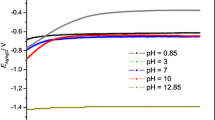
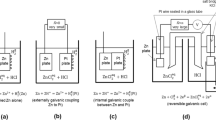
Abstract
The driving force for a reaction is the change in Gibbs free energy, AG, for reactants to products. Mathematically this may be expressed by
The summation signs are used as a general notation to indicate that all reactants and products are included in the calculation. From the nature of the free energy function, this calculation applies to initial (reactants) and final (product) states, and is independent of intervening states. The reaction may be investigated under controlled reversible conditions such as in an electrochemical cell, or under irreversible conditions such as in corrosion, and the same total free energy change (ΔG) will be appropriate. A quite general predictive capability may be applied to specific corrosion reactions since all the available thermodynamic data may be used for corrosion calculations directly. This enables the position of final equilibrium of the corrosion system to be established. The thermodynamic calculations have the limitation that no information of the rate of the reaction is provided, only what the final state will be for the process. Another limitation of the thermodynamic treatment is found when there are multiple reactions in a corrosion system but none are dominant, so that the final state is not well known. The limitations will not be described further here, but the influence of kinetics on corrosion behavior is significant and is the subject of the remaining sections. However, this section will briefly describe the sources of thermodynamic data and procedures for using it, since this remains a powerful resource despite the limitations.
This work was supported by United States Department of Energy (DOE) under contract DE-AC04-76DP00789. Sandia Laboratories is a DOE facility.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Preview
Unable to display preview. Download preview PDF.
Similar content being viewed by others
References
E. A. Guggenheim, Thermodynamics, North-Holland, Amsterdam (1959).
G. N. Lewis and M. Randall, Thermodynamics, revised by K. S. Pitzer and L. Brewer, McGraw-Hill, New York (1961).
NBS Technical Notes 270–3, 270–4, 270–5, U.S. Government Printing Office (1968–1971).
W. M. Latimer, Oxidation Potentials, Prentice-Hall, Englewood Cliffs, New Jersey (1952).
JANAF Thermochemical Tables, NSRDS-NBS 37, U.S. Government Printing Office (1971).
J. A. Plambeck, Fused Salt Systems, Encyclopedia of Electrochemistry of the Elements, Vol. X, A. J. Bard, ed., Marcel Dekker, New York (1976).
H. Leidheiser, Jr., The Corrosion of Copper, Tin, and Their Alloys, John Wiley and Sons, New York (1971).
M. Pourbaix, Atlas of Potential/pH Diagrams, Pergamon, Oxford (1962).
G. M. Florianovich and Ya. M. Kolotyrkin, Dokl. Akad. Nauk SSSR. 157, 422 (1964).
P. Delahay, Double Layer and Electrode Kinetics, Interscience, New York (1966).
K. J. Vetter, Electrochemical Kinetics, Academic Press, New York (1967).
J. O’M. Bockris and A. K. N. Reddy, Modern Electrochemistry, Plenum Press, New York (1970).
J. O’M. Bockris, in Modern Aspects of Electrochemistry, Vol. 1, J. O’M. Bockris, ed., Butterworths, London (1954).
T. N. Anderson and H. Eyring, Physical Chemistry, H. Eyring, D. Henderson, W. Jost, Eds., Academic Press, New York (1970).
N. Tanaka and R. Tamamushi, Electrochim. Acta 9, 963 (1964).
A. N. Frumkin, Advances in Electrochemistry and Electrochemical Engineering, Vol. 1, P. Delahay and C. W. Tobias, Eds., Interscience, New York (1961).
A. N. Frumkin, Advances in Electrochemistry and Electrochemical Engineering, Vol. III, P. Delahay and C. W. Tobias, Eds., Interscience, New York (1963).
J. P. Hoare, Advances in Electrochemistry and Electrochemical Engineering, Vol. VI, P. Delahay and C. W. Tobias, Eds., Interscience, New York (1967).
J. P. Hoare, The Electrochemistry of Oxygen, Interscience, New York (1968).
A. Damjanovic, Modern Aspects of Electrochemistry, Vol. 5, J. O’M. Bockris and B. E. Conway, Eds., Plenum Press, New York (1969).
C. Wagner and W. Traud, Z. Electrochem. 44, 391 (1938).
H. Kaesche, Z. Metallk. 61, 94 (1970).
M. Stern, Corrosion (Houston) 14, 440t (1958).
M. Stern and A. L. Geary, J. Electrochem. Soc. 104, 56 (1957).
F. Mansfeld, Advances in Corrosion Science and Corrosion Engineering, Vol. 8, M. Fontana and R. Staehle Eds., Plenum Press, New York (1978).
C. Wagner, J. Electrochem. Soc. 98, 116 (1951).
J. Newman, J. Electrochem. Soc. 113, 1235 (1966).
J. S. Newman, Electrochemical Systems, Prentice-Hall, Englewood Cliffs, New Jersey (1973).
J. O’M. Bockris, D. Drazic, and A. R. Despic, Electrochim. Acta 4, 325 (1961).
E. J. Kelly, J. Electrochem. Soc. 112, 124 (1965).
S. Asakura and K. Nobe, J. Electrochem. Soc. 118, 13 (1971); S. Asakura and K. Nobe, J. Electrochem. Soc. 118, 19 (1971); R. J. Chin and K. Nobe, J. Electrochem. Soc. 119, 1457 (1972); H. C. Kuo and K. Nobe, 125, 853 (1978).
H. A. Uhlig, Corrosion and Corrosion Control, (New York: John Wiley and Sons, New York (1971).
T. N. Anderson, M. H. Ghandehari, and H. Eyring, J. Electrochem. Soc. 122, 1580 (1975).
A. W. Tracy, in Corrosion Resistance of Metals and Alloys, F. L. LaQue and H. R. Copson, Eds., Reinhold, New York (1963).
U. Bertocci, Encyclopedia of Electrochemistry of the Elements, Vol. II, A. J. Bard, ed., Marcel Dekker, New York (1974).
U. Bertocci, Electrochim. Acta 11, 1261 (1966).
B. C. Y. Lu and W. F. Graydon, Can. J. Chem. 32, 153 (1954).
D. P. Gregory and A. C. Riddiford, J. Electrochem. Soc. 107, 950 (1960).
I. Cornet, E. A. Barrington, and G. U. Behrsing, J. Electrochem. Soc. 108, 947 (1961).
G. Demerits and A. P. van Peteghem, Corros. Sci. 18, 1041 (1978).
W. H. Smyrl, in Electrochemical Techniques for Corrosion Investigations, F. Mansfeld and U. Bertocci, Eds., American Society for Testing Materials, Philadelphia (1980).
E. Mattson and J. O’M. Bockris, Trans. Faraday Soc. 55, 1586 (1959).
W. H. Smyrl, unpublished data.
R. K. Astakhova and B. S. Krasikov, Zh. Prikl. Khim. 44, 363 (1972).
T. Hurlen, Acta Chem. Scand. 15, 1231 (1961).
M. Braun and K. Nobe, J. Electrochem. Soc. 126, 1666 (1979).
B. Miller and M. I. Bellavance, J. Electrochem. Soc. 119, 1510 (1972).
A. L. Baccarella and J. C. Griess, Jr., J. Electrochem. Soc. 120, 459 (1973).
R. V. Homsy and J. Newman, J. Electrochem. Soc. 121, 521 (1974).
J. O’M. Bockris and N. Pentland, Trans. Faraday Soc. 48, 833 (1952).
C. H. Bonfiglio, H. C. Albaya, and O. A. Cobo, Corros. Sci. 13, 717 (1973).
M. Turner and P. A. Brock, Corros. Sci. 13, 973 (1973).
G. Faita, G. Fiori, and D. Salvadore, Corros. Sci. 15, 383 (1975).
R. P. Frankenthal and J. Kruger, Eds., Proceedings of the Fourth International Symposium on Passivity, The Electrochemistry Society, Princeton, New Jersey (1978).
Proceedings of the Fifth International Congress on Metallic Corrosion, NACE, Houston (1974).
J. Newman, in Advances in Electrochemistry and Electrochemical Engineering, Vol. 5, P. Delahay and C. W. Tobias Eds., Interscience, New York (1967).
R. A. Robinson and R. H. Stokes, Electrolyte Solutions, Butterworths, London (1959).
C. Wagner, J. Electrochem. Soc. 99, 1 (1952).
J. Newman, in Localized Corrosion, R. Staehle, ed., NACE, Houston (1974).
W. H. Smyrl and J. Newman, J. Electrochem. Soc. 119, 208 (1972).
V. G. Levich, Physicochemical Hydrodynamics, Prentice-Hall, Englewood Cliffs, New Jersey (1962).
B. T. Ellison and I. Cornet, J. Electrochem. Soc. 118, 68 (1971).
F. L. LaQue, Corrosion (Houston) 13, 303t (1957).
J. R. Selman and C. W. Tobias, in Advances in Chemical Engineering, Vol. 10, T. B. Drew, G. R. Cokelet, J. W. Hoopes, Jr., and T. Vermeulen, Eds., Academic Press, New York (1978).
D. W. Siitari and R. C. Alkire, paper 238 presented at the Electrochemical Society Meeting, Los Angeles, October 1979.
J. Newman and W. Tiedeman, in Advances in Electrochemistry and Electrochemical Engineering, Vol. II, H. Gerischer and C. W. Tobias, Eds., Wiley Interscience, New York (1978).
R. C. Alkire and D. W. Siitari, J. Electrochemical Soc. 126, 15 (1979).
J. Newman, D. N. Hanson, and K. Vetter, Electrochim. Acta 22, 829 (1977).
W. H. Smyrl, unpublished data.
R. C. Alkire and G. Nicolaides, J. Electrochem. Soc. 121, 183 (1974).
M. Stern, Corrosion (Houston) 14, 329t (1958).
K. B. Oldham and F. Mansfeld, J. Appl. Electrochem. 2, 183 (1972).
V. G. Levich and A. Frumkin, Acta Physicochim. URSS 18, 1 (1943).
J. Newman, J. Electrochem. Soc. 113, 501 (1966).
L. Nanis and W. Kesselman, J. Electrochem. Soc. 118, 454 (1971).
J. T. Waber, J. Electrochem. Soc. 101, 271 (1954).
J. T. Waber and M. Rosenbluth, J. Electrochem. Soc. 102, 344 (1955).
J. T. Waber, J. Electrochem. Soc. 102, 420 (1955).
J. T. Waber, J. Morrissey, and J. Ruth, J. Electrochem. Soc. 103, 138 (1956).
J. T. Waber, J. Electrochem. Soc. 103, 567 (1956).
J. A. Simmons, S. R. Coriell, and F. Ogburn, J. Electrochem. Soc. 114, 782 (1967).
P. Doig and P. E. J. Flewitt, Philos. Mag. B 38, 27 (1978); Br. Corros. J. 13, 118 (1978).
E. McCafferty, J. Electrochem. Soc. 124, 1869 (1977).
L. Gal-Or, Y. Raz, and J. Yahalom, J. Electrochem. Soc. 120, 598 (1973).
W. H. Smyrl and J. Newman, J. Electrochem. Soc. 123, 1423 (1976).
W. H. Smyrl, unpublished data.
Author information
Authors and Affiliations
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 1981 Springer Science+Business Media New York
About this chapter
Cite this chapter
Smyrl, W.H. (1981). Electrochemistry and Corrosion on Homogeneous and Heterogeneous Metal Surfaces. In: Bockris, J.O., Conway, B.E., Yeager, E., White, R.E. (eds) Electrochemical Materials Science. Comprehensive Treatise of Electrochemistry, vol 4. Springer, Boston, MA. https://doi.org/10.1007/978-1-4757-4825-3_2
Download citation
DOI: https://doi.org/10.1007/978-1-4757-4825-3_2
Publisher Name: Springer, Boston, MA
Print ISBN: 978-1-4757-4827-7
Online ISBN: 978-1-4757-4825-3
eBook Packages: Springer Book Archive