86: 143–153
Hirsch EC (1993) Does oxidative stress participate in nerve cell death in Parkinson’s disease? Eur Neurol 33: 52–59
Hirsch EC, Faucheux BA (1998) Iron metabolism and Parkinson’s disease. Mov Disord 13 (S1): 3945
Hirsch EC, Graybiel AM, Agid Y (1988) Melanized dopaminergic neurons are differentially susceptible to degeneration in Parkinson’s disease. Nature 334: 345–348
Hunot S, Boissière F, Faucheux B, Brugg B, Mouatt-Prigent A, Agid Y, Hirsch EC (1996) Nitric oxide synthase and neuronal vulnerability in Parkinson’s disease. Neuroscience 72: 355–363
Hunot S, Dugas N, Faucheux B, Hartmann A, Tardieu M, Debré P, Agid Y, Dugas B, Hirsch EC (1999) Fc(epsilon)RII/CD23 is expressed in Parkinson’s disease and induces, in vitro, production of nitric oxide and tumor necrosis factor-alpha in glial cells. J Neurosci 19: 3440–3447
Jenner P (1996) Oxidative stress in Parkinson’s disease and other neurodegenerative disorders. Pathol Biol 44: 57–64
Jenner P (1998) Oxidative mechanisms in nigral cell death in Parkinson’s disease. Mov Disord 13: S24 - S34
Jenner P, Olanow CW (1996) Oxidative stress and the pathogenesis of Parkinson’s disease. Neurology 47: S161 - S170
Jenner P, Olanow CW (1998) Understanding cell death in Parkinson’s disease. Ann Neurol 44: S72 - S84
Juckett M, Zheng Y, Yuan H, Pastor T, Antholine W, Weber M, Vercellotti G (1998) Heme and the endothelium. Effects of nitric oxide on catalytic iron and heme degradation by heme oxygenase. J Biol Chem 273: 288–297
Kingsbury AE, Marsden CD, Foster OJ (1998) DNA fragmentation in human substantia nigra: apoptosis or perimortem effect? Mov Disord 13: 877–884
Langston JW, Forno LS, Tetrud J, Reeves AG, Kaplan JA, Karluk D (1999) Evidence of active nerve cell degeneration in the substantia nigra of humans years after 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine exposure. Ann Neurol 46: 598–605
Liberatore GT, Jackson-Lewis V, Vukosavic S, Mandir AS, Vila M, McAuliffe WG, Dawson VL, Dawson TM, Przedborski S (1999) Inducible nitric oxide synthase stimulates dopaminergic neurodegeneration in the MPTP model of Parkinson’s disease. Nature Med 5: 1403–1409
McGeer PL, Itagaki S, Boyes BE, McGeer EG (1988) Reactive microglia are positive for HLA-DR in the substantia nigra of Parkinson’s and Alzheimer’s disease brains. Neurology 38: 1285–1291
McNaught KS, Jenner P (1999) Altered glial function causes neuronal death and increases neuronal susceptibility to 1-methyl-4-phenylpyridinium and 6-hydroxydopamine-induced toxicity in astrocytic/ventral mesencephalic co-cultures. J Neurochem 73: 2469–2476
Mochizuki H, Goto K, Mori H, Mizuno Y (1996) Histochemical detection of apoptosis in Parkinson’s disease. J Neurol Sci 137: 120–123
Mogi M, Harada M, Riederer P, Narabayashi H, Fujita K, Nagatsu T (1994a) Tumor necrosis factor-a ( TNF-a) increases both in the brain and in the cerebrospinal fluid from parkinsonian patients. Neurosci Lett 165: 208–210
Mogi M, Harada M, Kondo J, Riederer P, Inagaki H, Minami M, Nagatsu T (1994b) Interleukin-1 ß, interleukin-6, epidermal growth factor and transforming growth factor-a are elevated in the brain from parkinsonian patients. Neurosci Lett 180: 147–150
Mogi M, Harada M, Kondo T, Riederer P, Nagatsu T (1996 a) Interleukin-2 but not basic fibroblast growth factor is elevated in parkinsonian brain. J Neural Transm 103: 1077–1081
Mogi M, Harada M, Narabayashi H, Inagaki H, Minami M, Nagatsu T (1996b) Interleukin (IL)-1 ß, IL-2, IL-4, IL-6 and transforming growth factor-a levels are elevated in ventricular cerebrospinal fluid in juvenile parkinsonism and Parkinson’s disease. Neurosci Lett 211: 13–16
Mossalayi MD, Paul-Eugène N, Ouaaz F, Arock M, Kolb J-P, Kilchherr E, Debré P, Dugas B (1994) Involvement of FcepsilonRll/CD23 and L-arginine-dependent pathway in IgE-mediated stimulation of human monocyte functions. Int Immunol 6: 931–934
Nicotera P, Leist M, Fava E, Berliocchi L, Volbracht C (2000) Energy requirement for caspase activation and neuronal cell death. Brain Pathol 10: 276–282
Olanow CW (1992) An introduction of the free radical hypothesis in Parkinson’s disease. Ann Neurol 32: S2 - S9
Olanow CW (1997) Attempts to obtain neuroprotection in Parkinson’s disease. Neurology 49: S26 - S33
Owen AD, Schapira AH, Jenner P, Marsden CD (1996) Oxidative stress and Parkinson’s disease. Ann NY Acad Sci 786: 217–223
Przedborski S, Jackson-Lewis V (1998) Experimental developments in movement disorders: update on proposed free radical mechanisms. Curr Opin Neurol 11: 335–339
Qureshi GA, Baig S, Bednar I, Sodersten P, Forsberg G, Siden A (1995) Increased cerebrospinal fluid concentration of nitrite in Parkinson’s disease. NeuroReport 6: 1642–1644
Tatton NA, Maclean-Fraser A, Tatton WGPDP, Olanow CW (1998) A fluorescent double-labeling method to detect and confirm apoptotic nuclei in Parkinson’s disease. Ann Neurol 44:5142S148
Tompkins MM, Basgall EJ, Zamrini E, Hill WD (1997) Apoptotic-like changes in Lewy-body-asso- ciated disorders and normal aging in substantia nigral neurons. Am J Pathol 150: 119–131
Turmel H, Hartmann A, Parain K, Douhou A, Srinivasan A, Agid Y, Hirsch EC (2001) Caspase-3 activation in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine ( MPTP)-treated mice. Mov Disord 16: 185–189
Vandenabeele P, Declercq W, Beyaert R, Fiers W (1994) Two tumor necrosis factor receptors: structure and function. Trends Cell Biol 5: 392–399
Werth JL, Deshmukh M, Cocabo J, Johnson EM Jr, Rothman SM (2000) Reversible physiological alterations in sympathetic neurons deprived of NGF but protected from apoptosis by caspase inhibition or Bax deletion. Exp Neurol 161: 203–211
Wullner U, Kornhuber J, Weller M, Schulz JB, Loschmann PA, Riederer P, Klockgether T (1999) Cell death and apoptosis regulating proteins in Parkinson’s disease: a cautionary note. Acta Neuropathol (Berl) 97: 408–412
Youdim MB, Ben Shachar D, Riederer P (1993) The possible role of iron in the etiopathology of Parkinson’s disease. Mov Disord 8: 1–12
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2001 Springer-Verlag Berlin Heidelberg
About this paper
Cite this paper
Hirsch, E.C. (2001). Apoptosis, Glial Cells and Parkinson’s Disease. In: Henderson, C.E., Green, D.R., Mariani, J., Christen, Y. (eds) Neuronal Death by Accident or by Design. Research and Perspectives in Neurosciences. Springer, Berlin, Heidelberg. https://doi.org/10.1007/978-3-662-04333-2_9
Download citation
DOI: https://doi.org/10.1007/978-3-662-04333-2_9
Publisher Name: Springer, Berlin, Heidelberg
Print ISBN: 978-3-642-07517-9
Online ISBN: 978-3-662-04333-2
eBook Packages: Springer Book Archive