Abstract
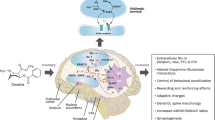
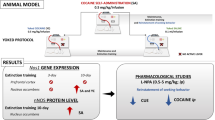
It has been described that development and persistent expression of addictive behaviors occur through the usurpation of natural learning and memory mechanisms within the limbic system. The hippocampus (HP) and medial prefrontal cortex (mPFC) have been implicated in the neuropathological mechanisms of drug addiction and withdrawal. In cocaine-abstinent human addicts and rats in similar conditions, neuronal activity of the mPFC is increased in response to cocaine reexposure or drug-associated cues and HP synaptic plasticity was increased after both cocaine and diazepam (DZ) repeated exposure in rats. Nitric oxide (NO) is a diffusible neuromodulator synthesized in the brain primarily by glutamatergic receptors activation, and may play a role in initiating and maintaining behavioral effects of psychostimulants and also in modulation of neuronal excitability and synaptic plasticity in different brain structures including the HP and mPFC. In this chapter we discuss some of the molecular and functional changes within the HP and mPFC induced by repeated cocaine or DZ administration and the pharmacological and methodological strategies used to prevent or reverse DZ dependence and cocaine vulnerability, such as those that interfere with NO signaling, HP plasticity or alter environmental cues related to withdrawal symptoms experience. The mechanisms and strategies presented can be explored as possible targets for development of new therapeutic agents to treat dependence of and addiction to psychoactive drugs.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Luddens H, Korpi ER, Seeburg PH. GABAA/benzodiazepine receptor heterogeneity: neurophysiological implications. Neuropharmacology. 1995;34(3):245–54.
Rabow LE, Russek SJ, Farb DH. From ion currents to genomic analysis: recent advances in GABAA receptor research. Synapse. 1995;21(3):189–274.
Sieghart W. Structure and pharmacology of gamma-aminobutyric acidA receptor subtypes. Pharmacol Rev. 1995;47(2):181–234.
Smith GB, Olsen RW. Functional domains of GABAA receptors. Trends Pharmacol Sci. 1995;16(5):162–8.
Chaudieu I, St-Pierre JA, Quirion R, Boksa P. GABAA receptor-mediated inhibition of N-methyl-D-aspartate-evoked [3H]dopamine release from mesencephalic cell cultures. Eur J Pharmacol. 1994;264(3):361–9.
Stelzer A, Slater NT, ten Bruggencate G. Activation of NMDA receptors blocks GABAergic inhibition in an in vitro model of epilepsy. Nature. 1987;326(6114):698–701.
Corda MG, Orlandi M, Lecca D, Giorgi O. Decrease in GABAergic function induced by pentylenetetrazol kindling in rats: antagonism by MK-801. J Pharmacol Exp Ther. 1992;262(2):792–800.
Giorgi O, Orlandi M, Geic M, Corda MG. MK-801 prevents the decrease in 35S-TBPS binding in the rat cerebral cortex induced by pentylenetetrazol kindling. Brain Res Bull. 1991;27(6):835–7.
Ohkuma S, Chen SH, Katsura M, Chen DZ, Kuriyama K. Muscimol prevents neuronal injury induced by NMDA. Jpn J Pharmacol. 1994;64(2):125–8.
Tsuda M, Suzuki T, Misawa M. Recovery of decreased seizure threshold for pentylenetetrazole during diazepam withdrawal by NMDA receptor antagonists. Eur J Pharmacol. 1997;324(1):63–6.
Tsuda M, Suzuki T, Misawa M. Role of the NMDA receptor complex in DMCM-induced seizure in mice. Neuroreport. 1997;8(3):603–6.
File SE. Tolerance to the behavioral actions of benzodiazepines. Neurosci Biobehav Rev. 1985;9(1):113–21.
Gonzalez JP, McCulloch AJ, Nicholls PJ, Sewell RD, Tekle A. Subacute benzodiazepine treatment: observations on behavioural tolerance and withdrawal. Alcohol Alcohol. 1984;19(4):325–32.
Hallstrom C, Lader M. Benzodiazepine withdrawal phenomena. Int Pharmacopsychiatry. 1981;16(4):235–44.
Rickels K, Case WG, Downing RW, Winokur A. Long-term diazepam therapy and clinical outcome. JAMA. 1983;250(6):767–71.
Petursson H. The benzodiazepine withdrawal syndrome. Addiction. 1994;89(11):1455–9.
Owen RT, Tyrer P. Benzodiazepine dependence. A review of the evidence. Drugs. 1983;25(4):385–98.
Petursson H, Lader MH. Benzodiazepine dependence. Br J Addict. 1981;76(2):133–45.
Cornish JW, O’Brien CP. Crack cocaine abuse: an epidemic with many public health consequences. Annu Rev Public Health. 1996;17:259–73.
Postma SW, Catterall WA. Inhibition of binding of [3H]batrachotoxinin A 20-alpha-benzoate to sodium channels by local anesthetics. Mol Pharmacol. 1984;25(2):219–27.
Reith ME, Kim SS, Lajtha A. Structural requirements for cocaine congeners to interact with [3H]batrachotoxinin A 20-alpha-benzoate binding sites on sodium channels in mouse brain synaptosomes. J Biol Chem. 1986;261(16):7300–5.
Wang GK, Wang SY. Altered stereoselectivity of cocaine and bupivacaine isomers in normal and batrachotoxin-modified Na+channels. J Gen Physiol. 1992;100(6):1003–20.
Li Y, Kauer JA. Repeated exposure to amphetamine disrupts dopaminergic modulation of excitatory synaptic plasticity and neurotransmission in nucleus accumbens. Synapse. 2004;51(1):1–10.
Thomas MJ, Malenka RC. Synaptic plasticity in the mesolimbic dopamine system. Philos Trans R Soc Lond B Biol Sci. 2003;358(1432):815–9.
Wolf ME, Mangiavacchi S, Sun X. Mechanisms by which dopamine receptors may influence synaptic plasticity. Ann N Y Acad Sci. 2003;1003:241–9.
Shen HW, Toda S, Moussawi K, Bouknight A, Zahm DS, Kalivas PW. Altered dendritic spine plasticity in cocaine-withdrawn rats. J Neurosci. 2009;29(9):2876–84.
Meyer JS, Quenzer LF. Psychopharmacology: drugs, the brain and behavior. Sunderland, MA: Sinauer Associates, Inc.; 2005.
Liddie S, Balda MA, Itzhak Y. Nitric Oxide (NO) Signaling as a potential therapeutic modality against psychostimulants. Curr Pharm Des. 2013;19(40):7092–102.
Ehrman RN, Robbins SJ, Childress AR, O'Brien CP. Conditioned responses to cocaine-related stimuli in cocaine abuse patients. Psychopharmacology (Berl). 1992;107(4):523–9.
Gawin FH, Kleber HD. Abstinence symptomatology and psychiatric diagnosis in cocaine abusers. Clinical observations. Arch Gen Psychiatry. 1986;43(2):107–13.
Weiss F, Martin-Fardon R, Ciccocioppo R, Kerr TM, Smith DL, Ben-Shahar O. Enduring resistance to extinction of cocaine-seeking behavior induced by drug-related cues. Neuropsychopharmacology. 2001;25(3):361–72.
Koob GF, Volkow ND. Neurocircuitry of addiction. Neuropsychopharmacology. 2010;35(1):217–38.
Parent A. Extrinsic connections of the basal ganglia. Trends Neurosci. 1990;13(7):254–8.
Koob GF, Ahmed SH, Boutrel B, Chen SA, Kenny PJ, Markou A, et al. Neurobiological mechanisms in the transition from drug use to drug dependence. Neurosci Biobehav Rev. 2004;27(8):739–49.
Pierce RC, Kumaresan V. The mesolimbic dopamine system: the final common pathway for the reinforcing effect of drugs of abuse? Neurosci Biobehav Rev. 2006;30(2):215–38.
Volkow ND, Wang GJ, Telang F, Fowler JS, Logan J, Childress AR, et al. Dopamine increases in striatum do not elicit craving in cocaine abusers unless they are coupled with cocaine cues. NeuroImage. 2008;39(3):1266–73.
Kelley AE. Ventral striatal control of appetitive motivation: role in ingestive behavior and reward-related learning. Neurosci Biobehav Rev. 2004;27(8):765–76.
Kalivas PW. Perspective: the manifest destiny of cocaine research. Neuropsychopharmacology. 2009;34(5):1089–90.
Kalivas PW, Stewart J. Dopamine transmission in the initiation and expression of drug- and stress-induced sensitization of motor activity. Brain Res Brain Res Rev. 1991;16(3):223–44.
Segal DS, Geyer MA, Schuckit MA. Stimulant-induced psychosis: an evaluation of animal methods. Essays Neurochem Neuropharmacol. 1981;5:95–129.
Robinson TE, Becker JB. Enduring changes in brain and behavior produced by chronic amphetamine administration: a review and evaluation of animal models of amphetamine psychosis. Brain Res. 1986;396(2):157–98.
Wolf ME. Addiction: making the connection between behavioral changes and neuronal plasticity in specific pathways. Mol Interv. 2002;2(3):146–57.
Itzhak Y, Roger-Sanchez C, Kelley JB, Anderson KL. Discrimination between cocaine-associated context and cue in a modified conditioned place preference paradigm: role of the nNOS gene in cue conditioning. Int J Neuropsychopharmacol. 2010;13(2):171–80.
Everitt BJ, Wolf ME. Psychomotor stimulant addiction: a neural systems perspective. J Neurosci. 2002;22(9):3312–20.
Steketee JD. Cortical mechanisms of cocaine sensitization. Crit Rev Neurobiol. 2005;17(2):69–86.
Belujon P, Grace AA. Hippocampus, amygdala, and stress: interacting systems that affect susceptibility to addiction. Ann N Y Acad Sci. 2011;1216:114–21.
Boudreau AC, Wolf ME. Behavioral sensitization to cocaine is associated with increased AMPA receptor surface expression in the nucleus accumbens. J Neurosci. 2005;25(40):9144–51.
Churchill L, Swanson CJ, Urbina M, Kalivas PW. Repeated cocaine alters glutamate receptor subunit levels in the nucleus accumbens and ventral tegmental area of rats that develop behavioral sensitization. J Neurochem. 1999;72(6):2397–403.
Pierce RC, Bell K, Duffy P, Kalivas PW. Repeated cocaine augments excitatory amino acid transmission in the nucleus accumbens only in rats having developed behavioral sensitization. J Neurosci. 1996;16(4):1550–60.
Perez MF, Gabach LA, Almiron RS, Carlini VP, De Barioglio SR, Ramirez OA. Different chronic cocaine administration protocols induce changes on dentate gyrus plasticity and hippocampal dependent behavior. Synapse. 2010;64(10):742–53.
White FJ, Hu XT, Zhang XF, Wolf ME. Repeated administration of cocaine or amphetamine alters neuronal responses to glutamate in the mesoaccumbens dopamine system. J Pharmacol Exp Ther. 1995;273(1):445–54.
Vorel SR, Liu X, Hayes RJ, Spector JA, Gardner EL. Relapse to cocaine-seeking after hippocampal theta burst stimulation. Science. 2001;292(5519):1175–8.
Jentsch JD, Taylor JR. Impulsivity resulting from frontostriatal dysfunction in drug abuse: implications for the control of behavior by reward-related stimuli. Psychopharmacology (Berl). 1999;146(4):373–90.
Marin RH, Perez MF, Duero DG, Ramirez OA. Preexposure to drug administration context blocks the development of tolerance to sedative effects of diazepam. Pharmacol Biochem Behav. 1999;64(3):473–7.
Perez MF, Nasif FJ, Marchesini GR, Maglio LE, Ramirez OA. Hippocampus and locus coeruleus activity on rats chronically treated with diazepam. Pharmacol Biochem Behav. 2001;69(3–4):431–8.
Tan KR, Brown M, Labouebe G, Yvon C, Creton C, Fritschy JM, et al. Neural bases for addictive properties of benzodiazepines. Nature. 2010;463(7282):769–74.
Perez MF, Maglio LE, Marchesini GR, Molina JC, Ramirez OA. Environmental changes modify the expression of diazepam withdrawal. Behav Brain Res. 2002;136(1):75–81.
Monti MC, Almiron RS, Bignante EA, Ramirez OA. Changes in hippocampal arc protein expression and synaptic plasticity by the presentation of contextual cues linked to drug experience. Synapse. 2010;64(1):39–46.
Siegel S. Evidence from rats that morphine tolerance is a learned response. J Comp Physiol Psychol. 1975;89(5):498–506.
Siegel S. Morphine tolerance acquisition as an associative process. J Exp Psychol Anim Behav Process. 1977;3(1):1–13.
Wikler A. Dynamics of drug dependence. Implications of a conditioning theory for research and treatment. Arch Gen Psychiatry. 1973;28(5):611–6.
Ludwig AM, Stark LH. Alcohol craving. Subjective and situational aspects. Q J Stud Alcohol. 1974;35(3):899–905.
O'Brien CP, Testa T, O'Brien TJ, Brady JP, Wells B. Conditioned narcotic withdrawal in humans. Science. 1977;195(4282):1000–2.
Hyman SE, Malenka RC. Addiction and the brain: the neurobiology of compulsion and its persistence. Nat Rev Neurosci. 2001;2(10):695–703.
Taubenfeld SM, Muravieva EV, Garcia-Osta A, Alberini CM. Disrupting the memory of places induced by drugs of abuse weakens motivational withdrawal in a context-dependent manner. Proc Natl Acad Sci U S A. 2010;107(27):12345–50.
Ciccocioppo R, Sanna PP, Weiss F. Cocaine-predictive stimulus induces drug-seeking behavior and neural activation in limbic brain regions after multiple months of abstinence: reversal by D(1) antagonists. Proc Natl Acad Sci U S A. 2001;98(4):1976–81.
Nestler EJ. Psychogenomics: opportunities for understanding addiction. J Neurosci. 2001;21(21):8324–7.
Eisch AJ, Mandyam CD. Drug dependence and addiction, II: adult neurogenesis and drug abuse. Am J Psychiatry. 2004;161(3):426.
del Olmo N, Miguens M, Higuera-Matas A, Torres I, Garcia-Lecumberri C, Solis JM, et al. Enhancement of hippocampal long-term potentiation induced by cocaine self-administration is maintained during the extinction of this behavior. Brain Res. 2006;1116(1):120–6.
Kim JJ, Fanselow MS. Modality-specific retrograde amnesia of fear. Science. 1992;256(5057):675–7.
Sinha R. How does stress increase risk of drug abuse and relapse? Psychopharmacology (Berl). 2001;158(4):343–59.
Phillips RG, LeDoux JE. Differential contribution of amygdala and hippocampus to cued and contextual fear conditioning. Behav Neurosci. 1992;106(2):274–85.
Martin SJ, Grimwood PD, Morris RG. Synaptic plasticity and memory: an evaluation of the hypothesis. Annu Rev Neurosci. 2000;23:649–711.
Gabach LA, Carlini VP, Monti MC, Maglio LE, De Barioglio SR, Perez MF. Involvement of nNOS/NO/sGC/cGMP signaling pathway in cocaine sensitization and in the associated hippocampal alterations: does phosphodiesterase 5 inhibition help to drug vulnerability? Psychopharmacology (Berl). 2013;229(1):41–50.
Thompson AM, Swant J, Gosnell BA, Wagner JJ. Modulation of long-term potentiation in the rat hippocampus following cocaine self-administration. Neuroscience. 2004;127(1):177–85.
Fuster JM. Frontal lobe and cognitive development. J Neurocytol. 2002;31(3–5):373–85.
Miller EK. The prefrontal cortex and cognitive control. Nat Rev Neurosci. 2000;1(1):59–65.
Quirk GJ, Beer JS. Prefrontal involvement in the regulation of emotion: convergence of rat and human studies. Curr Opin Neurobiol. 2006;16(6):723–7.
Sotres-Bayon F, Cain CK, LeDoux JE. Brain mechanisms of fear extinction: historical perspectives on the contribution of prefrontal cortex. Biol Psychiatry. 2006;60(4):329–36.
Tzschentke TM. Pharmacology and behavioral pharmacology of the mesocortical dopamine system. Prog Neurobiol. 2001;63(3):241–320.
Volkow ND, Ding YS, Fowler JS, Wang GJ. Cocaine addiction: hypothesis derived from imaging studies with PET. J Addict Dis. 1996;15(4):55–71.
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O'Brien CP. Limbic activation during cue-induced cocaine craving. Am J Psychiatry. 1999;156(1):11–8.
Li Y, Hu XT, Berney TG, Vartanian AJ, Stine CD, Wolf ME, et al. Both glutamate receptor antagonists and prefrontal cortex lesions prevent induction of cocaine sensitization and associated neuroadaptations. Synapse. 1999;34(3):169–80.
Pierce RC, Reeder DC, Hicks J, Morgan ZR, Kalivas PW. Ibotenic acid lesions of the dorsal prefrontal cortex disrupt the expression of behavioral sensitization to cocaine. Neuroscience. 1998;82(4):1103–14.
Wolf ME. The role of excitatory amino acids in behavioral sensitization to psychomotor stimulants. Prog Neurobiol. 1998;54(6):679–720.
Peterson JD, Wolf ME, White FJ. Altered responsiveness of medial prefrontal cortex neurons to glutamate and dopamine after withdrawal from repeated amphetamine treatment. Synapse. 2000;36(4):342–4.
Dong Y, Nasif FJ, Tsui JJ, Ju WY, Cooper DC, Hu XT, et al. Cocaine-induced plasticity of intrinsic membrane properties in prefrontal cortex pyramidal neurons: adaptations in potassium currents. J Neurosci. 2005;25(4):936–40.
Nasif FJ, Sidiropoulou K, Hu XT, White FJ. Repeated cocaine administration increases membrane excitability of pyramidal neurons in the rat medial prefrontal cortex. J Pharmacol Exp Ther. 2005;312(3):1305–13.
Mustafa AK, Gadalla MM, Snyder SH. Signaling by gasotransmitters. Sci Signal. 2009;2(68):re2.
Garthwaite J. New insight into the functioning of nitric oxide-receptive guanylyl cyclase: physiological and pharmacological implications. Mol Cell Biochem. 2010;334(1–2):221–32.
Garthwaite J. Concepts of neural nitric oxide-mediated transmission. Eur J Neurosci. 2008;27(11):2783–802.
Prast H, Philippu A. Nitric oxide as modulator of neuronal function. Prog Neurobiol. 2001;64(1):51–68.
Itzhak Y. Attenuation of cocaine kindling by 7-nitroindazole, an inhibitor of brain nitric oxide synthase. Neuropharmacology. 1996;35(8):1065–73.
Kim HS, Park WK. Nitric oxide mediation of cocaine-induced dopaminergic behaviors: ambulation-accelerating activity, reverse tolerance and conditioned place preference in mice. J Pharmacol Exp Ther. 1995;275(2):551–7.
Orsini C, Izzo E, Koob GF, Pulvirenti L. Blockade of nitric oxide synthesis reduces responding for cocaine self-administration during extinction and reinstatement. Brain Res. 2002;925(2):133–40.
Nasif FJ, Hu XT, Ramirez OA, Perez MF. Inhibition of neuronal nitric oxide synthase prevents alterations in medial prefrontal cortex excitability induced by repeated cocaine administration. Psychopharmacology (Berl). 2011;218(2):323–30.
Talarek S, Fidecka S. Role of nitric oxide in benzodiazepines-induced antinociception in mice. Pol J Pharmacol. 2002;54(1):27–34.
Talarek S, Fidecka S. Role of nitric oxide in anticonvulsant effects of benzodiazepines in mice. Pol J Pharmacol. 2003;55(2):181–91.
Talarek S, Fidecka S. Involvement of nitricoxidergic system in the hypnotic effects of benzodiazepines in mice. Pol J Pharmacol. 2004;56(6):719–26.
Talarek S, Listos J, Fidecka S. Role of nitric oxide in the development of tolerance to diazepam-induced motor impairment in mice. Pharmacol Rep. 2008;60(4):475–82.
Talarek S, Listos J, Fidecka S. Effect of nitric oxide synthase inhibitors on benzodiazepine withdrawal in mice and rats. Pharmacol Rep. 2011;63(3):680–9.
Groenewegen HJ, Wright CI, Beijer AV, Voorn P. Convergence and segregation of ventral striatal inputs and outputs. Ann N Y Acad Sci. 1999;877:49–63.
Di Ciano P, Cardinal RN, Cowell RA, Little SJ, Everitt BJ. Differential involvement of NMDA, AMPA/kainate, and dopamine receptors in the nucleus accumbens core in the acquisition and performance of pavlovian approach behavior. J Neurosci. 2001;21(23):9471–7.
French SJ, Totterdell S. Hippocampal and prefrontal cortical inputs monosynaptically converge with individual projection neurons of the nucleus accumbens. J Comp Neurol. 2002;446(2):151–65.
Thompson AM, Gosnell BA, Wagner JJ. Enhancement of long-term potentiation in the rat hippocampus following cocaine exposure. Neuropharmacology. 2002;42(8):1039–42.
Sammut S, Threlfell S, West AR. Nitric oxide-soluble guanylyl cyclase signaling regulates corticostriatal transmission and short-term synaptic plasticity of striatal projection neurons recorded in vivo. Neuropharmacology. 2010;58(3):624–31.
Boccia MM, Blake MG, Krawczyk MC, Baratti CM. Sildenafil, a selective phosphodiesterase type 5 inhibitor, enhances memory reconsolidation of an inhibitory avoidance task in mice. Behav Brain Res. 2011;220(2):319–24.
Puzzo D, Staniszewski A, Deng SX, Privitera L, Leznik E, Liu S, et al. Phosphodiesterase 5 inhibition improves synaptic function, memory, and amyloid-beta load in an Alzheimer's disease mouse model. J Neurosci. 2009;29(25):8075–86.
Sammut S, West AR. Acute cocaine administration increases NO efflux in the rat prefrontal cortex via a neuronal NOS-dependent mechanism. Synapse. 2008;62(9):710–3.
Bagetta G, Rodino P, Arabia A, Massoud R, Paoletti AM, Nistico R, et al. Systemic administration of cocaine, given alone or in combination with sensory stimuli, differentially affects L-arginine-nitric oxide metabolism in discrete regions of the brain of rat. Neurosci Lett. 1999;266(3):153–6.
Bhargava HN, Kumar S. Sensitization to the locomotor stimulant activity of cocaine is associated with increases in nitric oxide synthase activity in brain regions and spinal cord of mice. Pharmacology. 1997;55(6):292–8.
Monti MC, Gabach LA, Perez MF, Ramirez OA. Impact of contextual cues in the expression of the memory associated with diazepam withdrawal: involvement of hippocampal PKMzeta in vivo, and Arc expression and LTP in vitro. Eur J Neurosci. 2012;36(8):3118–25.
Frey U, Frey S, Schollmeier F, Krug M. Influence of actinomycin D, a RNA synthesis inhibitor, on long-term potentiation in rat hippocampal neurons in vivo and in vitro. J Physiol. 1996;490(Pt 3):703–11.
Nguyen PV, Kandel ER. A macromolecular synthesis-dependent late phase of long-term potentiation requiring cAMP in the medial perforant pathway of rat hippocampal slices. J Neurosci. 1996;16(10):3189–98.
Guzowski JF, McNaughton BL, Barnes CA, Worley PF. Environment-specific expression of the immediate-early gene Arc in hippocampal neuronal ensembles. Nat Neurosci. 1999;2(12):1120–4.
Bramham CR, Alme MN, Bittins M, Kuipers SD, Nair RR, Pai B, et al. The Arc of synaptic memory. Exp Brain Res. 2010;200(2):125–40.
Messaoudi E, Kanhema T, Soule J, Tiron A, Dagyte G, da Silva B, et al. Sustained Arc/Arg3.1 synthesis controls long-term potentiation consolidation through regulation of local actin polymerization in the dentate gyrus in vivo. J Neurosci. 2007;27(39):10445–55.
Pastalkova E, Serrano P, Pinkhasova D, Wallace E, Fenton AA, Sacktor TC. Storage of spatial information by the maintenance mechanism of LTP. Science. 2006;313(5790):1141–4.
Sacktor TC. How does PKMzeta maintain long-term memory? Nat Rev Neurosci. 2011;12(1):9–15.
Smith KM, Romanelli F. Recreational use and misuse of phosphodiesterase 5 inhibitors. J Am Pharm Assoc. 2005;45(1):63–72. quiz 3–5.
McCambridge J, Mitcheson L, Hunt N, Winstock A. The rise of Viagra among British illicit drug users: 5-year survey data. Drug Alcohol Rev. 2006;25(2):111–3.
Disclosures/Conflicts
None.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2015 Springer International Publishing Switzerland
About this chapter
Cite this chapter
de la Villarmois, E.A., Gabach, L.A., Pérez, M.F. (2015). New Insights in Glutamate-Mediated Mechanisms Underlying Benzodiazepines Dependence and Cocaine Vulnerability. In: Gargiulo, P., Arroyo, H. (eds) Psychiatry and Neuroscience Update. Springer, Cham. https://doi.org/10.1007/978-3-319-17103-6_13
Download citation
DOI: https://doi.org/10.1007/978-3-319-17103-6_13
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-17102-9
Online ISBN: 978-3-319-17103-6
eBook Packages: MedicineMedicine (R0)