Abstract
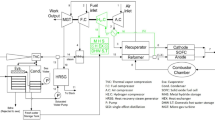
Hydrogen production via thermochemical water splitting using the Cu–Cl cycle is a promising alternative to conventional hydrogen production methods. Multi-generation systems are often attractive due to their higher energy and exergy efficiencies than individual cycles (e.g. steam or gas turbine cycles). An increase in efficiency often allows, for the same output, less resources (or exergy) to be used. This, in return, reduces the extraction from the environment of energy resources (e.g. fossil fuels), and decreases the associated environmental impacts. In this study, comprehensive thermodynamic analyses of two Cu–Cl based integrated systems for hydrogen production are reported. The systems considered here are developed to produce hydrogen and provide cooling. Hot water, drying air and oxygen are also by-products of the system. The first system also has the capability of generating electricity using a two-stage steam turbine cycle. The main sub-units of the system are the Cu–Cl thermochemical water splitting cycle and a LiBr–H2O absorption cooling system. Solar energy drives the first system using a heliostat solar tower, whereas waste/process heat from a Generation IV nuclear supercritical water cooled reactor (SCWR) is the energy source of the second system. The energy efficiency of the first system is found to be approximately 70 %, while the exergy efficiency is approximately 58 %. The second system, on the other hand, has an energy efficiency of 63 % and an exergy efficiency of 41 %.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others

References
Urbaniec K, Friedl A, Huisingh D, Claassen P (2010) Hydrogen for a sustainable global economy. J Cleaner Prod 18:S1–S3
Dincer I (2012) Green methods for hydrogen production. Int J Hydrogen Energy 37:1954–1971
Rosen MA (2010) Advances in hydrogen production by thermochemical water decomposition: a review. Energy 35:1068–1076
Dufour J, Serrano DP, Galvez JL, Moreno J, Garcia C (2009) Life cycle assessment of processes for hydrogen production: environmental feasibility and reduction of greenhouse gases emissions. Int J Hydrogen Energy 34:1370–1376
Lewis MA, Masin JG, O’Hare PA (2009) Evaluation of alternative thermochemical cycles. Part I: the methodology. Int J Hydrogen Energy 34:4115–4124
Pilavachi PA, Chatzinapagi AI, Spyropoulou AI (2009) Evaluation of hydrogen production methods using the analytic hierarchy process. Int J Hydrogen Energy 34:5294–5303
Naterer GF, Suppiah S, Stolberg L, Lewis M, Wang Z, Dincer I, Rosen MA, Gabriel K, Secnik E, Easton EB, Pioro I, Lvov S, Jiang J, Mostaghimi J, Ikeda BM, Rizvi G, Lu L, Oduyaka A, Spekkens P, Fowler M, Avsec J (2013) Progress of international hydrogen production network for the thermochemical Cu–Cl cycle. Int J Hydrogen Energy 38:740–759
Naterer GF, Fowler M, Cotton J, Gabriel K (2008) Synergistic roles of off-peak electrolysis and thermochemical production of hydrogen from nuclear energy in Canada. Int J Hydrogen Energy 33:6849–6857
Gebreslassie BH, Medrano M, Boer D (2010) Exergy analysis of multi-effect water–LiBr absorption systems: from half to triple effect. Renew Energy 35:1773–1782
Nezammahalleh H, Farhadi F, Tanhaemami M (2010) Conceptual design and techno-economic assessment of integrated solar combined cycle system with DSG technology. Solar Energy 84:1696–1705
Wang ZL, Naterer GF, Gabriel KS, Secnik E, Gravelsins R, Daggupati VN (2011) Thermal design of a solar hydrogen plant with a copper–chlorine cycle and molten salt energy storage. Int J Hydrogen Energy 36:11258–11272
Pioro IL, Duffey RB (2007) Heat transfer and hydraulic resistance at supercritical pressures in power engineering applications. ASME, New York
Dincer I, Rosen MA (2013) Exergy: energy, environment and sustainable development, 2nd edn. Elsevier, Oxford
Rosen MA, Dincer I (2001) Exergy as the confluence of energy, environment and sustainable development. Exergy Int J 1:3–13
Kotas TJ (1995) The exergy method of thermal plant analysis. Krieger, Malabar
Moran MJ (1989) Availability analysis: a guide to efficient energy use. ASME, New York
Acknowledgement
The authors acknowledge gratefully the financial support provided by the Ontario Research Excellence Fund.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Nomenclature
Nomenclature
- ex :
-
Specific exergy, MJ/kg
- Ex d :
-
Exergy destruction, MJ
- ex ch :
-
Specific chemical exergy, MJ/kg
- Ex Q :
-
Exergy of thermal energy, MJ
- h :
-
Specific enthalpy, kJ/kg
- LHV :
-
Lower heating value, MJ
- \( \dot{m} \) :
-
Mass flow rate, kg/s
- η en :
-
Energy efficiency
- η ex :
-
Exergy efficiency
- Q :
-
Heat, MJ
- S gen :
-
Entropy generation, MJ/K
- T i :
-
System temperature, K
- T 0 :
-
Reference-environment temperature, K
- W :
-
Work, MJ
- \( {\dot{W}}_{net} \) :
-
Net power output, MW
- ACS:
-
Absorption cooling system
- EES:
-
Engineering equation solver
- HP:
-
High pressure
- HTF:
-
Heat transfer fluid
- LP:
-
Low pressure
- SCWR:
-
Supercritical water cooled reactor
- STC:
-
Steam turbine cycle
Rights and permissions
Copyright information
© 2014 Springer International Publishing Switzerland
About this chapter
Cite this chapter
Ozbilen, A., Dincer, I., Rosen, M.A. (2014). Energy and Exergy Analyses of Copper–Chlorine (Cu–Cl) Based Integrated Systems for Hydrogen Production. In: Dincer, I., Midilli, A., Kucuk, H. (eds) Progress in Exergy, Energy, and the Environment. Springer, Cham. https://doi.org/10.1007/978-3-319-04681-5_10
Download citation
DOI: https://doi.org/10.1007/978-3-319-04681-5_10
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-319-04680-8
Online ISBN: 978-3-319-04681-5
eBook Packages: EnergyEnergy (R0)