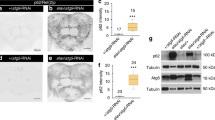
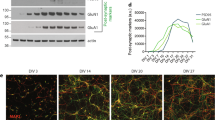
Abstract
Autophagy is crucial for neuronal integrity. Loss of key autophagic components leads to progressive neurodegeneration and structural defects in neuronal synapses. However, the molecular mechanisms regulating autophagy in the brain remain elusive. Similarly, while it is widely accepted that protein turnover is required for synaptic plasticity, the contribution of autophagy to the degradation of synaptic proteins is unknown. We find that BDNF signaling via the tropomyosin receptor kinase B (TrkB) and the phosphatidylinositol 3-kinase (PI3K)/Akt pathway suppresses autophagy in vivo. Autophagy is differentially regulated by fasting, in different brain regions. Suppression of autophagy is required for BDNF-induced synaptic plasticity and for memory enhancement, under conditions of nutritional stress. BDNF signaling suppresses autophagy in the forebrain of adult mice. Indeed, BDNF ablation in the neural lineage causes uncontrolled increase in autophagy. In turn, increased autophagy mediates the synaptic defects caused by BDNF deficiency. Thus, fasting suppresses autophagy in regions of the mouse forebrain, thereby promoting synaptic remodeling and memory through a BDNF-regulated mechanism. We identify three key remodelers of postsynaptic densities as cargo of autophagy. Our results establish autophagy as a pivotal component of BDNF signaling, which is essential for BDNF-induced synaptic plasticity. This molecular mechanism underlies behavioral adaptations that increase fitness in times of scarcity.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
Autophagy is crucial for neuronal integrity. Loss of key autophagic components leads to progressive neurodegeneration and structural defects in neuronal synapses. However, the molecular mechanisms regulating autophagy in the brain remain elusive. Similarly, while it is widely accepted that protein turnover is required for synaptic plasticity, the contribution of autophagy to the degradation of synaptic proteins is unknown. We find that BDNF signaling via the tropomyosin receptor kinase B (TrkB) and the phosphatidylinositol 3-kinase (PI3K)/Akt pathway suppresses autophagy in vivo. Autophagy is differentially regulated by fasting, in different brain regions. Suppression of autophagy is required for BDNF-induced synaptic plasticity and for memory enhancement, under conditions of nutritional stress. BDNF signaling suppresses autophagy in the forebrain of adult mice. Indeed, BDNF ablation in the neural lineage causes uncontrolled increase in autophagy. In turn, increased autophagy mediates the synaptic defects caused by BDNF deficiency. Thus, fasting suppresses autophagy in regions of the mouse forebrain, thereby promoting synaptic remodeling and memory through a BDNF-regulated mechanism. We identify three key remodelers of postsynaptic densities as cargo of autophagy. Our results establish autophagy as a pivotal component of BDNF signaling, which is essential for BDNF-induced synaptic plasticity. This molecular mechanism underlies behavioral adaptations that increase fitness in times of scarcity.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Tavernarakis, N. (2020). Regulation and Roles of Autophagy in the Brain. In: Vlamos, P. (eds) GeNeDis 2018. Advances in Experimental Medicine and Biology, vol 1195. Springer, Cham. https://doi.org/10.1007/978-3-030-32633-3_5
Download citation
DOI: https://doi.org/10.1007/978-3-030-32633-3_5
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-32632-6
Online ISBN: 978-3-030-32633-3
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)