Abstract
Background
Pulmonary tuberculosis (TB) lesion is acidic, and changing this acidic pH may affect growth of TB bacilli and response to therapy. We aimed to assess the effect of adjuvant inhalation of sodium bicarbonate (SB) 8.4% on clinical, radiological, and microbiological responses in patients with sputum-positive drug-sensitive pulmonary TB.
Patients and methods
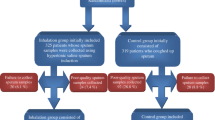
One hundred and three patients with pulmonary TB completed the study, and they were classified into two groups: group I included 55 patients who received standard anti-TB regimen plus SB inhalation, and group II included 48 patients who received anti-TB regimen only. The responses in both groups were evaluated clinically, microbiologically, and radiologically.
Results
There was no statistically significant difference between both groups in baseline bacillary load, clinical picture, and radiology. Both groups improved clinically 1 month after start of therapy. In group I only, there was a statistically significant improvement in chest radiograph after 1 month (P<0.001). The median duration of smear conversion for group I was 3 weeks (1–8) compared with 9.5 (2–17) in group II, with a statistically significant difference (P<0.001). Moreover, the median duration of culture conversion for group I was 1 month (1–3) compared with 3 months (1–4) in group II, with a statistically significant difference (P<0.001).
Conclusion
Adjuvant inhalation of SB in smear-positive pulmonary TB to standard anti-TB drugs accelerates smear conversion, culture conversion, and clinical and radiological improvement.
Article PDF
Similar content being viewed by others
References
Global Tuberculosis Report. Geneva: World Health Organization; 2015. Available at: http://apps.who.int/iris/bitstream/10665/191102/1/9789241565059_eng.pdf?ua=1 http://www.who.int/tb/publications/global_report/indicators_global_and_regional_summaries.pdf. [Accessed 13 September 2016].
Centers for Disease Control and Prevention. Core Curriculum on Tuberculosis: What the Clinician Should Know. Chapter 2 Transmission and Pathogenesis of Tuberculosis. Sixth Edition; 2013. Available at: https://www.cdc.gov/tb/education/corecurr/pdf/chapter2.pdf. [Accessed 13 September 2016].
Republic of South Africa Department of Health. South African National Tuberculosis Management Guidelines; 2014.
Raviglione MC, Snider DE, Kochi A. Global epidemiology of tuberculosis: morbidity and mortality of a worldwide epidemic. J Am Med Assoc 1995; 273:220–226.
Dannenberg AM. Pathogenesis of human pulmonary tuberculosis: insights from the rabbit model. Washington, DC: ASM Press 2006.
Saviola B. Response of Mycobacterial species to an acidic environment. In: Cardona PJ, editor. Understanding tuberculosis – deciphering the secret life of the bacilli. InTech; 2012. pp. 89–102.
Sabatini S, Kurtzman NA. Bicarbonate therapy in severe metabolic acidosis. J Am Soc Nephrol 2009; 20:692–695.
Abdalla D, El Badrawy M, AbouElela M, Abou El-khier N, Abdelgawad T, Moawad A, et al. Effect of bronchoalveolar lavage with sodium bicarbonate on lower respiratory tract pathogens. Chest 2016; 149:4S.
Treatment of Tuberculosis Guidelines. Fourth edition. World Health Organization; 2010. Available at: http://apps.who.int/iris/bitstream/10665/44165/1/9789241547833_eng.pdf?ua=1&ua=1. [Accessed 22 November 2016].
Lohmann EM, Koster BF, le Cessie S, Kamst-van Agterveld MP, van Soolingen D, Arend SM. Grading of a positive sputum smear and the risk of Mycobacterium tuberculosis transmission. Int J Tuberc Lung Dis 2012; 16:1477–1484.
National Tuberculosis Association of the USA. Diagnostic standards and classification of tuberculosis. New York, NY: National Tuberculosis Association; 1961.
Ng AW, Bidani A, Heming TA. Innate host defense of the lung: effects of lung-lining fluid pH. Lung 2004; 182:297–317.
Bodem CR, Lampton LM, Miller DP, Tarka EF, Everett ED. Endobronchial pH. Relevance of aminoglycoside activity in Gram-negative bacillary pneumonia. Am Rev Respir Dis 1983; 127:39–41.
Dalhoff A, Schubert S, Ullmann U. Effect of pH on the in vitro activity of and propensity for emergence of resistance to fluoroquinolones, macrolides, and a ketolide. Infection 2005; 33:36–43.
Bidani A, Heming TA. Effects of bafilomycin A1 on functional capabilities of LPS-activated alveolar macrophages. J Leukoc Biol 1995; 57: 275–281.
Bidani A, Reisner BS, Haque AK, Wen J, Helmer RE, Tuazon DM, et al. Bactericidal activity of alveolar macrophages is suppressed by V-ATPase inhibition. Lung 2000; 178:91–104.
Centers for Disease Control and Prevention. Guidelines for preventing the transmission of Mycobacterium tuberculosis in healthcare settings. MMWR Recomm Rep 2005; 54:1–141.
Agarwal SP, Chauhan LS. Tuberculosis Control In India. Directorate General of Health Services, Ministry of Health and Family Welfare. New Delhi, India: Government of India; 2005.
Frieden T, editor. Toman’s tuberculosis case detection, treatment, and monitoring – questions and answers. 2nd edition. Geneva, Switzerland: World Health Organisation 2004.
Parikh R, Nataraj G, Kanade S, Khatri V, Mehta P. Time to sputum conversion in smear positive pulmonary TB patients on category I DOTS and factors delaying it. J Assoc Physicians India 2012; 60:22–26.
Ngamtrakulpanit L, Yu Y, Adjei A, Amoah G, Gaston B, Hunt J. Identification of intrinsic airway acidification in pulmonary tuberculosis. Glob J Health Sci 2010; 2:106–110.
Treatment of Tuberculosis Disease. Available at: http://www.cdc.gov/TB/education/corecurr/pdf/chapter6.pdf. [Accessed 22 November 2016].
Hess D, Fisher D, Williams P, Pooler S, Kacmarek RM. Medication nebulizer performance. Effects of diluent volume, nebulizer flow, and nebulizer brand. Chest 1996; 110:498–505.
Iivanainen E, Martikainen PJ, Vaananen P, Katila ML. Environmental factors affecting the occurrence of myco-bacteria in brook sediments. J Appl Microbiol 1999; 86:673–681.
Parashar D, Kabra SK, Lodha R, Singh V, Mukherjee A, Arya T, et al. Does neutralization of gastric aspirates from children with suspected intrathoracic tuberculosis affect mycobacterial yields on MGIT culture? J Clin Microbiol 2013; 51:1753–1756.
MacMicking JD, Taylor GA, McKinney GD. Immune control of tuberculosis by IFN-gamma-inducible LRG-47. Science 2003; 302:654–659.
Schaible UE, Sturgill-Koszycki S, Schlesinger PH, Russell DG. Cytokine activation leads to acidification and increases maturation of Mycobacterium avium-containing phagosomes in murine macrophages. J Immunol 1998; 160:1290–1296.
Via LE, Fratti RA, McFalone M, Pagan-Ramos E, Deretic D, Deretic V. Effects of cytokines on mycobacterial phagosome maturation. J Cell Sci 1998; 111:897–905.
Vandal OH, Nathan CF, Ehrt S. Acid resistance in Mycobacterium tuberculosis. J Bacteriol 2009; 191:4714–4721.
Jackett PS, Aber VR, Lowrie DB. Virulence and resistance to superoxide, low pH and hydrogen peroxide among strains of Mycobacterium tuberculosis. J Gen Microbiol 1978; 104:37–45.
Vandal OH, Pierini LM, Schnappinger D. A membrane protein preserves intrabacterial pH in intraphagosomal Mycobacterium tuberculosis. Nat Med 2008; 14:849–854.
Armstrong JA, Hart PD. Phagosome-lysosome interactions in cultured macrophages infected with virulent tubercle bacilli. Reversal of the usual nonfusion pattern and observations on bacterial survival. J Exp Med 1975; 142:1–16.
Gomes MS, Paul S, Moreira AL, Appelberg R, Rabinovitch M, Kaplan G. Survival of Mycobacterium avium and Mycobacterium tuberculosis in acidified vacuoles of murine macrophages. Infect Immun 1999; 67:3199–3206.
Metchnikoff E. Immunity to infective disease. Cambridge, UK: Cambridge University Press 1905. p. 182.
Hoffmann C, Leis A, Niederweis M, Plitzko JM, Engelhardt H. Disclosure of the mycobacterial outer membrane: cryo-electron tomography and vitreous sections reveal the lipid bilayer structure. Proc Natl Acad Sci USA 2008; 105:3963–3967.
Zuber B, Chami M, Houssin C, Dubochet J, Griffiths G, Daffé M. Direct visualization of the outer membrane of mycobacteria and corynebacteria in their native state. J Bacteriol 2008; 190:5672–5680.
Mesquita ED, Gil-Santana L, Ramalho D, Tonomura E, Silva EC, Oliveira MM, et al. Associations between systemic inflammation, mycobacterial loads in sputum and radiological improvement after treatment initiation in pulmonary TB patients from Brazil: a prospective cohort study. BMC Infect Dis 2016; 16:368.
Rubin BK. Mucolytics, expectorants, and mucokinetic medications. Respir Care 2007; 52:859–865.
Author information
Authors and Affiliations
Corresponding author
Additional information
This is an open access journal, and articles are distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as appropriate credit is given and the new creations are licensed under the identical terms.
Rights and permissions
This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
El-Badrawy, M.K., Arram, E.O., Abdalla, D.A. et al. Effect of adding inhalation of sodium bicarbonate 8.4% to the usual treatment on smear-positive pulmonary tuberculosis: a prospective controlled study. Egypt J Bronchol 13, 531–538 (2019). https://doi.org/10.4103/ejb.ejb_18_19
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.4103/ejb.ejb_18_19