Abstract
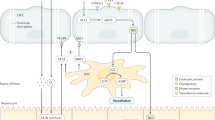
The fenestrated sinusoidal endothelium (‘liver sieve’) and space of Disse in the healthy liver do not impede the transfer of most substrates, including drugs and oxygen, from the sinusoidal lumen to the hepatocyte. Plasma components transfer freely in both directions through the endothelial fenestrations and into the space of Disse. The endothelium is attenuated, there is no basement membrane and there is minimum collagen in the space of Disse, thus minimising any barriers to substrate diffusion.
Both cirrhosis and aging are associated with marked structural changes in the sinusoidal endothelium and space of Disse that are likely to influence bulk plasma transfer into the space of Disse, and diffusion through the endothelium and space of Disse. These changes, termed capillarisation and pseudocapillarisation in cirrhosis and aging, respectively, impede the transfer of various substrates. Capillarisation is associated with exclusion of albumin, protein-bound drugs and macromolecules from the space of Disse, and the progressive transformation of flow-limited to barrier-limited distribution of some substrates.
There is evidence that the sinusoidal changes in cirrhosis and aging contribute to hepatocyte hypoxia, thus providing a mechanism for the apparent differential reduction of oxygen-dependent phase I metabolic pathways in these conditions. Structural change and subsequent dysfunction of the liver sieve warrant consideration as a significant factor in the impairment of overall substrate handling and hepatic drug metabolism in cirrhosis and aging.





Similar content being viewed by others
References
Roberts MS, Rowland M. Correlation between in-vitro microsomal enzyme activity and whole organ hepatic elimination kinetics: analysis with a dispersion model. J Pharm Pharmacol 1986; 38: 177–81
Branch RA, Shand DG. Propranolol disposition in chronic liver disease: a physiological approach. Clin Pharmacokinet 1976; 1: 264–79
McLean AJ, Morgan DJ. Clinical pharmacokinetics in patients with liver disease. Clin Pharmacokinet 1991; 21: 42–69
Pang KS, Rowland M. Hepatic clearance of drugs. I: theoretical considerations of a ‘well-stirred’ model and a ‘parallel tube’ model: influence of hepatic blood flow, plasma and blood cell binding, and the hepatocellular enzymatic activity on hepatic drug clearance. J Pharmacokinet Biopharm 1977; 5: 625–53
Goresky CA. The modeling of tracer exchange and sequestration in the liver. Fed Proc 1984; 43: 154–60
Goresky CA, Pang KS, Schwab AJ, et al. Uptake of a protein-bound polar compound, acetaminophen sulfate, by perfused rat liver. Hepatology 1992; 16: 173–90
Roberts MS, Rowland M. A dispersion model of hepatic elimination. I: formulation of the model and bolus considerations. J Pharmacokinet Biopharm 1986; 14: 227–60
Mellick GD, Roberts MS. Structure-hepatic disposition relationships for phenolic compounds. Toxicol Appl Pharmacol 1999; 158: 50–60
Wood AJ, Villeneuve JP, Branch RA, et al. Intact hepatocyte theory of impaired drug metabolism in experimental cirrhosis in the rat. Gastroenterology 1979; 76: 1358–62
Morgan DJ, McLean AJ. Clinical pharmacokinetic and pharmacodynamic considerations in patients with liver disease: an update. Clin Pharmacokinet 1995; 29: 1–22
Le Couteur DG, McLean AJ. The aging liver: drug clearance and an oxygen diffusion barrier hypothesis. Clin Pharmacokinet 1998; 34: 359–73
Schaffner R, Popper H. Capillarisation of hepatic sinusoids in man. Gastroenterology 1963; 44: 239–42
Fraser R, Dobbs BR, Rogers GW. Lipoproteins and the liver sieve: the role of fenestrated sinusoidal endothelium in lipoprotein metabolism, atherosclerosis, and cirrhosis. Hepatology 1995; 21: 863–74
Mori T, Okanoue T, Sawa Y, et al. Defenestration of the sinusoidal endothelial cell in a rat model of cirrhosis. Hepatology 1993; 17: 891–7
Le Couteur DG, Cogger VC, Markus AM, et al. Pseudocapillarization and associated energy limitation in the aged rat liver. Hepatology 2001; 33: 537–43
McLean AJ, Cogger VC, Chong GC, et al. Age-related pseudocapillarisation of the human liver. J Pathol 2003; 200: 112–7
Wisse W, De Zanger RB, Charels K, et al. The liver sieve: considerations concerning the structure and function of endothelial fenestrae, the sinusoidal wall and the space of Disse. Hepatology 1985; 5: 683–92
Huet PM, Goresky CA, Villeneuve JP, et al. Assessment of liver microcirculation in human cirrhosis. J Clin Invest 1982; 70: 1234–44
Varin F, Huet PM. Hepatic microcirculation in the perfused cirrhotic rat liver. J Clin Invest 1985; 76: 1904–12
Reichen J. The role of the sinusoidal endothelium in liver function. News Physiol Sci 1999; 14: 117–21
Hung DY, Chang P, Cheung K, et al. Quantitative evaluation of altered hepatic spaces and membrane transport in fibrotic rat liver. Hepatology 2002; 36: 1180–9
Braet F, Wisse E. Structural and functional aspects of liver sinusoidal endothelial cell fenestrae: a review. BMC Compar Hepatol 2002; 1: 1–17
Bendayan M. Vascular permeability in blood capillary. Microsc Res Tech 2002; 57: 263–8
Fraser R, Bosanquet AG, Day WA. Filtration of chylomicrons by the liver may influence cholesterol metabolism and atherosclerosis. Atherosclerosis 1978; 29: 113–23
Popescu D, Movileanu L, Ion S, et al. Hydrodynamic effects on the solute transport across endothelial pores and hepatocyte membranes. Phys Med Biol 2000; 45: N157–65
Heath T, Lowden S. Pathways of interstitial fluid and lymph flow in the liver acinus of the sheep and mouse. J Anat 1998; 192: 351–8
Goresky CA, Bach GC, Nadeau BE. On the uptake of materials by the intact liver: the transport and net removal of galactose. J Clin Invest 1973; 52: 991–1009
Goresky CA, Nadeau BA. Uptake of materials by the intact liver: the exchange of glucose across the cell membranes. J Clin Invest 1974; 53: 634–46
Pang KS, Schwab AJ, Goresky CA, et al. Transport, binding, and metabolism of sulfate conjugates in the liver. Chem Biol Interact 1994; 92: 179–207
Goresky CA. A linear method for determining liver sinusoidal and extravascular volumes. Am J Physiol 1963; 204: 626–40
Yu K, Cooper AD. Postprandial lipoproteins and atherosclerosis. Frontiers Biosci 2001; 6: 332–54
Romero EL, Morilla MJ, Regts J, et al. On the mechanism of hepatic transendothelial passage of large liposomes. FEBS Lett 1999; 448: 193–6
Floren CH. Binding of apolipoprotein E-rich remnant lipoproteins to human liver membranes. Scand J Gastroenterol 1984; 19: 473–9
Hardonk MJ, Harms G, Koudstaal J. Zonal heterogeneity of rat hepatocytes in the in vivo uptake of 17nm colloidal gold granules. Histochemistry 1985; 83: 473–7
Huet PM, Villeneuve JP, Pomier-Layrargues G, et al. Hepatic circulation in cirrhosis. Clin Gastroenterol 1985; 14: 155–68
Okanoue T, Mori T, Sakamoto S, et al. Role of sinusoidal endothelial cells in liver disease. J Gastroenterol Hepatol 1995; 10: S35–7
Horn T, Christoffersen P, Henriksen JH. Alcoholic liver injury: defenestration in noncirrhotic livers: a scanning electron microscopic study. Hepatology 1987; 7: 77–82
Hirooka N, Iwasaki I, Horie H, et al. Hepatic microcirculation of liver cirrhosis studied by corrosion cast/scanning electron microscope examination. Acta Pathol Jpn 1986; 36: 375–87
Clark SA, Cook HB, Oxner RB, et al. Defenestration of hepatic sinusoids as a cause of hyperlipoproteinaemia in alcoholics. Lancet 1988; II: 1225–7
Cogger VC, Warren A, Fraser R, et al. Hepatic sinusoidal pseudocapillarization with aging in the non-human primate. Exp Gerontol 2003; 38: 1101–7
Popper H. Aging and the liver. Prog Liver Dis 1986; 8: 659–83
Villeneuve JP, Dagenais M, Huet PM, et al. The hepatic microcirculation in the isolated perfused human liver. Hepatology 1996; 23: 24–31
Miki K, Kubota K, Inoue Y, et al. Receptor measurements via Tc-GSA kinetic modeling are proportional to functional hepatocellular mass. J Nucl Med 2001; 42: 733–7
Wakabayashi H, Nishiyama Y, Ushiyama T, et al. Evaluation of the effect of age on functioning hepatocyte mass and liver blood flow using liver scintigraphy in preoperative estimations for surgical patients: comparison with CT volumetry. J Surg Res 2002; 106: 246–53
Rippe B, Rosengren BI, Carlsson O, et al. Transendothelial transport: the vesicle controversy. J Vasc Res 2002; 39: 375–90
Bodega F, Zocchi L, Agostoni E. Macromolecule transfer through mesothelium and connective tissue. J Appl Physiol 2000; 89: 2165–73
Fenvyes D, Gariepy L, Villeneuve JP. Clearance by the liver in cirrhosis. I: relationship between propranolol metabolism in vitro and its extraction by the perfused liver in the rat. Hepatology 1993; 17: 301–6
Gariepy L, Fenyves D, Kassissia I, et al. Clearance by the liver in cirrhosis. II: characterization of propranolol uptake with the multiple-indicator dilution technique. Hepatology 1993; 18: 823–31
Molino G, Avagnina P, Belforte G, et al. Assessment of the hepatic circulation in humans: new concepts based on evidence derived from a D-sorbitol clearance method. J Lab Clin Med 1998; 131: 393–405
Ott P, Clemmesen O, Keiding S. Interpretation of simultaneous measurements of hepatic extraction fractions of indocyanine green and sorbitol: evidence of hepatic shunts and capillarization? Dig Dis Sci 2000; 45: 359–65
Hung DY, Chang P, Cheung K, et al. Cationic drug pharmacokinetics in diseased livers determined by fibrosis index, hepatic protein content, microsomal activity, and nature of drug. J Pharmacol Exp Ther 2002; 301: 1079–87
Materne R, Annet L, Dechambre S, et al. Dynamic computed tomography with low- and high-molecular-mass contrast agents to assess microvascular permeability modifications in a model of liver fibrosis. Clin Sci (Lond) 2002; 103: 213–6
Mastai R, Laganiere S, Wanless IR, et al. Hepatic sinusoidal fibrosis induced by cholesterol and stilbestrol in the rabbit. II: hemodynamic and drug disposition studies. Hepatology 1996; 24: 865–70
Fraser R, Courtice FC. The transport of cholesterol in thoracic duct lymph of animals fed cholesterol with varying trigylceride loads. Aust J Exp Biol Med Sci 1969; 47: 723–32
Fraser R, Cliff WJ, Courtice FC. The effect of dietary fat load on the size and composition of chylomicrons in thoracic duct lymph. Q J Exp Physiol Cogn Med Sci 1968; 53: 390–8
Fraser R, Clark SA, Day WA, et al. Nicotine decreases the porosity of the rat liver sieve: a possible mechanism for hypercholesterolaemia. Br J Exp Pathol 1988; 69: 345–50
Fraser R, Bowler LM, Day WA, et al. High perfusion pressure damages the sieving ability of sinusoidal endothelium in rat livers. Br J Exp Pathol 1980; 61: 222–8
Le Couteur DG, Fraser R, Cogger VC, et al. Hepatic pseudocapillarisation and atherosclerosis in ageing. Lancet 2002; 359: 1612–5
McLean AJ, Le Couteur DG. The effect of age on hepatocyte uptake of solutes [abstract]. Clin Pharmacol Ther 1998; 63: 223
Le Couteur DG, Rivory LP, Yi C, et al. Aging, acute oxidative injury and hepatocellular glucose transport in the rat. Int Hepatol Commun 1995; 3: 244–53
Kroker R, Hegner D, Anwer MS. Altered hepatobiliary transport of taurocholic acid in aged rats. Mech Ageing Dev 1980; 12: 367–73
Handler JA, Genell CA, Goldstein RS. Hepatobiliary function in senescent male Sprague-Dawley rats. Hepatology 1994; 19: 1496–503
Kassissia I, Rose CP, Goresky CA, et al. Flow-limited tracer oxygen distribution in the isolated perfused rat liver: effects of temperature and hematocrit. Hepatology 1992; 16: 763–75
Le Couteur DG, Yin ZL, Rivory LP, et al. Carbon monoxide disposition in the perfused rat liver. Am J Physiol 1999; 277: G725–30
Jones DP. Hypoxia and drug metabolism. Biochem Pharmacol 1981; 30: 1019–23
Hickey PL, McLean AJ, Angus PW, et al. Increased sensitivity of propranolol clearance to reduced oxygen delivery in the isolated perfused cirrhotic rat liver. Gastroenterology 1996; 111: 1039–48
Rodighiero V. Effects of liver disease on pharmacokinetics: an update. Clin Pharmacokinet 1999; 37: 399–431
Levy M, Caraco Y, Geisslinger G. Drug acetylation in liver disease. Clin Pharmacokinet 1998; 34: 219–326
Morgan DJ, McLean AJ. Therapeutic implications of impaired oxygen diffusion in chronic liver disease. Hepatology 1991; 14: 1280–2
Choo EF, Angus PW, Morgan DJ. Effect of cirrhosis on sulphation by the isolated perfused rat liver. J Hepatol 1999; 30: 498–502
Froomes PR, Morgan DJ, Smallwood RA, et al. Comparative effects of oxygen supplementation on theophylline and acetaminophen clearance in human cirrhosis. Gastroenterology 1999; 116: 915–20
Le Couteur DG, Hickey H, Harvey P, et al. Hepatic artery flow and propranolol metabolism in the perfused cirrhotic rat liver. J Pharmacol Exp Ther 1999; 289: 1553–8
Harvey PJ, Gready JE, Hickey HM, et al. 31p and 1H NMR spectroscopic studies of liver extracts of carbon tetrachloridetreated rats. NMR Biomed 1999; 12: 395–401
Harvey PJ, Gready JE, Yin ZL, et al. Acute oxygen supplementation restores markers of hepatocyte energy status and hypoxia in cirrhotic rats. J Pharmacol Exp Ther 2000; 293: 52–5
Hickey PL, Angus PW, McLean AJ, et al. Oxygen supplementation restores theophylline clearance to normal in cirrhotic rats. Gastroenterology 1995; 108: 1504–9
Rose CP, Goresky CA. Limitation of tracer oxygen uptake in the canine coronary circulation. Circ Res 1985; 56: 57–71
Cho CS, McLean AJ, Rivory LP, et al. Carbon monoxide wash-in method to determine gas transfer in vascular beds: application to rat hind limb. Am J Physiol 2001; 280: H1802–6
Sangalli MR, McLean AJ, Peek MJ, et al. Carbon monoxide disposition and permeability-surface area product in the fetal circulation of the perfused term human placenta. Placenta 2003; 24: 8–11
Greenblatt DJ, Harmatz JS, Shader RI. Clinical pharmacokinetics of anxiolytics and hypnotics in the elderly: therapeutic considerations (Pt I). Clin Pharmacokinet 1991; 21: 165–77
Schmucker DL. Liver function and phase I drug metabolism in the elderly: a paradox. Drugs Aging 2001; 18: 837–51
Sotaneimi EA, Arranto AJ, Pelkonen O, et al. Age and cytochrome P450-linked drug metabolism in humans: an analysis of 226 subjects with equal histopathological conditions. Clin Pharmacol Ther 1997; 61: 331–9
Woodhouse KW, Wynne HA. Age-related changes in liver size and hepatic blood flow: the influence on drug metabolism in the elderly. Clin Pharmacokinet 1988; 15: 287–94
Le Couteur DG, Rivory LP, Pond SM. The effects of aging and nutritional state on hypoxia-reoxygenation injury in the perfused rat liver. Transplantation 1994; 58: 531–6
Le Couteur DG, Rivory LP, Roberts MS, et al. Aging and the response of the isolated perfused rat liver to vasoactive drugs. Biochem Pharmacol 1992; 43: 913–5
Richardson DI, Withrington PG. Liver blood flow. II: effects of drugs and hormones on liver blood flow. Gastroenterology 1981; 81: 356–75
Le Couteur DG, Muller M, Yang MC, et al. Age-environment and gene-environment interactions in the pathogenesis of Parkinson’s disease. Rev Environ Health 2002; 17: 51–65
Acknowledgements
We acknowledge funding from the National Health and Medical Research Council, Department of Veterans Affairs, and the Ageing and Alzheimer’s Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Le Couteur, D.G., Fraser, R., Kilmer, S. et al. The Hepatic Sinusoid in Aging and Cirrhosis. Clin Pharmacokinet 44, 187–200 (2005). https://doi.org/10.2165/00003088-200544020-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003088-200544020-00004