Abstract
The marriage of maleic anhydride and vegetable oils as a unique class of environmentally benign macromonomers and sustainable polymers has attracted a great deal of interests in the last few decades. The rich chemistry and versatile reactivity of maleic anhydride, combined with the biodegradable and renewable nature of vegetable oils, have rendered infinite possibilities for the maleic anhydride–vegetable oil derivatives. This section provides a survey of the recent literatures on the synthetic routes of the various maleic anhydride derivatives based on the reactive sites in the vegetable oil, as well as the properties and applications of these novel macromonomers and polymers.
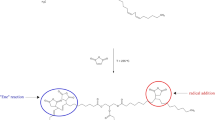
Maleimides and derivatives are important building blocks in chemical synthesis, and some of them demonstrate biological activity. A special feature of the reactivity of maleimides is their susceptibility to additions across the double bond, either by Michael additions or via Diels–Alder reactions. The carbon–carbon double bond in maleimide is also capable of free-radical or anionic polymerization or copolymerization to produce functional polymers that are utilized in high temperature applications, such as adhesives in the semiconductor industry. Biological applications of maleimide derivatives result from their high and specific reactivity/binding toward the thiol groups in biological substrate (e.g., chemical probes), site-directed modification for proteins and peptides, bioconjugates, and prodrugs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Lu Y, Larock RC (2009) Novel polymeric materials from vegetable oils and vinyl monomers: preparation, properties, and applications. ChemSusChem 2(2):136–147
Miao S, Wang P, Su Z, Zhang S (2014) Vegetable-oil-based polymers as future polymeric biomaterials. Acta Biomater 10(4):1692–1704
Mazo P, Estenoz D, Sponton M, Rios L (2012) Kinetics of the transesterification of castor oil with maleic anhydride using conventional and microwave heating. J Am Oil Chem Soc 89(7):1355–1361
Saied MA, Mansour SH, Eweis M, El-Sabee MZ, Saad ALG, Abdel Nour KN (2008) Some biophysical properties of castor oil esterified with some acid anhydrides. Eur J Lipid Sci Technol 110(10):926–934
Saied MA, Mansour SH, El Sabee MZ, Saad ALG, Abdel-Nour KN (2012) Some electrical and physical properties of castor oil adducts dissolved in 1-propanol. J Mol Liq 172:1–7
Wang HJ, Rong MZ, Zhang MQ, Hu J, Chen HW, Czigany T (2008) Biodegradable foam plastics based on castor oil. Biomacromolecules 9(2):615–623
Wang H, Rong M, Zhang M (2014) Plastic foams based on castor oil modified with the reaction product of maleic anhydride and β-hydroxyethyl acrylate. Gaofenzi Cailiao Kexue Yu Gongcheng 30(2):21–26
Sponton M, Casis N, Mazo P, Raud B, Simonetta A, Rios L, Estenoz D (2013) Biodegradation study by Pseudomonas sp. of flexible polyurethane foams derived from castor oil. Int Biodeterior Biodegrad 85:85–94
Echeverri DA, Perez WA, Rios LA (2013) Maleinization of soybean oil glycerides obtained from biodiesel-derived crude glycerol. J Am Oil Chem Soc 90(12):1877–1882
Echeverri DA, Jaramillo F, Rios LA (2015) Curing copolymerization kinetics of styrene with maleated castor oil glycerides obtained from biodiesel-derived crude glycerol. J Appl Polym Sci 132(4)
Echeverri DA, Perez WA, Rios LA (2013) Synthesis of maleated-castor oil glycerides from biodiesel-derived crude glycerol. Ind Crops Prod 49:299–303
Can E, Wool RP, Kusefoglu S (2006) Soybean and castor oil based monomers: synthesis and copolymerization with styrene. J Appl Polym Sci 102(3):2433–2447
Can E, Kusefoglu S, Wool RP (2001) Rigid, thermosetting liquid molding resins from renewable resources. I. Synthesis and polymerization of soybean oil monoglyceride maleates. J Appl Polym Sci 81(1):69–77
Can E, Kusefoglu S, Wool RP (2002) Rigid thermosetting liquid molding resins from renewable resources. II. Copolymers of soybean oil monoglyceride maleates with neopentyl glycol and bisphenol A maleates. J Appl Polym Sci 83(5):972–980
Bora MM, Gogoi P, Deka DC, Kakati DK (2014) Synthesis and characterization of yellow oleander (Thevetia peruviana) seed oil-based alkyd resin. Ind Crops Prod 52:721–728
Bora MM, Deka R, Ahmed N, Kakati DK (2014) Karanja (Millettia pinnata (L.) Panigrahi) seed oil as a renewable raw material for the synthesis of alkyd resin. Ind Crops Prod 61:106–114
Boruah M, Gogoi P, Adhikari B, Dolui SK (2012) Preparation and characterization of Jatropha curcas oil based alkyd resin suitable for surface coating. Prog Org Coat 74(3):596–602
Dutta N, Karak N, Dolui SK (2004) Synthesis and characterization of polyester resins based on Nahar seed oil. Prog Org Coat 49(2):146–152
Ahmad S, Ashraf SM, Zafar F (2007) Development of linseed oil based polyesteramide without organic solvent at lower temperature. J Appl Polym Sci 104(2):1143–1148
Ahmad S, Ashraf SM, Naqvi F, Yadav S, Hasnat A (2003) A polyesteramide from Pongamia glabra oil for biologically safe anticorrosive coating. Prog Org Coat 47(2):95–102
Mahapatra SS, Karak N (2004) Synthesis and characterization of polyesteramide resins from Nahar seed oil for surface coating applications. Prog Org Coat 51(2):103–108
Pramanik S, Sagar K, Konwar BK, Karak N (2012) Synthesis, characterization and properties of a castor oil modified biodegradable poly(ester amide) resin. Prog Org Coat 75(4):569–578
Pramanik S, Konwarh R, Sagar K, Konwar BK, Karak N (2013) Bio-degradable vegetable oil based hyperbranched poly(ester amide) as an advanced surface coating material. Prog Org Coat 76(4):689–697
Thames SF, Smith OW, Evans JM, Dutta S, Chen L (2005) Functionalized vegetable oil derivatives used in latex and coating compositions and their preparation. US7361710
Musa OM, Shih JS (2010) Performance-boosting UV-absorbing compounds for personal care and performance compositions. US8557226
Candy L, Vaca-Garcia C, Borredon E (2005) Synthesis and characterization of oleic succinic anhydrides: structure-property relations. J Am Oil Chem Soc 82(4):271–277
Tomoda H, Sugimoto Y, Tani Y, Watanabe S (1998) Characteristic properties of cutting fluid additives derived from the reaction products of hydroxyl fatty acids with some acid anhydrides. J Surfact Deterg 1(4):533–537
Bertz SH, Miksza FM, Zucker E (2001) Adduct characterized by absence of free maleic anhydride; personal care products. 4p
Espana JM, Sanchez-Nacher L, Boronat T, Fombuena V, Balart R (2012) Properties of biobased epoxy resins from epoxidized soybean oil (ESBO) cured with maleic anhydride (MA). J Am Oil Chem Soc 89(11):2067–2075
Gerbase AE, Petzhold CL, Costa APO (2002) Dynamic mechanical and thermal behavior of epoxy resins based on soybean oil. J Am Oil Chem Soc 79(8):797–802
Samper MD, Fombuena V, Boronat T, Garcia-Sanoguera D, Balart R (2012) Thermal and mechanical characterization of epoxy resins (ELO and ESO) cured with anhydrides. J Am Oil Chem Soc 89(8):1521–1528
Huang Y, Pang L, Wang H, Zhong R, Zeng Z, Yang J (2013) Synthesis and properties of UV-curable tung oil based resins via modification of Diels-Alder reaction, nonisocyanate polyurethane and acrylates. Prog Org Coat 76(4):654–661
Ge Q, Wang H, She Y, Jiang S, Cao M, Zhai L, Jiang S (2015) Synthesis, characterization, and properties of acrylate-modified tung-oil waterborne insulation varnish. J Appl Polym Sci 132(10):41608/1–41608/8
Tran P, Seybold K, Graiver D, Narayan R (2005) Free radical maleation of soybean oil via a single-step process. J Am Oil Chem Soc 82(3):189–194
Force CG, Starr FS (1988) Vegetable oil adducts as emollients in skin and hair care products. US4740367
Bidulescu G, Stere EA, Tarko L (1986) Manufacture of maleimides from aromatic primary amines. RO89171A2
Yamamoto T, Mizuno S, Watanabe M (1994) Preparation of N-arylmaleimides from maleic anhydride and aromatic amines. JP06184104A
Yamamoto T, Mizuno S, Watanabe M (1995) Preparation and purification of N-substituted maleimide. JP07109258A
Yamamoto T, Mizuno S, Watanabe M (1995) Preparation of maleimide derivatives. JP07053513A
Fujita T, Irie T, Takayanagi Y, Narita T, Yano Y (1989) Preparation of N-substituted maleimides. DE3905872A1
Sudo I, Watabe Y (1992) Preparation of maleimides. JP04198167A
Oonuma Y, Chiba H, Kanayama K (1994) Preparation of maleimides. JP06016627A
Ikeda I, Yamashita W, Tamai S (1998) Preparation of (poly)maleimides as monomers. JP10175952A
Le Z-G, Chen Z-C, Hu Y, Zheng Q-G (2004) Organic reactions in ionic liquids: ionic liquid-promoted efficient synthesis of N-alkyl and N-arylimides. Synthesis 7:995–998
Reddy PY, Kondo S, Toru T, Ueno Y (1997) Lewis acid and hexamethyldisilazane-promoted efficient synthesis of N-alkyl- and N-arylimide derivatives. J Org Chem 62(8):2652–2654
Doi S, Takayanagi Y (1986) N-substituted maleimides. EP177031A1
Ueda H, Kita J, Kishino K (1993) Preparation of maleimides. JP05213869A
Wu K-C, Tung Y-L, Lee C-H, Dai J-C, Huang C-H (2003) Imidation method and catalysts for the production of maleimides from maleic anhydride and primary amines. US6630595
Manjula KS, Rai KML, Umesha KB, Babu MS, Jagadeesha RL (2003) Microwave-assisted synthesis of N-aryl maleimide under solvent free conditions. Bulg Chem Commun 35(3):192–194
Matsukawa M, Wada M, Minazu H, Furuya M, Nagata T (1991) Preparation of N-substituted maleimides. JP03011060A
Gaina V, Gaina C (2004) Dehydrochlorination of α-chlorosuccinimides, a new method for synthesis of high-purity maleimides. Mater Plast (Bucharest) 41(3):169–172
Walker MA (1994) The Mitsunobu reaction - a novel method for the synthesis of bifunctional maleimide linkers. Tetrahedron Lett 35(5):665–668
Ahn KD, Koo DI, Willson CG (1995) Synthesis and polymerization of t-Boc protected maleimide monomers - N-(t-butyloxycarbonyloxy) maleimide and N-p-(t-butyloxycarbonyloxy) phenyl-maleimide. Polymer 36(13):2621–2628
Wang ZY (1990) Syntheses of some N-alkylmaleimides. Synth Commun 20(11):1607–1610
Katritzky AR, Fan WQ, Li QL, Bayyuk S (1989) Novel chromophoric heterocycles based on maleimide and naphthoquinone. J Heterocyc Chem 26(4):885–892
Oishi T, Fujimoto M (1992) Synthesis and polymerization of N-4-N′-(alpha-methylbenzyl)aminocarbonylphenyl maleimide. J Polym Sci A Polym Chem 30(9):1821–1830
Oishi T, Kagawa K, Fujimoto M (1993) Synthesis and polymerization of N-N′-(alpha-methylbenzyl)amino carbonyl methyl maleimide. Macromolecules 26(1):24–29
Kitagawa O, Izawa H, Sato K, Dobashi A, Taguchi T, Shiro M (1998) Optically active axially chiral anilide and maleimide derivatives as new chiral reagents: synthesis and application to asymmetric Diels-Alder reaction. J Org Chem 63(8):2634–2640
Yanase M, Kagawa T (2005) Optically active polymaleimides, their manufacture, and uses for separation of optically active compounds. JP2005255795A
Cai H, He XH, Zheng DY, Qiu JA, Li ZC, Li FM (1996) Vinyl monomers bearing chromophore moieties and their polymers. 2. Fluorescence and initiation behavior of N-(4-N′, N′-dimethylaminophenyl)maleimide and its polymer. J Polym Sci A Polym Chem 34(7):1245–1250
Reddy PY, Kondo S, Fujita S, Toru T (1998) Efficient synthesis of fluorophore-linked maleimide derivatives. Synthesis 7:999–1002
Palmer M, Buchkremer M, Valeva A, Bhakdi S (1997) Cysteine-specific radioiodination of proteins with fluorescein maleimide. Anal Biochem 253(2):175–179
Majima E, Shinohara Y, Yamaguchi N, Hong YM, Terada H (1994) Importance of loops of mitochondrial ADP/ATP carrier for its transport activity deduced from reactivities of its cysteine residues with the sulfhydryl reagent eosin-5-maleimide. Biochemistry 33(32):9530–9536
Houstek J, Pedersen PL (1985) Adenine nucleotide and phosphate transport systems of mitochondria. Relative location of sulfhydryl groups based on the use of the novel fluorescent probe eosin-5-maleimide. J Biol Chem 260(10):6288–6295
Liu SQJ, Knauf PA (1993) Lys-430, site of irreversible inhibition of band-3 Cl(-) flux by eosin-5-maleimide, is not at the transport site. Am J Physiol 264(5):C1155–C1164
Majima E, Koike H, Hong YM, Shinohara Y, Terada H (1993) Characterization of cysteine residues of mitochondrial ADP/ATP carrier with the SH-reagents eosin 5-maleimide and N-ethylmaleimide. J Biol Chem 268(29):22181–22187
Blackman SM, Cobb CE, Beth AH, Piston DW (1996) The orientation of eosin-5-maleimide on human erythrocyte band 3 measured by fluorescence polarization microscopy. Biophys J 71(1):194–208
Kedar PS, Colah RB, Kulkarni S, Ghosh K, Mohanty D (2003) Experience with eosin-5′-maleimide as a diagnostic tool for red cell membrane cytoskeleton disorders. Clin Lab Haematol 25(6):373–376
King MJ, Smythe JS, Mushens R (2004) Eosin-5-maleimide binding to band 3 and Rh-related proteins forms the basis of a screening test for hereditary spherocytosis. Br J Haematol 124(1):106–113
Girodon F, Garcon L, Bergoin E, Largier M, Delaunay J, Feneant-Thibault M, Maynadie M, Couillaud G, Moreira S, Cynober T (2008) Usefulness of the eosin-5′-maleimide cytometric method as a first-line screening test for the diagnosis of hereditary spherocytosis: comparison with ektacytometry and protein electrophoresis. Br J Haematol 140(4):468–470
Kamata T, Akasaka K, Ohrui H, Meguro H (1993) A sensitive fluorometric assay of glutathione reductase activity with N-(9-acridinyl)maleimide. Anal Sci 9(6):867–870
Aykin N, Neal R, Yusof M, Ercal N (2001) Determination of captopril in biological samples by high performance liquid chromatography with ThioGlo(TM)3 derivatization. Biomed Chromatogr 15(7):427–432
Penugonda S, Wu W, Mare S, Ercal N (2004) Liquid chromatography analysis of N-(2-mercaptopropionyl)-glycine in biological samples by ThioGlo 3 derivatization. J Chromatogr B Anal Technol Biomed Life Sci 807(2):251–256
Mare S, Penugonda S, Ercal N (2005) High-performance liquid chromatography analysis of MESNA (2-mercaptoethane sulfonate) in biological samples using fluorescence detection. Biomed Chromatogr 19(1):80–86
Nakashima K, Umekawa C, Yoshida H, Nakatsuji S, Akiyama S (1987) High-performance liquid chromatography-fluorometry for the determination of thiols in biological samples using N-4-(6-dimethylamino-2-benzofuranyl) phenyl-maleimide. J Chromatogr 414(1):11–17
Lo KKW, Hui WK, Ng DCM, Cheung KK (2002) Synthesis, characterization, photophysical properties, and biological labeling studies of a series of luminescent rhenium(I) polypyridine maleimide complexes. Inorg Chem 41(1):40–46
DiGleria K, Hill HAO, Wong LL (1996) N-(2-ferrocene-ethyl)maleimide: a new electroactive sulphydryl-specific reagent for cysteine-containing peptides and proteins. FEBS Lett 390(2):142–144
Gandini A (2013) The furan/maleimide Diels-Alder reaction: a versatile click-unclick tool in macromolecular synthesis. Prog Polym Sci 38(1):1–29
Durmaz H, Colakoclu B, Tunca U, Hizal G (2006) Preparation of block copolymers via diels alder reaction of maleimide- and anthracene-end functionalized polymers. J Polym Sci A Polym Chem 44(5):1667–1675
Gacal B, Durmaz H, Tasdelen MA, Hizal G, Tunca U, Yagci Y, Demirel AL (2006) Anthracene-maleimide-based Diels-Alder “click chemistry” as a novel route to graft copolymers. Macromolecules 39(16):5330–5336
Yameen B, Rodriguez-Emmenegger C, Preuss CM, Pop-Georgievski O, Verveniotis E, Trouillet V, Rezek B, Barner-Kowollik C (2013) A facile avenue to conductive polymer brushes via cyclopentadiene-maleimide Diels-Alder ligation. Chem Commun 49(77):8623–8625
Liu YL, Yu JM, Chou CI (2004) Preparation and properties of novel benzoxazine and polybenzoxazine with maleimide groups. J Polym Sci A Polym Chem 42(23):5954–5963
Ishida H, Ohba S (2005) Synthesis and characterization of maleimide and norbornene functionalized benzoxazines. Polymer 46(15):5588–5595
Chaisuwan T, Ishida H (2006) High-performance maleimide and nitrile-functionalized benzoxazines with good processability for advanced composites applications. J Appl Polym Sci 101(1):548–558
Musa OM, Xhang R, Zhang R Curable liquid composition used as adhesive or encapsulant for affixing semiconductor devices onto substrate, comprises bisoxazoline resin, and resin having one or more reactive carbon to carbon double bonds e.g. 1,6-hexanediol diacrylate. WO2005085384-A1; EP1716215-A1; US2007032578-A1; US7390430-B2; TW200606231-A; EP1716215-B1; DE602004027549-E; TW384045-B1; SG116203-B
Musa OM, Sridhar LM, Yuan-Huffman QW Synthesizing product having triazole functionality, useful in film adhesive, comprises bulk polymerization of reactants having e.g. azide functionality using copper without a reducing agent in the absence of solvent. WO2008048733-A1; TW200829625-A; EP2078050-A1; KR2009088371-A; JP2010506940-W; CN101652405-A; US2010121022-A1
Musa OM (2004) Cycloaliphatic epoxy compounds containing styrenic, cinnamyl, or maleimide functionality. US6716992
Musa OM (2008) Maleimide resin with cyanurate core. US7456280
Phelan JC, Sung CSP (1997) Cure characterization in bis(maleimide)/diallylbisphenol A resin by fluorescence, FT-IR, and UV-reflection spectroscopy. Macromolecules 30(22):6845–6851
Marr B, Musa OM (2010) Diphenyl oxide compound for forming curable composition used as, e.g. coating, has diphenyl oxide backbone and pendant from backbone which is hydrocarbon chain containing ester functionality and terminated with maleimide functional group. US2010311207
Marr B, Musa OM, Zhuo Q (2009) Curable composition useful as adhesives, coatings, and encapsulants for various fabrication steps in the semiconductor packaging industry comprises oligomeric adduct of bismaleimide, diamine and dithiol; and a curing initiator. WO2009145779-A1
Olsen LC, Boyce BJ, Musa OM Vibrating screen assembly for separating e.g. rock in aggregate business, has screen deck and three cross braces diagonally disposed within frame, where one cross brace is positioned in center portion of deck to stiffen deck. US2004222135-A1; WO2004098797-A2; US6953121-B2; CN100344674-C; WO2004098797-A3
Musa OM, Marr B (2010) Alcohols containing imide moieties and reactive oligomers prepared therefrom. US20100190998-A1
Musa OM (2009) New oligomeric compound obtained by Michael addition reaction of a thiol with a maleimide compounds useful as a curable composition, adhesives, coatings and encapsulants, and for various fabrication steps in semiconductor packaging. US2010036136-A1
Molly H, Askym FS, Charles EH, Joe BW, Charles MW, Garfield TW (2005) Maleimide—Vinyl ether-based polymer dispersed liquid crystals, in stimuli-responsive polymeric films and coatings. American Chemical Society, Washington, DC, pp 195–213
Decker C, Bianchi C, Morel F, Jonsson S, Hoyle C (2000) Mechanistic study of the light-induced copolymerization of maleimide/vinyl ether systems. Macromol Chem Phys 201(13):1493–1503
Hoyle CE, Clark SC, Jönsson ES (2005) Polymerization processes using aliphatic maleimides. Google Patents
Jonsson S, Viswanathan K, Hoyle CE, Clark SC, Miller C, Nguyen C, Zhao W, Shao L, Morel F, Decker C (2000) Recent development in free radical photopolymerization. Direct and sensitized excitation of maleimides. J Photopolym Sci Technol 13(1):125–143
Fan SL, Boey FYC, Abadie MJM (2007) UV curing of a liquid based bismaleimide-containing polymer system. eXPRESS Polym Lett 1(6):397–405
Hoyle CE, Viswanathan K, Clark SC, Miller CW, Nguyen C, Jonsson S, Shao LY (1999) Sensitized polymerization of an acrylate/maleimide system. Macromolecules 32(8):2793–2795
Bongiovanni R, Sangermano M, Malucelli G, Priola A (2005) UV curing of photoinitiator-free systems containing bismaleimides and diacrylate resins: bulk and surface properties. Prog Org Coat 53(1):46–49
Herr D, Schultz RA, Xu P, McLaughlin SR (2001) Die attach adhesives for use in microelectronic devices. US6265530
Dershem SM, Patterson DB, Osuna JA (2006) Maleimide compounds in liquid form. US7102015
Horai A, Funaki K (2002) Maleimides for photoresist monomers, and their manufacture. JP2002069056A
Osuch CE, McFarland MJ (1990) Blocked monomer and polymers therefrom for use as photoresists. US 4962171
Zhang X, Chen G-C, Collins A, Jacobson S, Morganelli P, Dar YL, Musa OM (2009) Thermally degradable maleimides for reworkable adhesives. J Polym Sci A Polym Chem 47(4):1073–1084
Khosravi E, Musa OM (2011) Thermally degradable thermosetting materials. Eur Polym J 47(4):465–473
Khosravi E, Iqbal F, Musa OM (2011) Thermosetting ROMP materials with thermally degradable linkages. Polymer 52(2):243–249
Canary SA, Stevens MP (1992) Thermally reversible cross-linking of polystyrene via the furan-maleimide Diels-Alder reaction. J Polym Sci A Polym Chem 30(8):1755–1760
McElhanon JR, Wheeler DR (2001) Thermally responsive dendrons and dendrimers based on reversible furan-maleimide Diels-Alder adducts. Org Lett 3(17):2681–2683
Szalai ML, McGrath DV, Wheeler DR, Zifer T, McElhanon JR (2007) Dendrimers based on thermally reversible furan-maleimide Diels-Alder adducts. Macromolecules 40(4):818–823
Ondrus V, Fisera L, Polborn K, Ertl P, Pronayova N (1995) Diels-Alder reaction of fulvenes with N-(3,5-dichlorophenyl)-maleimide. Monatshefte Fur Chemie 126(8-9):961–969
Liu YL, Hsieh CY (2006) Crosslinked epoxy materials exhibiting thermal remendability and removability from multifunctional maleimide and furan compounds. J Polym Sci A Polym Chem 44(2):905–913
Stenzel MH (2013) Bioconjugation using thiols: old chemistry rediscovered to connect polymers with nature’s building blocks. ACS Macro Lett 2(1):14–18
Elliott JT, Prestwich GD (2000) Maleimide-functionalized lipids that anchor polypeptides to lipid bilayers and membranes. Bioconjug Chem 11(6):832–841
Le Sann C (2006) Maleimide spacers as versatile linkers in the synthesis of bioconjugates of anthracyclines. Nat Prod Rep 23(3):357–367
Warnecke A, Fichtner I, Garmann D, Jaehde U, Kratz F (2004) Synthesis and biological activity of water-soluble maleimide derivatives of the anticancer drug carboplatin designed as albumin-binding prodrugs. Bioconjug Chem 15(6):1349–1359
Warnecke A, Kratz F (2003) Maleimide-oligo(ethylene glycol) derivatives of camptothecin as albumin-binding prodrugs: synthesis and antitumor efficacy. Bioconjug Chem 14(2):377–387
Bykov VJN, Issaeva N, Zache N, Shilov A, Hultcrantz M, Bergman J, Selivanova G, Wiman KG (2005) Reactivation of mutant p53 and induction of apoptosis in human tumor cells by maleimide analogs. J Biol Chem 280(34):30384–30391
Ghosh SS, Kao PM, McCue AW, Chappelle HL (1990) Use of maleimide-thiol coupling chemistry for efficient syntheses of oligonucleotide-enzyme conjugate hybridization probes. Bioconjug Chem 1(1):71–76
Acknowledgments
The authors would like to express our sincere gratitude to Helen Gerardi, Tian Lu, Guanglou Cheng and Chi-san Wu of Ashland, Inc. for many helpful conversations/suggestions for the contents, and their thorough review of the manuscript and valuable feedback prior to publication.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2016 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Wu, F., Musa, O.M. (2016). Vegetable Oil–Maleic Anhydride and Maleimide Derivatives: Syntheses and Properties. In: Musa, O. (eds) Handbook of Maleic Anhydride Based Materials. Springer, Cham. https://doi.org/10.1007/978-3-319-29454-4_3
Download citation
DOI: https://doi.org/10.1007/978-3-319-29454-4_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-29453-7
Online ISBN: 978-3-319-29454-4
eBook Packages: Chemistry and Materials ScienceChemistry and Material Science (R0)