Abstract
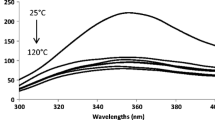
The thermal stability (60°C, 80°C, 100°C), antioxidant activity, and ultraviolet C light (UV-C) stability of standard polyphenols solutions (catechin, gallic acid, and vanillic acid) and of vegetal extracts from spruce bark and grape seeds were investigated. Exposure of the standard solutions and vegetal extracts to high temperatures revealed that phenolic compounds were also relatively stable (degradations ranged from 15 % to 30 % after 4 h of exposure). The highest antioxidant activity was obtained for ascorbic acid and gallic acid followed by catechin and caffeic acid and the grape seeds. The results show that, after 3 h of UV-C exposure, approximately 40 % of vanillic acid, 50 % of gallic acid, and 83 % of catechin were removed. Similar degradation rates were observed for vegetal extracts, with the exception of the degradation of catechin (40 %) from grape seeds. In addition, the photo-oxidation of polyphenols in the presence of food constituents such as citric acid, ascorbic acid, sodium chloride, and sodium nitrate was assessed.
Similar content being viewed by others
References
Almela, L., Sánchez-Muñoz, B., Fernández-López, J. A., Roca, M. J., & Rabe, V. (2006). Liquid chromatograpic-mass spectrometric analysis of phenolics and free radical scavenging activity of rosemary extract from different raw material. Journal of Chromatography A, 1120, 221–229. DOI: 10.1016/j.chroma.2006.02.056.
Bąkowska, A., Kucharska, Z. A., & Oszmiański, J. (2003). The effects of heating, UV irradiation, and storage on stability of the anthocyanin-polyphenol copigment complex. Food Chemistry, 81, 349–355. DOI: 10.1016/s0308-8146(02)00429-6.
Benitez, F. J., Real, F. J., Acero, J. L., Leal, A. I., & Garcia, C. (2005). Gallic acid degradation in aqueous solutions by UV/H2O2 treatment, Fenton’s reagent and the photo-Fenton system. Journal of Hazardous Materials, 126, 31–39. DOI: 10.1016/j.jhazmat.2005.04.040.
Castañeda-Ovando, A., Pacheco-Hernández, M. L., Páez-Hernández, M. E., Rodríguez, J. A., & Galán-Vidal, C. A. (2009). Chemical studies of anthocyanins: A review. Food Chemistry, 113, 859–871. DOI: 10.1016/j.foodchem.2008.09.001.
Díaz-García, M. C., Obón, J. M., Castellar, M. R., Collado, J., & Alacid, M. (2013). Quantification by UHPLC of total individual polyphenols in fruit juices. Food Chemistry, 138, 938–949. DOI: 10.1016/j.foodchem.2012.11.061.
Fischer, U. A., Carle, R., & Kammerer, D. R. (2013). Thermal stability of anthocyanins and colourless phenolics in pomegranate (Punica granatum L.) juices and model solutions. Food Chemistry, 138, 1800–1809. DOI: 10.1016/j.foodchem.2012.10.072.
Giusti, M. M., & Wrolstad, R. E. (2003). Acylated anthocyanins from edible sources and their applications in food systems. Biochemical Engineering Journal, 14, 217–225. DOI: 10.1016/s1369-703x(02)00221-8.
Hainal, A. R., Ignat, I., Volf, I., & Popa, V. I. (2011). Transformation of polyphenols from biomass by some yeast species. Cellulose Chemistry and Technology, 45, 211–219.
Ignat, I., Volf, I., & Popa, V. I. (2011a). A critical review of methods for characterization of polyphenolic compounds in fruits and vegetables. Food Chemistry, 126, 1821–1835. DOI: 10.1016/j.foodchem.2010.12.026.
Ignat, I., Stingu, A., Volf, I., & Popa, V. I. (2011b). Characterization of grape seeds aqueous extract and possible application in biological systems. Cellulose Chemistry and Technology, 45, 205–209.
Karou, D., Dicko, M. H., Simpore, J., & Traore, A. S. (2005). Antioxidant and antibacterial activities of Polyphenols from ethnomedicinal plants of Burkina Faso. African Journal of Biotechnology, 4, 823–828.
Kırca, A., & Arslan, E. (2008). Antioxidant capacity and total phenolic content of selected plants from Turkey. International Journal of Food Science and Technology, 43, 2038–2046. DOI: 10.1111/j.1365-2621.2008.01818.x.
Koutchma, T. (2008). UV-light for processing foods. Ozone: Science and Engineering: The Journal of the International Ozone Association, 30, 93–98. DOI: 10.1080/01919510701816346.
Koutchma, T. (2009). Advances in ultraviolet light technology for non-thermal processing of liquid foods. Food Bioprocess Technology, 2, 138–155. DOI: 10.1007/s11947-008-0178-3.
Mack, J., & Bolton, J. R. (1999). Photochemistry of nitrite and nitrate in aqueous solution: a review. Journal of Photochemistry and Photobiology A: Chemistry, 128, 1–13. DOI: 10.1016/s1010-6030(99)00155-0.
Montoro, P., Tuberoso, C. I. G., Piacente, S., Perrone, A., De Feo, V., Cabras, P., & Pizza, C. (2006). Stability and antioxidant activity of polyphenols in extracts of Myrtus communis L. berries used for the preparation of myrtle liqueur. Journal of Pharmaceutical and Biomedical Analysis, 41, 1614–1619. DOI: 10.1016/j.jpba.2006.02.018.
Muanda, F. N., Soulimani, R., Diop, B., & Dicko, A. (2011). Study on chemical composition and biological activities of essential oil and extracts from Stevia rebaudiana Bertoni leaves. LWT — Food Science and Technology, 44, 1865–1872. DOI: 10.1016/j.lwt.2010.12.002.
Naczk, M., & Shahidi, F. (2006). Phenolics in cereals, fruits and vegetables: Occurrence, extraction and analysis. Journal of Pharmaceutical and Biomedical Analysis, 41, 1523–1542. DOI: 10.1016/j.jpba.2006.04.002.
Neamtu, M., & Frimmel, F. H. (2006). Degradation of endocrine disrupting bisphenol A by 254 nm irradiation in different water matrices and effect on yeast cells. Water Research, 40, 3745–3750. DOI: 10.1016/j.watres.2006.08.019.
Neo, Y. P., Ariffin, A., Tan, C. P., & Tan, Y. A. (2008). Determination of oil palm fruit phenolic compounds and their antioxidant activities using spectrophotometric methods. International Journal of Food Science & Technology, 43, 1832–1837. DOI: 10.1111/j.1365-2621.2008.01717.x.
Oppenlaender, T. (2003). Photochemical purification of water and air. New York, NY, USA: Wiley-VCH.
Parr, A. J., & Bolwell, G. P. (2000). Phenols in plant and in man. The potential for possible nutritional enhancement of the diet by modifying the phenols content or profile. Journal of the Science of Food and Agriculture, 80, 985–1012. DOI: 10.1002/(sici)1097-0010(20000515)80:7〈985::aidjsfa572〉3.0.co;2-7.
Sajiki, J., & Yonekubo, J. (2003). Leaching of bisphenol A (BPA) to seawater from polycarbonate plastic and its degradation by reactive oxygen species. Chemosphere, 51, 55–62. DOI: 10.1016/s0045-6535(02)00789-0.
Sarikurkcu, C., Tepe, B., Daferera, D., Polissiou, M., & Harmandar, M. (2008). Studies on the antioxidant activity of the essential oil and methanol extract of Marrubium globosum subsp. globosum (lamiaceae) by three different chemical assays. Bioresource Technology, 99, 4239–4246. DOI: 10.1016/j.biortech.2007.08.058.
Schindelin, A. J., & Frimmel, F. H. (2000). Nitrate and natural organic matter in aqueous solutions irradiated by simulated sunlight. Environmental Science and Pollution Research, 7, 205–210. DOI: 10.1007/bf02987349.
Tikekar, R. V., Anantheswaran, R. C., & LaBorde, L. F. (2011a). Ascorbic acid degradation in a model apple juice system and in apple juice during ultraviolet processing and storage. Journal of Food Science, 76, H62–H71. DOI: 10.1111/j.1750-3841.2010.02015.x.
Tikekar, R. V., Anantheswaran, R. C., Elias, R. J., & LaBorde, L. F. (2011b). Ultraviolet-induced oxidation of ascorbic acid in a model juice system: Identification of degradation products. Journal of Agricultural and Food Chemistry, 59, 8244–8248. DOI: 10.1021/jf201000x.
Tikekar, R. V., Anantheswaran, R. C., & LaBorde, L. F. (2012). Patulin degradation in a model apple juice system and in apple juice during ultraviolet processing. Journal of Food Processing and Preservation. DOI: 10.1111/jfpp.12047. (in press)
Food and Drug Administration (2000). Irradiation in the production, processing and handling of food. Federal Register, 65, 71056–71058.
Warneck, P., & Wurzinger, C. (1988). Product quantum yields for the 305-nm photodecomposition of nitrate in aqueous solution. Journal of Physical Chemistry, 92, 6278–6283. DOI: 10.1021/j100333a022.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Volf, I., Ignat, I., Neamtu, M. et al. Thermal stability, antioxidant activity, and photo-oxidation of natural polyphenols. Chem. Pap. 68, 121–129 (2014). https://doi.org/10.2478/s11696-013-0417-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.2478/s11696-013-0417-6