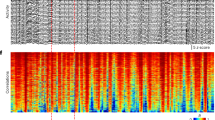
Abstract
Self-organizing systems acquire their structures and functions without patterned input from the outside world. In the interconnected architectures of the neocortex, spontaneous activity—that is, activity that arises without external sensory or electrical stimulus—predominates over sensory-evoked activity. Thus, spontaneous neuronal activity provides a means to characterizing the structure, function and dynamics of neocortical networks. We have recorded spontaneous, asynchronous network activity from hundreds of neurons constituting local cortical circuits in mice with high-density microelectrode arrays (MEAs) in vitro. The spontaneous activity in the network displayed features of a system at criticality and scale-free structures, such as fluctuation scaling and multiple frequency bands. To investigate dynamical parameters, we have investigated the linear and nonlinear components of the network dynamics. The former allows us not only to define a linear measure of functional connectivity, but also to determine the linear stability of the system through its eigenvalues. Similarly, the latter allows us to define a measure of nonlinear functional connectivity. An important feature revealed by this approach is the large number of eigenvalues with positive real parts and the high density of eigenvalues near the imaginary axis, which demonstrate respectively that this high-dimensional system is linearly unstable and critical on long time scales (>1s). The function of critical dynamics in these networks is discussed with respect to exploratory behavior in rodents.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Ahmed, B., Anderson, J.C., Douglas, R.J., et al.: Polyneuronal innervation of spiny stellate neurons in cat visual cortex. J. Comput. Neurol. 341, 39–49 (1994). https://doi.org/10.1002/cne.903410105
Ahrens, K.F., Kleinfeld, D.: Current flow in vibrissa motor cortex can phase-lock with exploratory rhythmic whisking in rat. J. Neurophysiol. 92, 1700–1707 (2004). https://doi.org/10.1152/jn.00404.2004
Alstrom, P.: Mean-field exponents for self-organized critical phenomena. Phys. Rev. A 38, 4905–4906 (1988). https://doi.org/10.1103/PhysRevA.38.4905
Bak, P., Stassinopoulos, D.: Democratic reinforcement 51 (1995)
Bak, P., Tang, C., Wiesenfeld, K.: Self-organized criticality: An explanation of the 1/ f noise. Phys. Rev. Lett. 59, 381–384 (1987). https://doi.org/10.1103/physrevlett.59.381
Bédard, C., Kröger, H., Destexhe, A.: Does the 1/f frequency scaling of brain signals reflect self-organized critical states? Phys. Rev. Lett. 97, 1–4 (2006). https://doi.org/10.1103/physrevlett.97.118102
Beggs, J.M., Plenz, D.: Neuronal avalanches in neocortical circuits. J. Neurosci. 23, 11167–11177 (2003). doi:23/35/11167 [pii]
Berg, R.W., Kleinfeld, D.: Rhythmic whisking by rat: retraction as well as protraction of the vibrissae is under active muscular control. J. Neurophysiol. 89, 104–117 (2003). https://doi.org/10.1152/jn.00600.2002
Buice, M.A., Cowan, J.D.: Statistical mechanics of the neocortex. Prog. Biophys. Mol. Biol. 99, 53–86 (2009). https://doi.org/10.1016/j.pbiomolbio.2009.07.003
Cabral, J., Kringelbach, M., Deco, G.: Functional connectivity dynamically evolves on multiple time-scales over a static structural connectome: models and mechanisms. Neuroimage, 0–1 (2017). https://doi.org/10.1016/j.neuroimage.2017.03.045
Chialvo, D., Bak, P.: Commentary: learning from mistakes. Neuroscience 90, 1137–1148 (1999)
Chiel, H.J., Beer, R.D.: The brain has a body: adaptive behavior emerges from interactions of nervous system, body and environment. Trends Neurosci. 20, 553–557 (1997)
Cowan, J.D., Neuman, J., Kiewiet, B., Van Drongelen, W.: Self-organized criticality in a network of interacting neurons. J. Stat. Mech. Theory Exp. 2013:. https://doi.org/10.1088/1742-5468/2013/04/p04030
Cowan, J.D., Neuman, J., Van Drongelen, W.: Self-organized criticality and near-criticality in neural networks. In: Criticality in Neural Systems, pp 465–484 (2014)
De Arcangelis, L., Perrone-Capano, C., Herrmann, H.J.: Self-organized criticality model for brain plasticity. Phys. Rev. Lett. 96, 1–4 (2006). https://doi.org/10.1103/physrevlett.96.028107
Douglas, R.J., Koch, C., Mahowald, M., et al.: Recurrent excitation in neocortical circuits. Science 269, 981–985 (1995)
Eguíluz, V.M., Chialvo, D.R., Cecchi, G.A., et al.: Scale-free brain functional networks. Phys. Rev. Lett. 94, 1–4 (2005). https://doi.org/10.1103/physrevlett.94.018102
Friedman, N., Ito, S., Brinkman, B.A.W., et al.: Universal critical dynamics in high resolution neuronal avalanche data. Phys. Rev. Lett. 108, 1–5 (2012). https://doi.org/10.1103/physrevlett.108.208102
Galán, R.F.: On how network architecture determines the dominant patterns of spontaneous neural activity. PLoS One 3, (2008). https://doi.org/10.1371/journal.pone.0002148
Gireesh, E.D., Plenz, D.: Neuronal avalanches organize as nested theta- and beta/gamma-oscillations during development of cortical layer 2/3. Proc. Natl. Acad. Sci. 105, 7576–7581 (2008). https://doi.org/10.1073/pnas.0800537105
Gutenberg B, Richter C (1954) Seismicity of the earth: Princeton, NJ
Hájos, N., Ellender, T.J., Zemankovics, R., et al.: Maintaining network activity in submerged hippocampal slices: importance of oxygen supply. Eur. J. Neurosci. 29, 319–327 (2009). https://doi.org/10.1111/j.1460-9568.2008.06577.x
Hájos, N., Mody, I.: Establishing a physiological environment for visualized in vitro brain slice recordings by increasing oxygen supply and modifying aCSF content. J. Neurosci. Methods 183, 107–113 (2009). https://doi.org/10.1016/j.jneumeth.2009.06.005
Haken, H.: Synergetics: An Introduction. Springer, Berlin (1978)
Haken, H.: Advanced Synergetics: Instability Hierarchies of Self-organizing Systems. Springer, Berlin (1983)
Herz, A.V.M., Hopfield, J.J.: Earthquake cycles and neural reverberations: collective oscillations in systems with pulse-coupled threshold elements andreas. Phys. Rev. Lett. 75, 4–7 (1995)
Hoffman, K.L., Battaglia, F.P., Harris, K., et al.: The upshot of up states in the neocortex: from slow oscillations to memory formation. J. Neurosci. 27, 11838–11841 (2007). https://doi.org/10.1523/JNEUROSCI.3501-07.2007
Kodama, N.X., Feng, T., Ullett, J.J., et al.: Anti-correlated cortical networks arise from spontaneous neuronal dynamics at slow timescales. Sci. Rep. (2017)
Koyama, S.: On the spike train variability characterized by variance-to-mean power relationship. Neural Comput. 27, 1530–1548 (2015). https://doi.org/10.1162/NECO_a_00748
Landau, I.D., Sompolinsky, H.: Coherent chaos in a recurrent neural network with structured connectivity, 1–29 (2018). https://doi.org/10.1101/350801
Langton, C.G.: Computation at the edge of chaos: phase transitions and emergent computation. Phys. D Nonlinear Phenom. 42, 12–37 (1990). https://doi.org/10.1016/0167-2789(90)90064-V
Levina, A., Priesemann, V.: Subsampling scaling. Nat. Commun. 8, 1–9 (2017). https://doi.org/10.1038/ncomms15140
Luczak, A., Maclean, J.N.: Default activity patterns at the neocortical microcircuit level. Front. Integr. Neurosci. 6, 30 (2012). https://doi.org/10.3389/fnint.2012.00030
MacLean, J.N., Watson, B.O., Aaron, G.B., Yuste, R.: Internal dynamics determine the cortical response to thalamic stimulation. Neuron 48, 811–823 (2005). https://doi.org/10.1016/j.neuron.2005.09.035
Malamud, B.D.: Forest fires: an example of self-organized critical behavior. Science 1840, 1998–2001 (2008). https://doi.org/10.1126/science.281.5384.1840
Mehta, M.L.: Random Matrices (2005)
Millman, D., Mihalas, S., Kirkwood, A., Niebur, E.: Self-organized criticality occurs in non-conservative neuronal networks during “up” states. Nat. Phys. 6, 801–805 (2010). https://doi.org/10.1038/nphys1757
Mitra, P., Pesaran, B.: Analysis of dynamic brain imaging data. Biophys. J. 76, 691–708 (1999)
Nelder, J., Wedderburn, R. Generalized linear models. J. R. Stat. Soc. (1972). https://doi.org/10.2307/2344614
Neske, G.T., Patrick, S.L., Connors, B.W.: Contributions of diverse excitatory and inhibitory neurons to recurrent network activity in cerebral cortex. J. Neurosci. 35, 1089–1105 (2015). https://doi.org/10.1523/JNEUROSCI.2279-14.2015
Nicolelis, M.A.L., Baccala, L.A., Lin, R.C.S., Chapin, J.K.: Sensorimotor encoding by synchronous neural ensemble activity at multiple levels of the somatosensory system. Science 268, 1353–1358 (1995)
Nicolelis, M.A.L., Fanselow, E.E.: Thalamocortical optimization of tactile processing according to behavioral state. Nat. Neurosci. 5, 517–523 (2002). https://doi.org/10.1038/nn0602-517
Pais-Vieira, M., Kunicki, C., Tseng, P.-H., et al.: Cortical and thalamic contributions to response dynamics across layers of the primary somatosensory cortex during tactile discrimination. J. Neurophysiol. 114, 1652–1676 (2015). https://doi.org/10.1152/jn.00108.2015
Petermann, T., Thiagarajan, T.C., Lebedev, M.A., et al.: Spontaneous cortical activity in awake monkeys composed of neuronal avalanches. Proc. Natl. Acad. Sci. 106, 15921–15926 (2009). https://doi.org/10.1073/pnas.0904089106
Pikovsky, A.: Reconstruction of a neural network from a time series of firing rates. Phys. Rev. E 93, 062313 (2016)
Priesemann, V.: Spike avalanches in vivo suggest a driven, slightly subcritical brain state 8, 1–17 (2014). https://doi.org/10.3389/fnsys.2014.00108
Prigogine, I., Lefever, R.: Symmetry breaking instabilities in dissipative systems. II. J Chem Phys 48, 1695–1700 (1968). https://doi.org/10.1063/1.1668896
Prigogine, I., Nicolis, G.: On symmetry-breaking instabilities in dissipative systems. J. Chem. Phys. 46, 3542–3550 (1967). https://doi.org/10.1063/1.1841255
Puzerey, P.A., Kodama, N.X., Galán, R.F.: Abnormal cell-intrinsic and network excitability in the neocortex of serotonin-deficient Pet-1 knockout mice. J. Neurophysiol. (2016). https://doi.org/10.1152/jn.00996.2014
Quiroga, R.Q., Nadasdy, Z., Ben-Shaul, Y.: Unsupervised spike detection and sorting with wavelets and superparamagnetic clustering. Neural Comput. 16, 1661–1687 (2004). https://doi.org/10.1162/089976604774201631
Rajan, K., Abbott, L.F.: Eigenvalue spectra of random matrices for neural networks. Phys. Rev. Lett. 97, 2–5 (2006). https://doi.org/10.1103/physrevlett.97.188104
Renart, A., de la Rocha, J., Bartho, P., et al.: The asynchronous state in cortical circuits. Science 327, 587–590 (2010). https://doi.org/10.1126/science.1179850
Sadovsky, A.J., MacLean, J.N.: Scaling of topologically similar functional modules defines mouse primary auditory and somatosensory microcircuitry. J. Neurosci. 33, 14048–14060 (2013). https://doi.org/10.1523/JNEUROSCI.1977-13.2013
Sederberg, A.J., Palmer, S.E., MacLean, J.N.: Decoding thalamic afferent input using microcircuit spiking activity. J. Neurophysiol. 113, 2921–2933 (2015). https://doi.org/10.1152/jn.00885.2014
Sporns, O., Zwi, J.D.: The small world of the cerebral cortex. Neuroinformatics 2, 145–162 (2004). https://doi.org/10.1385/NI:2:2:145
Steinke, G.K., Galán, R.F.: Brain rhythms reveal a hierarchical network organization. PLoS Comput. Biol. 7, e1002207 (2011). https://doi.org/10.1371/journal.pcbi.1002207
Tomen, N., Rotermund, D., Ernst, U.: Marginally subcritical dynamics explain enhanced stimulus discriminability under attention. Front. Syst. Neurosci. 8, 1–15 (2014). https://doi.org/10.3389/fnsys.2014.00151
Touboul, J., Destexhe, A.: Power-law statistics and universal scaling in the absence of criticality. Phys. Rev. E 012413, 1–15 (2017). https://doi.org/10.1103/PhysRevE.95.012413
Watson, B.O., MacLean, J.N., Yuste, R.: UP States protect ongoing cortical activity from thalamic inputs. PLoS One 3, (2008). https://doi.org/10.1371/journal.pone.0003971
Wilting, J., Priesemann, V.: Inferring collective dynamical states from widely unobserved systems (2018)
Zapperi, S., Lauritsen, K.B., Stanley, H.E.: Self-organized branching processes: Mean-field theory for avalanches. Phys. Rev. Lett. 75, 4071–4074 (1995). https://doi.org/10.1103/physrevlett.75.4071
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2019 Springer Nature Switzerland AG
About this chapter
Cite this chapter
Kodama, N.X., Galán, R.F. (2019). Linear Stability of Spontaneously Active Local Cortical Circuits: Is There Criticality on Long Time Scales?. In: Tomen, N., Herrmann, J., Ernst, U. (eds) The Functional Role of Critical Dynamics in Neural Systems . Springer Series on Bio- and Neurosystems, vol 11. Springer, Cham. https://doi.org/10.1007/978-3-030-20965-0_8
Download citation
DOI: https://doi.org/10.1007/978-3-030-20965-0_8
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-20964-3
Online ISBN: 978-3-030-20965-0
eBook Packages: Biomedical and Life SciencesBiomedical and Life Sciences (R0)