Abstract
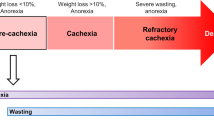
Cachexia is a clinical syndrome of wasting that accompanies many chronic diseases including cancer, renal failure, and heart failure. This condition is marked by an increase in energy expenditure and preferential loss of lean body mass, creating a striking catabolic state. Cachexia contrasts with starvation, a state in which energy expenditure decreases and muscle mass is maintained while fat stores are consumed. In contrast, cachexia is accompanied by a paradoxical anorexia that occurs despite ongoing weight loss and negative calorie balance. This loss of appetite is a significant component of the decreased quality of life experienced by patients with cachexia. Few treatments have proved to be of significant benefit to patients suffering from cachexia. One new treatment that shows promise is pharmacologic blockade of the central melanocortin system. The importance of this system in maintaining normal body weight in humans is highlighted by the finding that disordered melanocortin signaling results in early-onset morbid obesity and dramatic increases in lean body mass in humans. Emerging evidence suggests that blocking this system via pharmacologic antagonists to the type 4 melanocortin receptor (MC4R) may restore appetite and lean body mass in subjects with cachexia caused by a variety of underlying disorders. This review will focus on current pathophysiologic mechanisms involved in cachexia and will outline the central melanocortin pathway as pertaining to these diseases and summarize early animal data using MC4R antagonists to treat cachexia.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
Abstract
Katz, AM and Katz, PB, Diseases of the heart in the works of Hippokrates. Br Heart J, 1962. 24: p. 257–264.
Deans, C and Wigmore, SJ, Systemic inflammation, cachexia and prognosis in patients with cancer. Curr Opin Clin Nutr Metab Care, 2005. 8(3): p. 265–269.
Larkin, M, Thwarting the dwindling progression of cachexia. Lancet, 1998. 351: p. 1336.
Laviano, A, Meguid, MM, Inui, A, et al., Therapy insight: Cancer anorexia-cachexia syndrome–when all you can eat is yourself. Nat Clin Pract Oncol, 2005. 2(3): p. 158–165.
Maltoni, M, Caraceni, A, Brunelli, C, et al., Prognostic factors in advanced cancer patients: evidence-based clinical recommendations–a study by the Steering Committee of the European Association for Palliative Care. J Clin Oncol, 2005. 23(25): p. 6240–6248.
Guenter, P, Muurahainen, N, Simons, G, et al., Relationships among nutritional status, disease progression, and survival in HIV infection. J Acquir Immune Defic Syndr, 1993. 6(10): p. 1130–1138.
Anker, SD and Sharma, R, The syndrome of cardiac cachexia. Int J Cardiol, 2002. 85(1): p. 51–66.
Mehrotra, R and Kopple, JD, Nutritional management of maintenance dialysis patients: why aren’t we doing better? Annu Rev Nutr, 2001. 21: p. 343–379.
Qureshi, AR, Alvestrand, A, Divino-Filho, JC, et al., Inflammation, malnutrition, and cardiac disease as predictors of mortality in hemodialysis patients. J Am Soc Nephrol, 2002. 13 Suppl 1: p. S28–36.
Tisdale, MJ, Cachexia in cancer patients. Nat Rev Cancer, 2002. 2(11): p. 862–871.
Gelin, J, Moldawer, LL, Lonnroth, C, et al., Role of endogenous tumor necrosis factor alpha and interleukin 1 for experimental tumor growth and the development of cancer cachexia. Cancer Res, 1991. 51(1): p. 415–421.
Plata-Salaman, CR, Immunomodulators and feeding regulation: A humoral link between the immune and nervous systems. Br Behav Immun, 1989. 3: p. 193–213.
Moldawer, LL, Andersson, C, Gelin, J, et al., Regulation of food intake and hepatic protein synthesis by recombinant-derived cytokines. Am J Physiol, 1988. 254(3 Pt 1): p. G450–456.
Plata-Salaman, CR, Immunomodulators and feeding regulation: a humoral link between the immune and nervous systems. Brain Behav Immun, 1989. 3(3): p. 193–213.
Scott, HR, McMillan, DC, Crilly, A, et al., The relationship between weight loss and interleukin 6 in non-small-cell lung cancer. Br J Cancer, 1996. 73(12): p. 1560–1562.
Abad, LW, Schmitz, HR, Parker, R, et al., Cytokine responses differ by compartment and wasting status in patients with HIV infection and healthy controls. Cytokine, 2002. 18(5): p. 286–293.
Bonfield, TL, Panuska, JR, Konstan, MW, et al., Inflammatory cytokines in cystic fibrosis lungs. Am J Respir Crit Care Med, 1995. 152(6 Pt 1): p. 2111–2118.
Levine, B, Kalman, J, Mayer, L, et al., Elevated circulating levels of tumor necrosis factor in severe chronic heart failure. N Engl J Med, 1990. 323(4): p. 236–241.
Roubenoff, R, Roubenoff, RA, Cannon, JG, et al., Rheumatoid cachexia: cytokine-driven hypermetabolism accompanying reduced body cell mass in chronic inflammation. J Clin Invest, 1994. 93(6): p. 2379–2386.
Torre-Amione, G, Kapadia, S, Benedict, C, et al., Proinflammatory cytokine levels in patients with depressed left ventricular ejection fraction: a report from the Studies of Left Ventricular Dysfunction (SOLVD). J Am Coll Cardiol, 1996. 27(5): p. 1201–1206.
Wigmore, SJ, Fearon, KC, Maingay, JP, et al., Down-regulation of the acute-phase response in patients with pancreatic cancer cachexia receiving oral eicosapentaenoic acid is mediated via suppression of interleukin-6. Clin Sci (Lond), 1997. 92(2): p. 215–221.
Inui, A, Cancer Anorexia-Cachexia Syndrome: Are Neuropeptides the Key? Cancer Res, 1999. 59: p. 4493–4501.
Plata-Salaman, CR, Brain mechanisms in cytokine-induced anorexia. Psychoneuroendocrinology, 1998. 24: p. 25–41.
Turrin, NP, Ilyin, SE, Gayle, DA, et al., Interleukin-1beta system in anorectic catabolic tumor-bearing rats. Curr Opin Clin Nutr Metab Care, 2004. 7(4): p. 419–426.
Plata-Salaman, CR and Borkoski, JP, Chemokines/intercrines and central regulation of feeding. Am J Physiol, 1994. 266: p. R1711–R1715.
Plata-Salaman, CR, Sonti, G, Borkoski, JP, et al., Anorexia induced by chronic central administration of cytokines at estimated pathophysiological concentrations. Phys Behav, 1996. 60: p. 867–875.
Szelenyi, J, Cytokines and the central nervous system. Brain Res Bull, 2001. 54(4): p. 329–338.
Barber, MD, Fearon, KC, Delmore, G, et al., Should cancer patients with incurable disease receive parenteral or enteral nutritional support? Eur J Cancer, 1998. 34(3): p. 279–285.
Della Cuna, GR, Pellegrini, A, and Piazzi, M, Effect of methylprednisolone sodium succinate on quality of life in preterminal cancer patients: a placebo-controlled, multicenter study. The Methylprednisolone Preterminal Cancer Study Group. Eur J Cancer Clin Oncol, 1989. 25(12): p. 1817–1821.
Loprinzi, CL, Schaid, DJ, Dose, AM, et al., Body-composition changes in patients who gain weight while receiving megestrol acetate. J Clin Oncol, 1993. 11(1): p. 152–154.
Mann, M, Koller, E, Murgo, A, et al., Glucocorticoidlike activity of megestrol. A summary of Food and Drug Administration experience and a review of the literature. Arch Intern Med, 1997. 157(15): p. 1651–1656.
Eriksson, C, Nobel, S, Winblad, B, et al., Expression of interleukin 1 alpha and beta, and interleukin 1 receptor antagonist mRNA in the rat central nervous system after peripheral administration of lipopolysaccharides. Cytokine, 2000. 12(5): p. 423-431.
Gayle, D, Ilyin, SE, Flynn, MC, et al., Lipopolysaccharide (LPS)- and muramyl dipeptide (MDP)-induced anorexia during refeeding following acute fasting: Characterization of brain cytokine and neuropeptide systems mRNAs. Brain Res, 1998. 795: p. 77–86.
Laye, S, Gheusi, G, Cremona, S, et al., Endogenous brain IL-1 mediates the response to peripheral LPS. Am J Phys Reg Integr Comp Physiol, 2000. 279(1): p. R93–R98.
Takao, T, Hashimoto, K, and De Souza, EB, Modulation of interleukin-1 receptors in the neuro-endocrine-immune axis. Int J Dev Neurosci, 1995. 13(3-4): p. 167–178.
Ericsson, A, C., L, Hart, RP, et al., Type 1 interleukin-1 receptor in the rat brain: distribution, regulation, and relationship to sites of IL-1-induced cellular activation. J Comp Neurol, 1995. 361(4): p. 681–698.
Nadeau, S and Rivest, S, Effects of circulating tumor necrosis factor on the neuronal activity and expression of the genes encoding the tumor necrosis factor receptors (p55 and p75) in the rat brain: a view from the blood-brain barrier. Neuroscience, 1999. 93(4): p. 1449–1464.
Utsuyama, M and Hirokawa, K, Differential expression of various cytokine receptors in the brain after stimulation with LPS in young and old mice. Exp Gerontol, 2002. 37(2-3): p. 411–420.
Yamakuni, H, Minami, M, and Satoh, M, Localization of mRNA for leukemia inhibitory factor receptor in the adult rat brain. J Neuroimmunol, 1996. 70(1): p. 45–53.
Elmquist, JK, Scammell, TE, Jacobsen, CD, et al., Distribution of Fos-like immunoreactivity in the rat brain following intravenous lipopolysaccharide administration. J Comp Neurol, 1996. 371(1): p. 85–103.
Laflamme, N and Rivest, S, Effects of systemic immunogenic insults and circulating proinflammatory cytokines on the transcription of the inhibitory factor kappaB alpha within specific cellular populations of the rat brain. J Neurochem, 1999. 73(1): p. 309–321.
Broback, JR, Tepperman, J, and Long, CNH, Experimental hypothalamic hyperphagia in the albino rat. Yale Journal of Biological Medicine, 1943. 15: p. 831–853.
Hotheriogtoa, AW and Ranson, SW, Hypothalamic lesions and adiposity in the rat. Anatomical Record, 1940. 78: p. 149–172.
Muller, HL, Bruhnken, G, Emser, A, et al., Longitudinal study on quality of life in 102 survivors of childhood craniopharyngioma. Childs Nerv Syst, 2005. 21(11): p. 975–980.
Cone, RD, Cowley, MA, Butler, AA, et al., The arcuate nucleus as a conduit for diverse signals relevant to energy homeostasis. Int J Obes Relat Metab Disord, 2001. 25 Suppl 5: p. S63–67.
Cowley, MA, Smith, RG, Diano, S, et al., The distribution and mechanism of action of ghrelin in the CNS demonstrates a novel hypothalamic circuit regulating energy homeostasis. Neuron, 2003. 37(4): p. 649–661.
Schwartz, MW, Seeley, RJ, Campfield, LA, et al., Identification of targets of leptin action in rat hypothalamus. J Clin Invest, 1996. 98(5): p. 1101–1106.
Rizk, NM, Joost, HG, and Eckel, J, Increased hypothalamic expression of the p75 tumor necrosis factor receptor in New Zealand obese mice. Horm Metab Res, 2001. 33(9): p. 520–524.
Yabuuchi, K, Minami, M, Katsumata, S, et al., Localization of type-1 interleukin-1 receptor mRNA in the rat brain. Brain Res Mol Brain Res, 1994. 27(1): p. 27–36.
Jacobowitz, DM and O’Donohue, TL, a-Melanocyte-stimulating hormone: immunohistochemical identification and mapping in neurons of rat brain. Proc Natl Acad Sci USA, 1978. 75: p. 6300–6304.
Benoit, S, Schwartz, M, Baskin, D, et al., CNS melanocortin system involvement in the regulation of food intake. Horm Behav, 2000. 37(4): p. 299–305.
Cone, RD, Anatomy and regulation of the central melanocortin system. Nat Neurosci, 2005. 8(5): p. 571–578.
Fan, W, Boston, BA, Kesterson, RA, et al., Role of melanocortinergic neurons in feeding and the agouti obesity syndrome. Nature, 1997. 385: p. 165–168.
Huszar, D, Lynch, CA, Fairchild-Huntress, V, et al., Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell, 1997. 88: p. 131–141.
Vaisse, C, Clement, K, Guy-Grand, B, et al., A frameshift mutation in human MC4R is associated with a dominant form of obesity. Nature Genetics, 1998. 20: p. 113–114.
Yeo, GS, Farooqi, IS, Aminian, S, et al., A frameshift mutation in MC4R associated with dominantly inherited human obesity. Nat Genet, 1998. 20(2): p. 111–112.
Farooqi, IS, Keogh, JM, Yeo, GS, et al., Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N Engl J Med, 2003. 348(12): p. 1085–1095.
Fan, W, Dinulescu, DM, Butler, AA, et al., The central melanocortin system can directly regulate serum insulin levels. Endocrinology, 2000. 141(9): p. 3072–3079.
Haskell-Luevano, C, Chen, P, Li, C, et al., Characterization of the neuroanatomical distribution of agouti-related protein immunoreactivity in the rhesus monkey and the rat. Endocrinology, 1999. 140(3): p. 1408–1415.
Fong, TM, Mao, C, MacNeil, C, et al., ART (protein product of agouti-related transcript) as an antagonist of MC-3 and MC-4 receptors. Biochem Biophys Res Commun, 1997. 237: p. 629–631.
Ollmann, MM, Wilson, BD, Yang, YK, et al., Antagonism of central melanocortin receptors in vitro and in vivo by agouti-related protein. Science, 1997. 278(5335): p. 135–138.
Mizuno, TM and Mobbs, CV, Hypothalamic agouti-related protein messenger ribonucleic acid is inhibited by leptin and stimulated by fasting. Endocrinology, 1999. 140(2): p. 814–817.
Hagan, MM, Rushing, PA, Pritchard, LM, et al., Long-term orexigenic effects of AgRP-(83-132) involve mechanisms other than melanocortin receptor blockade. Am J Physiol Regul Integr Comp Physiol, 2000. 279(1): p. R47–R52.
Nijenhuis, WA, Oosterom, J, and Adan, RA, AGRP (83-132) acts as an inverse agonist on the human melanocortin-4 receptor. Mol Endocrinol, 2001. 15(1): p. 164–171.
Rivest, S and Laflamme, N, Neuronal activity and neuropeptide gene transcription in the brains of immune-challenged rats. J Neuroendocrinol, 1995. 7(7): p. 501–525.
Scarlett, JM, Joppa, MA, Markison, S, et al., Direct activation of the hypothalamic melanocortin system by interleukin-1. Abs Endocr Soc (2004):OR6-3, 2005.
Lennie, TA, Relationship of body energy status to inflammation-induced anorexia and weight loss. Physiol Behav, 1998. 64(4): p. 475–481.
Lennie, TA, Wortman, MD, and Seeley, RJ, Activity of body energy regulatory pathways in inflammation-induced anorexia. Physiol Behav, 2001. 73(4): p. 517–523.
Sergeyev, V, Broberger, C, and Hokfelt, T, Effect of LPS administration on the expression of POMC, NPY, galanin, CART and MCH mRNAs in the rat hypothalamus. Brain Res Mol Brain Res, 2001. 90(2): p. 93–100.
Huang, QH, Hruby, VJ, and Tatro, JB, Role of central melanocortins in endotoxin-induced anorexia. Am J Physiol, 1999. 276(3 pt 2): p. R864–R871.
Tatro, JB, Huszar, D, Fairchild-Huntress, V, et al., Role of the melanocortin-4 receptor in thermoregulatory responses to central IL-1 Society for Neuroscience Abstracts, 1999. 25(624.4): p. 558.
Lawrence, CB and Rothwell, NJ, Anorexic but not pyrogenic actions of interleukin-1 are modulated by central melanocortin-3/4 receptors in the rat. J Neuroendocrinol, 2001. 13(6): p. 490–495.
Marks, DL, Butler, AA, Turner, R, et al., Differential role of melanocortin receptor subtypes in cachexia. Endocrinology, 2003. 144(4): p. 1513–1523.
Marks, DL, Ling, N, and Cone, RD, Role of the Central Melanocortin System in Cachexia. Cancer Res, 2001. 61(4): p. 1432–1438.
Wisse, BE, Frayo, RS, Schwartz, MW, et al., Reversal of cancer anorexia by blockade of central melanocortin receptors in rats. Endocrinology, 2001. 142(8): p. 3292–3301.
Cheung, W, Yu, PX, Little, BM, et al., Role of leptin and melanocortin signaling in uremia-associated cachexia. J Clin Invest, 2005. 115(6): p. 1659–1665.
Vos, TJ, Caracoti, A, Che, JL, et al., Identification of 2-[2-[2-(5-bromo-2- methoxyphenyl)-ethyl]-3-fluorophenyl]-4,5-dihydro-1H-imidazole (ML00253764), a small molecule melanocortin 4 receptor antagonist that effectively reduces tumor-induced weight loss in a mouse model. J Med Chem, 2004. 47(7): p. 1602–1604.
Markison, S, Foster, AC, Chen, C, et al., The regulation of feeding and metabolic rate and the prevention of murine cancer cachexia with a small-molecule melanocortin-4 receptor antagonist. Endocrinology, 2005. 146(6): p. 2766–2773.
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2007 Humana Press Inc.
About this chapter
Cite this chapter
Marks, D.L. (2007). The Role of Central Melanocortins in Cachexia. In: Donohoue, P.A. (eds) Energy Metabolism and Obesity. Contemporary Endocrinology. Humana Press. https://doi.org/10.1007/978-1-60327-139-4_4
Download citation
DOI: https://doi.org/10.1007/978-1-60327-139-4_4
Publisher Name: Humana Press
Print ISBN: 978-1-58829-671-9
Online ISBN: 978-1-60327-139-4
eBook Packages: MedicineMedicine (R0)