Abstract
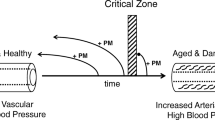
Arterial stiffness is a perfect example of translational research spanning the understanding of the molecular determinants of the arterial wall constituents and their organization to the physiology of normal and early vascular ageing.
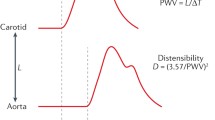
The most widely used parameter to investigate arterial stiffness in rodents is pulse wave velocity (PWV). The relation between strain and stress is also established to characterize the intrinsic behaviour of the arterial wall independent of geometric factors.
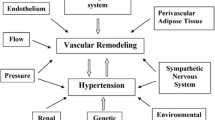
The first attempts to explain arterial stiffness by the properties of the structural components of the arterial wall addressed the role of the principal constituents, elastin and collagen fibres and smooth muscle cells. To complete this approach, the roles of the adhesion molecules, inflammation, blood pressure variability, NO, integrins and metalloproteinases in arterial stiffness were also investigated by attempting to try and interfere directly with these different factors.
Hypertensive rodents were the first experimental models used and employed to test remodelling and vascular function in an environment mimicking human physiology. Then, investigations were completed with other cardiovascular animal models (mainly kidney disease, obesity, blood pressure variability or ageing) to discover more specific therapeutic targets related to other mechanisms that trigger arterial stiffness. Nowadays, the advances in mouse genetics have provided numerous genotypes and phenotypes to study changes in arterial mechanics with disease progression and treatment. The aim of modifying a single gene to understand the mechanism of this polygenic disease has led to complete animal models using in vitro cellular approaches.
The response to these questions will ultimately come from more complete knowledge of the mechanisms involved, using pharmacological and other tools in existing and/or newly emerging animal models.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Safar ME, Levy BI, Struijker-Boudier H. Current perspectives on arterial stiffness and pulse pressure in hypertension and cardiovascular diseases. Circulation. 2003;107(22):2864–9.
Pinto YM, Paul M, Ganten D. Lessons from rat models of hypertension: from Goldblatt to genetic engineering. Cardiovasc Res. 1998;39(1):77–88.
Monassier L, Combe R, El Fertak L. Mouse models of hypertension. Drug Discov Today. 2006;3(3):273–81.
Cvetkovic B, Sigmund CD. Understanding hypertension through genetic manipulation in mice. Kidney Int. 2000;57(3):863–74.
Amin M, Le VP, Wagenseil JE. Mechanical testing of mouse carotid arteries: from newborn to adult. J Vis Exp. 2012;60:e3733.
Sehgel N, Zhu Y, Sun Z, et al. Increased vascular smooth muscle cell stiffness: a novel mechanism for aortic stiffness in hypertension. Am J Physiol Heart Circ Physiol. 2013;305(9):H1281–7.
Meaume S, Benetos A, Henry OF, et al. Aortic pulse wave velocity predicts cardiovascular mortality in subjects >70 years of age. Arterioscler Thromb Vasc Biol. 2001;21(12):2046–50.
Milnor W. Properties of the vascular wall. In: Collins N, editor. Hemodynamics. Baltimore: Williams and Wilkins; 1989. p. 58–101.
Glaser E, Lacolley P, Boutouyrie P, et al. Dynamic versus static compliance of the carotid artery in living Wistar-Kyoto rats. J Vasc Res. 1995;32(4):254–65.
Hayoz D, Rutschmann B, Perret F, et al. Conduit artery compliance and distensibility are not necessarily reduced in hypertension. Hypertension. 1992;20(1):1–6.
Lacolley P, Bezie Y, Girerd X, et al. Aortic distensibility and structural changes in sinoaortic-denervated rats. Hypertension. 1995;26(2):337–40.
Levy B. Mechanics of the large artery wall. In: Lévy BI, Tedgui A, editors. Biology of the arterial wall. Dordrecht: Kluwer; 1999. p. 13–24.
van Gorp AW, van Ingen Schenau DS, Hoeks AP, et al. Aortic wall properties in normotensive and hypertensive rats of various ages in vivo. Hypertension. 1995;26(2):363–8.
Ruiz-Feria CA, Yang Y, Thomason DB, et al. Pulse wave velocity and age- and gender-dependent aortic wall hardening in fowl. Comp Biochem Physiol A Mol Integr Physiol. 2009;154(4):429–36.
Bezie Y, Lamaziere JM, Laurent S, et al. Fibronectin expression and aortic wall elastic modulus in spontaneously hypertensive rats. Arterioscler Thromb Vasc Biol. 1998;18(7):1027–34.
Pauletto P, Scannapieco G, Vescovo G, et al. Catecholamine-induced cardiovascular disease in the spontaneously hypertensive and atherosclerotic turkey. Methods Find Exp Clin Pharmacol. 1988;10(6):357–62.
Hadjiisky P, Bourdillon MC, Grosgogeat Y. Experimental models of atherosclerosis. Contribution, limits and trends. Arch Mal Coeur Vaiss. 1991;84(11):1593–603.
Ng K, Hildreth CM, Phillips JK, et al. Aortic stiffness is associated with vascular calcification and remodeling in a chronic kidney disease rat model. Am J Physiol Renal Physiol. 2011;300(6):F1431–6.
Sloboda N, Feve B, Thornton SN, et al. Fatty acids impair endothelium-dependent vasorelaxation: a link between obesity and arterial stiffness in very old Zucker rats. J Gerontol A Biol Sci Med Sci. 2012;67(9):927–38.
Sista AK, O’Connell MK, Hinohara T, et al. Increased aortic stiffness in the insulin-resistant Zucker fa/fa rat. Am J Physiol Heart Circ Physiol. 2005;289(2):H845–51.
Isabelle M, Simonet S, Ragonnet C, et al. Chronic reduction of nitric oxide level in adult spontaneously hypertensive rats induces aortic stiffness similar to old spontaneously hypertensive rats. J Vasc Res. 2012;49(4):309–18.
Qiu H, Zhu Y, Sun Z, Trzeciakowski JP, et al. Short communication: vascular smooth muscle cell stiffness as a mechanism for increased aortic stiffness with aging. Circ Res. 2010;107(5):615–9.
Cantini C, Kieffer P, Corman B, et al. Aminoguanidine and aortic wall mechanics, structure, and composition in aged rats. Hypertension. 2001;38(4):943–8.
Osborne-Pellegrin M, Labat C, Mercier N, et al. Changes in aortic stiffness related to elastic fiber network anomalies in the Brown Norway rat during maturation and aging. Am J Physiol Heart Circ Physiol. 2010;299(1):H144–52.
Basta G, Schmidt AM, De Caterina R. Advanced glycation end products and vascular inflammation: implications for accelerated atherosclerosis in diabetes. Cardiovasc Res. 2004;63(4):582–92.
Slove S, Lannoy M, Behmoaras J, et al. Potassium channel openers increase aortic elastic fiber formation and reverse the genetically determined elastin deficit in the BN rat. Hypertension. 2013;62(4):794–801.
Dabire H, Lacolley P, Chaouche-Teyara K, et al. Relationship between arterial distensibility and low-frequency power spectrum of blood pressure in spontaneously hypertensive rats. J Cardiovasc Pharmacol. 2002;39(1):98–106.
Bouissou C, Lacolley P, Dabire H, et al. Increased stiffness and cell-matrix interactions of abdominal aorta in two experimental non-hypertensive models: long-term chemical sympathectomized and sinoaortic-denervated rats. J Hypertens. 2014;32:652–8.
Lacolley P, Glaser E, Challande P, et al. Structural changes and in situ aortic pressure-diameter relationship in long-term chemical-sympathectomized rats. Am J Physiol. 1995;269(2 Pt 2):H407–16.
Li ZY, Xu TY, Zhang SL, et al. Telemetric ambulatory arterial stiffness index, a predictor of cardio-cerebro-vascular mortality, is associated with aortic stiffness-determining factors. CNS Neurosci Ther. 2013;19(9):667–74.
Cox RH. Basis for the altered arterial wall mechanics in the spontaneously hypertensive rat. Hypertension. 1981;3(4):485–95.
Ito H. Vascular connective tissue change in hypertension. In: Lee RE, editor. Blood vessel changes in hypertension: structure and function. Boca Raton: CRC Press; 1989. p. 99–122.
Cox RH. Mechanical properties of arteries in hypertension. In: Lee RMKW, editor. Blood vessel changes in hypertension: structure and function. Boca Raton: CRC Press; 1989.
Wagenseil JE, Mecham RP. Vascular extracellular matrix and arterial mechanics. Physiol Rev. 2009;89(3):957–89.
Wagenseil JE, Ciliberto CH, Knutsen RH, et al. Reduced vessel elasticity alters cardiovascular structure and function in newborn mice. Circ Res. 2009;104(10):1217–24.
Wan W, Yanagisawa H, Gleason Jr RL. Biomechanical and microstructural properties of common carotid arteries from fibulin-5 null mice. Ann Biomed Eng. 2010;38(12):3605–17.
Small J, North A. Architecture of the smooth muscle cell. In: Schwartz SM, Mecham RP, editors. The vascular smooth muscle cell. New York: Academic; 1995. p. 169–88.
Bezie Y, Lacolley P, Laurent S, et al. Connection of smooth muscle cells to elastic lamellae in aorta of spontaneously hypertensive rats. Hypertension. 1998;32(1):166–9.
Clark JM, Glagov S. Structural integration of the arterial wall. I. Relationships and attachments of medial smooth muscle cells in normally distended and hyperdistended aortas. Lab Invest. 1979;40(5):587–602.
Osborne-Pellegrin MJ. Some ultrastructural characteristics of the renal artery and abdominal aorta in the rat. J Anat. 1978;125(Pt 3):641–52.
Galmiche G, Labat C, Mericskay M, et al. Inactivation of serum response factor contributes to decrease vascular muscular tone and arterial stiffness in mice. Circ Res. 2013;112(7):1035–45.
Retailleau K, Toutain B, Galmiche G, et al. Selective involvement of serum response factor in pressure-induced myogenic tone in resistance arteries. Arterioscler Thromb Vasc Biol. 2013;33(2):339–46.
Lacolley P, Challande P, Boumaza S, et al. Mechanical properties and structure of carotid arteries in mice lacking desmin. Cardiovasc Res. 2001;51(1):178–87.
Benetos A, Pannier B, Brahimi M, et al. Dose-related changes in the mechanical properties of the carotid artery in WKY rats and SHR following relaxation of arterial smooth muscle. J Vasc Res. 1993;30(1):23–9.
Fridez P, Zulliger M, Bobard F, et al. Geometrical, functional, and histomorphometric adaptation of rat carotid artery in induced hypertension. J Biomech. 2003;36(5):671–80.
Gabella G. Structural apparatus for force transmission in smooth muscles. Physiol Rev. 1984;64(2):455–77.
Ingber D. Integrins as mechanical transducers. Curr Opin Cell Biol. 1991;3(5):841–8.
Wilson E, Sudhir K, Ives HE. Mechanical strain of rat vascular smooth muscle cells is sensed by specific extracellular matrix/integrin interactions. J Clin Invest. 1995;96(5):2364–72.
Saouaf R, Takasaki I, Eastman E, et al. Fibronectin biosynthesis in the rat aorta in vitro. Changes due to experimental hypertension. J Clin Invest. 1991;88:1182–9.
Boumaza S, Arribas SM, Osborne-Pellegrin M, et al. Fenestrations of the carotid internal elastic lamina and structural adaptation in stroke-prone spontaneously hypertensive rats. Hypertension. 2001;37(4):1101–7.
Zanchi A, Wiesel P, Aubert JF, et al. Time course changes of the mechanical properties of the carotid artery in renal hypertensive rats. Hypertension. 1997;29(5):1199–203.
Fitch RM, Vergona R, Sullivan ME, et al. Nitric oxide synthase inhibition increases aortic stiffness measured by pulse wave velocity in rats. Cardiovasc Res. 2001;51:351–8.
Kato H, Hou J, Chobanian AV, et al. Effects of angiotensin II infusion and inhibition of nitric oxide synthase on the rat aorta. Hypertension. 1996;28:153–8.
Labat C, Lacolley P, Lajemi M, et al. Effects of valsartan on mechanical properties of the carotid artery in spontaneously hypertensive rats under high-salt diet. Hypertension. 2001;38(3):439–43.
Mercier N, Labat C, Louis H, et al. Sodium, arterial stiffness, and cardiovascular mortality in hypertensive rats. Am J Hypertens. 2007;20(3):319–25.
Lacolley P, Labat C, Pujol A, et al. Increased carotid wall elastic modulus and fibronectin in aldosterone-salt-treated rats: effects of eplerenone. Circulation. 2002;106(22):2848–53.
Ammarguellat FZ, Gannon PO, Amiri F, et al. Fibrosis, matrix metalloproteinases, and inflammation in the heart of DOCA-salt hypertensive rats: role of ET(A) receptors. Hypertension. 2002;39(2 Pt 2):679–84.
Cox RH. Effects of deoxycorticosterone on arterial wall properties in two-kidney rats. J Hypertens. 1986;4(5):557–65.
Takasaki I, Chobanian AV, Sarzani R, et al. Effect of hypertension on fibronectin expression in the rat aorta. J Biol Chem. 1990;265(35):21935–9.
Intengan HD, Schiffrin EL. Structure and mechanical properties of resistance arteries in hypertension: role of adhesion molecules and extracellular matrix determinants. Hypertension. 2000;36(3):312–8.
Intengan HD, Schiffrin EL. Vascular remodeling in hypertension: roles of apoptosis, inflammation, and fibrosis. Hypertension. 2001;38(3 Pt 2):581–7.
Louis H, Kakou A, Regnault V, et al. Role of alpha1beta1-integrin in arterial stiffness and angiotensin-induced arterial wall hypertrophy in mice. Am J Physiol Heart Circ Physiol. 2007;293(4):H2597–604.
Mao X, Said R, Louis H, et al. Cyclic stretch-induced thrombin generation by rat vascular smooth muscle cells is mediated by the integrin αvβ3 pathway. Cardiovasc Res. 2012;96(3):513–23.
Et-Taouil K, Schiavi P, Lévy BI, et al. Sodium intake, large artery stiffness, and proteoglycans in the spontaneously hypertensive rat. Hypertension. 2001;38(5):1172–6.
Gandley RE, McLaughlin MK, Koob TJ, et al. Contribution of chondroitin-dermatan sulfate-containing proteoglycans to the function of rat mesenteric arteries. Am J Physiol. 1997;273(2 Pt 2):H952–60.
Calvier L, Miana M, Rebou LP, et al. Galectin-3 mediates aldosterone-induced vascular fibrosis. Arterioscler Thromb Vasc Biol. 2013;33(1):67–75.
López-Andrés N, Calvier L, Labat C, et al. Absence of cardiotrophin 1 is associated with decreased age-dependent arterial stiffness and increased longevity in mice. Hypertension. 2013;61(1):120–9.
Nguyen Dinh Cat A, Griol-Charhbili V, Loufrani L, et al. The endothelial mineralocorticoid receptor regulates vasoconstrictor tone and blood pressure. FASEB J. 2010;24(7):2454–63.
Galmiche G, Pizard A, Gueret A, et al. Smooth muscle cell mineralocorticoid receptors are mandatory for aldosterone-salt to induce vascular stiffness. Hypertension. 2014;63:520–6.
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2014 Springer-Verlag London
About this chapter
Cite this chapter
Lacolley, P., Thornton, S.N., Bezie, Y. (2014). Animal Models for Studies of Arterial Stiffness. In: Safar, M., O'Rourke, M., Frohlich, E. (eds) Blood Pressure and Arterial Wall Mechanics in Cardiovascular Diseases. Springer, London. https://doi.org/10.1007/978-1-4471-5198-2_6
Download citation
DOI: https://doi.org/10.1007/978-1-4471-5198-2_6
Published:
Publisher Name: Springer, London
Print ISBN: 978-1-4471-5197-5
Online ISBN: 978-1-4471-5198-2
eBook Packages: MedicineMedicine (R0)