Abstract
Asthma and chronic obstructive pulmonary disease (COPD) are common disorders that are associated with increasing morbidity and mortality in older people. Bronchodilators are used widely in patients with these conditions, but even when used in inhaled form can have systemic as well as local effects. Older people experience more adverse drug effects because of pharmacodynamic and pharmacokinetic changes and particularly drug-drug and drug-disease interactions.
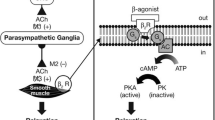
Cardiovascular disease is common in older people and β-adrenoceptor agonists (β-agonists) have inotropic and chronotropic effects that can increase arrhythmias and cardiomyopathy. They can also worsen or induce myocardial ischaemia and cause electrolyte disturbances that contribute to arrhythmias. Tremor is a well known distressing adverse effect of β-agonist administration. Long-term β-agonist use can be associated with tolerance, poor disease control, sudden life-threatening exacerbations and asthma-related deaths. Functional β2-adrenoceptors are present in osteoblasts, and chronic use of β-agonists has been implicated in osteoporosis.
Inhaled anticholinergics are usually well tolerated but may cause dry mouth, which can be troublesome in older people. Pupillary dilatation, blurred vision and acute glaucoma can occur from escape of droplets from loosely fitting nebulizer masks. Although ECG changes have not been seen in randomized controlled trials of long-acting inhaled anticholinergics, supraventricular tachycardias have been observed in a 5-year randomized controlled trial of ipratropium bromide. Paradoxical bronchoconstriction can occur with inhaled anticholinergics as well as with β-agonists, but tolerance has not been reported with anticholinergics. Anticholinergic drugs also cause central effects, most notably impairment of cognitive function, and these effects have been noted with inhaled agents.
Use of theophylline is limited by its adverse effects, which range from commonly occurring gastrointestinal symptoms to palpitations, arrhythmias and reports of myocardial infarction. Seizures have been reported, but are rare. Theophylline is metabolized primarily by the liver, and commonly interacts with other medications. Its concentration in plasma should be monitored closely, especially in older people.
Although many clinical trials have been conducted on bronchodilators in obstructive airways disease, the results of these clinical trials need to be interpreted with caution as older people are often under-represented and subjects with co-morbidities actively excluded from these trials.












Similar content being viewed by others
References
Soriano JB, Kiri VA, Maier WC, et al. Increasing prevalence of asthma in UK primary care during the 1990s. Int J Tuberc Lung Dis 2003 May; 7(5): 415–21
Horsley JR, Sterling IJ, Waters WE, et al. Respiratory symptoms among elderly people in the New Forest area as assessed by postal questionnaire. Age Aging 1991 Sep; 20(5): 325–31
Halbert RJ, Natoli JL, Gano A, et al. Global burden of COPD: systematic review and meta-analysis. Eur Respir J 2006; 28(3): 523–32
Britton M. The burden of COPD in the UK: results from the Confronting COPD survey. Respir Med 2003; 97Suppl. C: S71–9
Hardie JA, Vollmer WM, Buist SA, et al. Respiratory symptoms and obstructive pulmonary disease in a population aged over 70 years. Respir Med 2005 Feb; 99(2): 186–95
Diette GB, Krishnan JA, Dominici F, et al. Asthma in older patients: factors associated with hospitalization. Arch Intern Med 2002 May 27; 162(10): 1123–32
Cydulka RK, McFadden Jr ER, Emerman CL, et al. Patterns of hospitalization in elderly patients with asthma and chronic obstructive pulmonary disease. Am J Respir Crit Care Med 1997; 156(6): 1807–12
Banerji A, Clark S, Afilalo M, et al. Prospective multicenter study of acute asthma in younger versus older adults presenting to the emergency department. J Am Geriatr Soc 2006 Jan; 54(1): 48–55
Carrasco Garrido P, de Miguel Diez JM, Rejas Gutiérrez J, et al. Negative impact of chronic obstructive pulmonary disease on the health-related quality of life of patients: results of the EPIDEPOC study. Health Qual Life Outcomes 2006 May 23; 4: 31
Connolly MJ, Lowe D, Anstey K, et al. on behalf of the British Thoracic Society and the Royal College of Physicians Clinical Effectiveness Evaluation Unit (CEEU). Admissions to hospital with exacerbations of chronic obstructive pulmonary disease: effect of age related factors and service organization. Thorax 2006; 61: 843–8
Roberts CM, Lowe D, Bucknall CE, et al. Clinical audit indicators of outcome following admission to hospital with acute exacerbation of chronic obstructive pulmonary disease. Thorax 2002; 57: 137–41
Seneff MG, Wagner DP, Wagner RP, et al. Hospital and 1-year survival of patients admitted to intensive care units with acute exacerbation of chronic obstructive pulmonary disease. JAMA 1995; 274: 1852–7
Mannino DM, Brown C, Giovino GA. Obstructive lung disease deaths in the United States from 1979 through 1993: an analysis using multiple-cause mortality data. Am J Respir Crit Care Med 1997 Sep; 156(3): 814–8
Wijnhoven HA, Kriegsman DM, Hesselink AE, et al. Determinants of different dimensions of disease severity in asthma and COPD: pulmonary function and health-related quality of life. Chest 2001; 119: 1034–42
Yeo J, Karimova G, Bansal S. Co-morbidity in older patients with COPD: its impact on health service utilization and quality of life, a community study. Age Aging; 35(1): 33–7
Cully JA, Graham DP, Stanley MA, et al. Quality of life in patients with chronic obstructive pulmonary disease and co-morbid anxiety or depression. Psychosomatics 2006 Aug; 47: 312–9
BTS/SIGN guideline for treatment for asthma 2005 [online]. Available from URL: http://www.brit-thoracic.org.uk [Accessed 2008 Mar 13]
National Collaborating Centre for Chronic Conditions. Chronic obstructive pulmonary disease: national clinical guideline for management of chronic obstructive pulmonary disease in adults in primary and secondary care. Thorax 2004; 59Suppl. I: 1–232
Parameswaran K, Hildreth AJ, Chadha D, et al. Asthma in the elderly: underperceived, underdiagnosed and undertreated: a community survey. Respir Med 1998; 92: 573–7
McLean AJ, Le Conteur DG. Aging biology and geriatric clinical pharmacology. Pharmacol Rev 2004; 56: 163–84
Soriano JB, Visick GT, Muellerova H, et al. Patterns of comorbidities in newly diagnosed COPD and asthma in primary care. Chest 2005 Oct; 128(4): 2099–107
McGarvey LP, Matthias J, Anderson J, et al. Ascertainment of cause-specific mortality in COPD: operations of the TORCH Clinical Endpoint Committee. Thorax 2007; 62: 411–5
Curkendall SM, de Luise C, Jones JK, et al. Cardiovascular disease in patients with chronic obstructive pulmonary disease: Saskatchewan Canada cardiovascular disease in COPD patients. Ann Epidemiol 2006 Jan; 16(1): 63–70
National Academy of Aging Society. Challenges for the 21st century: chronic but preventable conditions - heart disease 2000 January [online]. Available from URL: http://www.agingsociety.org/agingsociety/pdf/heart.pdf [Accessed 2008 Mar 13]
Gottdiener JS, McCLelland RL, Marshall R, et al. Outcome of congestive heart failure in elderly persons: influence of left ventricular systolic function. The Cardiovascular Health Study. Ann Intern Med 2002 Oct 15; 137(8): 631–9
Kupari M, Lindroos M, Iivanainen AM, et al. Congestive heart failure in old age: prevalence, mechanisms and 4-year prognosis in the Helsinki Aging Study. J Intern Med 1997 May; 241(5): 387–94
Kitzman DW, Gardin JM, Gottdiener JS, et al. Importance of heart failure with preserved systolic function in patients ≥65 years of age. CHS Research Group. Am J Cardiol 2001 Feb 15; 87(4): 413–9
Rutten FH, Cramer MJM, Grobbee DE, et al. Unrecognized heart failure in elderly patients with stable COPD. Eur Heart J 2005 Sep; 26(18): 1887–94
Jenneck C, Juergens U, Buecheler M, et al. Pathogenesis, diagnosis and treatment of aspirin intolerance. Ann Allergy Asthma Immunol 2007; 99(1): 13–21
Kotlyar E, Keogh AM, Macdonald PS, et al. Tolerability of carvedilol in patients with heart failure and concomitant chronic obstructive pulmonary disease or asthma. J Heart Lung Transplant 2002 Dec; 21(12): 1290–5
Norwood R, Balkissoon R. Current perspectives on management of co-morbid depression in COPD. COPD 2005 Mar; 2(1): 185–93
Yohannes AM, Baldwin RC, Connolly MJ. Depression and anxiety in elderly patients with chronic obstructive pulmonary disease. Age Aging 2006; 35(5): 457–9
Yohannes AM, Connolly MJ, Baldwin RC. A feasibility study of antidepressant drug therapy in depressed elderly patients with chronic obstructive pulmonary disease. Int J Geriatr Psychiatry 2001 May; 16(5): 451–4
Lavoie KL, Bacon SL, Barone S, et al. What is worse for asthma control and quality of life: depressive disorders, anxiety disorders, or both? Chest 2006 Oct; 130(4): 1039–47
Jorgensen NR, Schwarz P, Holme I, et al. The prevalence of osteoporosis in patients with chronic obstructive pulmonary disease: a cross sectional study. Respiration 2007 Jan; 101(1): 177–85
Bolton CE, Ionescu AA, Shiels KM, et al. Associated loss of fat-free mass and bone mineral density in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2004 Dec 15; 170(12): 1286–93
de Vries F, Pouwels S, Bracke M, et al. Use of beta-2 agonists and risk of hip/femur fracture: a population-based case-control study. Pharmacoepidemiol Drug Saf 2007 Jun; 16(6): 612–9
de Vries F, van Staa TP, Bracke M, et al. Severity of obstructive airway disease and risk of osteoporotic fracture. Eur Respir J 2005 May; 25(5): 879–84
Fain SB. Detection of age-dependent changes in healthy lungs with diffusion weighted 3He MRI. Acad Radiol Nov 2005; 12(11): 1385–93
Tager IB, Segal MR, Speizer FE, et al. The natural history of forced expiratory volumes: effect of cigarette smoking and respiratory symptoms. Am Rev Respir Dis 1988 Oct; 138(4): 837–49
Pride NB. Aging and changes in lung mechanics. Eur Respir J 2005; 26: 563–5
Meyer KC, Rosenthal NS, Soergel P, et al. Neutrophils and low-grade inflammation in the seemingly normal aging human lung. Mech Aging Dev 1998; 104: 169–81
Morris JF. Physiological changes due to age: implications for respiratory drug therapy. Drugs Aging 1994 Mar; 4(3): 207–20
Svartengren M, Falk R, Philipson K. Long-term clearance from small airways decreases with age. Eur Respir J 2005; 26: 609–15
Scichilone N, Messina M, Battaglia S, et al. Airway hyper responsiveness in the elderly: prevalence and clinical implications. Eur Respir J 2005; 25: 364–75
O’Riordan T, Faris M. Inhaled antimicrobial therapy. Respir Care Clin N Am 1999 Dec; 5(4): 617–31
Patton JS, Fishburn CS, Weers JG. The lungs as a portal of entry for systemic drug delivery. Proc Am Thorac Soc 2004; 1(4): 338–44
Dolovich M, Labiris R. Imaging drug delivery and drug responses in the lung. Proc Am Thorac Soc 2004; 1: 329–37
Taylor J, Kotch A, Rice K, et al. Ipratropium bromide hydrofluoroalkane inhalation aerosol is safe and effective in patients with COPD. Chest 2001 Oct; 120(4): 1253–61
Appleton S, Jones T, Poole P, et al. Ipratropium bromide vs short acting β-2 agonists for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2006; (2): CD001387
Colice GL. Nebulized bronchodilators for outpatient management of stable chronic obstructive pulmonary disease. Am J Med 1996; 100Suppl. 1A: 11–7S
Friedman M. A multicenter study of nebulized bronchodilator solutions in chronic obstructive pulmonary disease. Am J Med 1996; 100Suppl. 1A: 30–9s
Rennard SI, Serby CW, Ghafouri M, et al. Extended therapy with ipratropium is associated with improved lung function in patients with COPD: a retrospective analysis of data from seven clinical trials. Chest 1996; 110: 62–70
COMBIVENT Inhalation Aerosol Study Group. Chronic obstructive pulmonary disease: a combination of ipratropium and albuterol is more effective than either agent alone. Chest 1994; 105(5): 1411–9
COMBIVENT Inhalation Study Group. Routine nebulized ipratropium and albuterol together are better than either alone in COPD. Chest 1997; 112(6): 1514–21
Gross N, Tashkin D, Miller R, et al. Inhalation by nebulization of albuterol-ipratropium combination (Dey Combination) is superior to either agent alone in the treatment of chronic obstructive pulmonary disease. Respiration 1998; 65: 354–62
Levin DC, Little KS, Laughlin KR, et al. Addition of anticholinergic solution prolongs bronchodilator effect of beta2 agonists in patients with chronic obstructive pulmonary disease. Am J Med 1996; 100Suppl. 1A: 40s–8s
Tashkin DP, Bleeker ER, Braun S, et al. Results of a multicenter study of nebulized inhalant bronchodilator solutions. Am J Med 1996 Jan 29; 100Suppl. 1A: 62S–9S
Appleton S, Jones T, Poole P, et al. Ipratropium bromide vs long acting β-2 agonists for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2006; 3: CD006101
Dahl R, Greefhorst LA, Nowak D, et al. Inhaled formoterol dry powder versus ipratropium bromide in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001; 164: 778–84
Stahl E, Wadbo M, Bengtsson T, et al. Health-related quality of life, symptoms, exercise capacity and lung function during treatment for moderate to severe COPD. J Drug Assess 2002; 5: 81–94
Wadbo M, Lofdahl CG, Larsson K, et al. Effects of formoterol and ipratropium bromide in COPD: a 3-month placebo-controlled study. Eur Respir J 2002; 20(5): 1138–46
Rennard S, Anderson W, ZuWallack R, et al. Use of a long-acting inhaled beta-2 adrenergic agonist, salmeterol xinafoate, in patients with chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001; 163(5): 1087–92
Appleton S, Poole P, Smith B, et al. Long acting β-2 agonists for poorly reversible chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2006; 3: CD001104
Mahler DA, Donohue JF, Barbee RA, et al. Efficacy of salmeterol xinafoate in the treatment of COPD. Chest 1999; 115: 957–65
Mahler DA, Wire P, Horstman D, et al. Effectiveness of fluticasone propionate and salmeterol combination delivered via the Diskus device in the treatment of chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2002; 166: 1084–91
van Noord JA, de Munck DR, Bantje TA, et al. Long-term treatment of chronic obstructive pulmonary disease with salmeterol and the additive effect of ipratropium. Eur Respir J 2000; 15: 878–85
Boyd G, Morice AH, Pounsford JC, et al. An evaluation of salmeterol in the treatment of chronic obstructive pulmonary disease (COPD). Eur Respir J 1997; 10(4): 815–21
Chapman K, Arvidsson P, Chuchalin A, et al. The addition of salmeterol 50(µg bid to anticholinergic treatment in patients with COPD: a randomised placebo controlled trial. Can Resp J 2002; 9(3): 178–85
Chapman K, James MH, Kuipers AF, et al. Addition of salmeterol 50µg bid to anticholinergic treatment in COPD [abstract]. Am J Respir Crit Care Med 1999; 159(3): A523
Dal Negro R, Pomari C, Tognella S, et al. Salmeterol and fluticasone 50(µg/250(µg bid in combination provides a better long-term control than salmeterol 50µg bid alone and placebo in COPD patients already treated with theophylline. Pulm Pharmacol Ther 2003; 16: 241–6
Dauletbaev N, Viel K, Bargon J. Salmeterol or ipratropium bromide/fenoterol in stable mild-moderate COPD [abstract]. Eur Respir J 2001; 18Suppl. 33: 426s
Gupta R, Chhabra S. An evaluation of salmeterol in the treatment of chronic obstructive pulmonary disease. Indian J Chest Dis Allied Sci 2002; 44: 165–72
Stockley R, Chopra N. Salmeterol added to usual therapy, is an effective bronchodilator over 12 months of treatment in chronic obstructive pulmonary disease [abstract]. 12th European Respiratory Society Annual Congress; 2002 Sep 14–18; Stockholm, P1568
Ulrik CS. Efficacy of inhaled salmeterol in the management of smokers with chronic obstructive pulmonary disease: a single centre randomised, double blind, placebo controlled, cross over study. Thorax 1995; 50: 750–4
Calverley P, Pauwels R, Vestbo J, et al. Combined salmeterol and fluticasone in the treatment of chronic obstructive pulmonary disease: a randomised controlled trial. Lancet 2003; 361(9356): 449–56
Celli B, Halpin D, Hepburn R, et al. Symptoms are an important outcome in chronic obstructive pulmonary disease clinical trials: results of a 3-month comparative study using the Breathlessness, Cough and Sputum Scale (BCSS). Respir Med 2003; 97Suppl. A: 535–43
Hanania N, Darken P, Horstman D, et al. The efficacy and safety of fluticasone propionate (250µg)/salmeterol (50µg) combined in the Diskus Inhaler for the treatment of COPD. Chest 2003; 124: 834–43
Rossi A, Kristufek P, Levine BE, et al. Comparison of the efficacy, tolerability, and safety of formoterol dry powder and oral, slow-release theophylline in the treatment of COPD. Chest 2002 Apr; 121: 1058–69
ZuWallack RL, Mahler DA, Reilly D, et al. Salmeterol plus theophylline combination therapy in the treatment of COPD. Chest 2001; 119: 1661–70
Cazzola M, Di Lorenzo G, Di Perna F, et al. Additive effects of salmeterol and fluticasone or theophylline in COPD. Chest 2000 Dec; 118(6): 1576–81
Ram FSF, Jones PW, Castro AA, et al. Oral theophylline for chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2002; (4): CD003902
Newman D, Tamir J, Speed L, et al. Physiological and neuropsychological effects of theophylline in chronic obstructive pulmonary disease. Israel J Med Sci 1994; 30: 811–6
Power CK, Morris AM, Sreenan SK, et al. An assessment of oral theophylline in patients with “reversible” chronic obstructive pulmonary disease [abstract]. Irish J Med Sci 1992; 161(5): 509
Thomas P, Pugsley JA, Stewart JH. Theophylline and salbutamol improve pulmonary function in patients with irreversible chronic obstructive pulmonary disease. Chest 1992; 101(1): 160–5
Barr RG, Bourbeau J, Camargo CA, et al. Inhaled tiotropium for stable chronic obstructive pulmonary disease. Cochrane Database Syst Rev 2005; (2): CD002876
van Noord JA, Bantje TA, Eland ME, et al. The Dutch Tiotropium Study Group: a randomised controlled comparison of tiotropium and ipratropium in the treatment of chronic obstructive pulmonary disease. Thorax 2000; 55(4): 289–94
Vincken W, van Noord JA, Greefhorst AP, et al. Dutch/Belgium Tiotropium Study Group: improved health outcomes in patients with COPD during 1 yr’s treatment with tiotropium. Eur Respir J 2002; 19: 209–16
Brasasco V, Hodder R, Miravitlles M, et al. Health outcomes following treatment for six months with once daily tiotropium compared with twice daily salmeterol in patients with COPD. Thorax 2003; 58: 399–404
Donohue JF, van Noord JA, Bateman ED, et al. A 6-month, placebo-controlled study comparing lung function and health status changes in COPD patients treated with tiotropium or salmeterol. Chest 2002; 122: 47–55
Beeh KM, Beier J, Stark-Lorenzen P, et al. Efficacy of tiotropium in patients with mild-to-moderate COPD [abstract]. Am J Respir Crit Care Med 2004; 169: A519
Casaburi R, Briggs Jr DD, Donohue JF, et al. The spirometric efficacy of once-daily dosing with tiotropium in stable COPD: a 13-week multicenter trial. The US Tiotropium Study Group. Chest 2000; 118(5): 1294–302
Niewoehner D, Rice K, Cote C, et al. Reduced COPD exacerbations and associated health care utilization with once-daily tiotropium in the VA Medical System [abstract]. Am J Respir Crit Care Med 2004; 169: A207
Casaburi R, Mahler DA, Jones PW, et al. A long-term evaluation of once-daily inhaled tiotropium in chronic obstructive pulmonary disease. Eur Respir J 2002 Feb; 19(2): 217–24
Chen CY, Yang KY, Lee YC, et al. Effect of oral aminophylline on pulmonary function improvement and tolerability in different age groups of COPD patients. Chest 2005 Oct; 128(4): 2088–92
Cook D, Guyatt G, Wong E, et al. Regular versus as-needed short-acting inhaled beta-agonist therapy for chronic obstructive pulmonary disease. Am J Respir Crit Care Med 2001 Jan; 163(1): 85–90
Anthonisen NR, Connett JE, Enright PL, et al. Hospitalizations and mortality in the Lung Health Study. Am J Respir Crit Care Med 2002 Aug 1; 166(3): 333–9
Castle W, Fuller R, Hall J, et al. Serevent Nationwide Surveillance Study: comparison of salmeterol with salbutamol in asthmatic patients who require regular bronchodilator treatment. BMJ 1993; 306: 1034–7
Walters EH, Walters JAE, Gibson PW. Regular treatment with long acting beta agonists vs daily regular treatment with short acting beta agonists in adults and children with stable asthma. Cochrane Database Syst Rev 2002; (4): CD003901
Britton MC, Earnshaw JS, Palmer JBD. A twelve month comparison of salmeterol with salbutamol in asthmatic patients. Eur Respir J 1992; 5: 1062–7
D’Alonzo GE. Efficacy of inhaled salmeterol in the treatment of asthma. Eur Resp Rev 1995; 5: 128–32
De Carli G, Arpinelli F, Irvine SH, et al. Change of quality of life induced by salmeterol and salbutamol in adult asthmatic patients [abstract]. Therapie 1995; 50 Suppl.: N118
Ekstrom T, Ringdal N, Tukiainen P, et al. A 3 month comparison of formoterol with terbutaline via turbohaler: a placebo controlled study. Ann Allergy Asthma Immunol 1998; 81: 225–30
Ekstrom T, Ringdal N, Sobradillo V, et al. Low-dose formoterol Turbohaler™ (Oxis™) b.i.d., a 3-month placebo-controlled comparison with terbutaline (q.i.d.). Respir Med 1998; 92(8): 1040–5
FitzGerald J, Chapman K, Della Cioppa G, et al. Sustained bronchoprotection, bronchodilatation, and symptom control during regular formoterol use in asthma of moderate or greater severity. J Allergy Clin Immunol 1999; 103(31): 427–35
Hekking P, Maesen F, Greefhorst A, et al. Long-term efficacy of formoterol compared with salbutamol. Lung 1990; 168 Suppl.: 76–82
Kemp J, Wolfe J, Grady J, et al. Salmeterol powder compared with albuterol aerosol as maintenance therapy for asthma in adolescent and adult patients. Clin Ther 1998; 20(2): 270–82
Kesten S, Chapman KR, Broder I, et al. A three month comparison of inhaled formoterol versus four times daily inhaled albuterol in the management of stable asthma. Am Rev Resp Dis 1991; 144: 622–5
Leblanc P, Knight A, Kreisman H, et al. A placebo controlled crossover comparison of salmeterol and salbutamol in patients with asthma. Am J Respir Crit Care Med 1996; 154: 324–8
Lundback B, Rawlinson DW, Palmer JBD. Twelve month comparison of salmeterol and salbutamol as dry powder formulations in asthmatic patients. Thorax 1993; 153: 148–53
Pearlman DS, Chervinsky P, LaForce C, et al. A comparison of salmeterol with albuterol in the treatment of mild-to-moderate asthma. N Engl J Med 1992; 327: 1420–5
Steffensen I, Faurschou P, Riska H, et al. Inhaled formoterol DP in the treatment of patients with ROAD: a 3 month, placebo controlled comparison of the efficacy and safety of formoterol and salbutamol. Allergy 1995; 50: 657–63
Taylor DR, Town GI, Herbison GP, et al. Asthma control during long term treatment with regular inhaled salbutamol and salmeterol. Thorax 1998; 53: 744–52
Wenzel SE, Lumry W, Manning M, et al. Efficacy, safety, and effects on quality of life of salmeterol versus albuterol in patients with mild to moderate persistent asthma. Ann Allergy 1998; 80(6): 463–70
Bensch G, Lapidus R, Levine B, et al. A randomized, 12-week, double-blind, placebo-controlled study comparing formoterol dry powder inhaler with albuterol metered-dose inhaler. Ann Allergy Asthma Immunol 2001; 86: 9–27
Cloosterman SG, Bijl-Hofland ID, van Herwaarden CL, et al. A placebo-controlled clinical trial of regular monotherapy with short-acting and long-acting beta-2 agonists in allergic asthmatic patients. Chest 2001; 119: 1306–15
Shah L, Wilson AJ, Gibson PG, et al. Long acting β-2 agonists vs theophylline for maintenance treatment of asthma. Cochrane Database Syst Rev 2003; 3: CD001281
Muir JF, Bertin D, French Multicentre Study Group. Salmeterol versus slow-release theophylline combined with ketotifen in nocturnal asthma: a multicentre trial. Eur Respir J 1992; 5: 1197–2000
Nutini S, Martini T, Righi R. Long-term treatment of asthmatic patients with salmeterol vs slow-release theophylline. Respir Med 1998; 92: 683–90
Pastorello AE, Mauro M, Incorvaia C. Comparison of efficacy and safety of inhaled salmeterol and slow-release oral theophylline in patients with moderate/severe asthma. L’Internista 1998; 6: 101–7
Pollard SJ, Sheldon LS, Yancey SW, et al. Salmeterol versus theophylline in the treatment of asthma. Ann Allergy Asthma Immunol 1997; 78: 457–64
Walters EH, Walters J. Inhaled short acting β-2 agonist use in chronic asthma: regular versus as needed treatment. Cochrane Database Syst Rev 2003; (2): CD001285
Apter AJ, Reisine ST, Willard A, et al. The effect of inhaled albuterol in moderate to severe asthma. J Allergy Clin Immunol 1996; 98: 295–301
Hancox RJ, Subbarao P, Kamada D, et al. Beta2-agonist tolerance and exercise-induced bronchospasm. Am J Respir Crit Care Med 2002; 165(8): 1068–70
Dennis S, Sharp S, Vickers M, et al. Regular inhaled salbutamol and asthma control: the TRUST randomised trial. Lancet 2000; 355: 1657–79
Drazen JM, Israel E, Boushey HA, et al. Comparison of regularly scheduled with as-needed use of albuterol in mild asthma. N Engl J Med 1996; 335: 841–7
Manolitsas ND, Wang J, Devalia JL, et al. Regular albuterol, nedocromil sodium, and bronchial inflammation in asthma. Am J Respir Crit Care Med 1995; 151: 1925–30
Sears MR, Taylor DR, Print CG, et al. Regular inhaled beta-agonist treatment in bronchial asthma. Lancet 1990; 336: 1391–6
Nelson HS, Weiss ST, Bleecker ER, et al. The Salmeterol Multicenter Asthma Research Trial: a comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest 2006; 129: 5–26
Walters EH, Walters JAE, Gibson MDP. Long-acting beta2-agonists for chronic asthma in adults and children. Cochrane Database Review, 2003; (4): CD001385
Adinoff A, Schwartz H, Rickard K, et al. Salmeterol compared with current therapies in chronic asthma. J Fam Pract 1998; 47(4): 278–84
Booth H, Fishwick K, Harkawat R, et al. Changes in methacholine induced bronchoconstriction with the long acting beta2 agonist salmeterol in mild to moderate asthmatic patients. Thorax 1993; 48(11): 1121–4
Boulet LP, Laviolette M, Boucher S, et al. A twelve week comparison of salmeterol and salbutamol in the treatment of mild to moderate asthma: a Canadian multicentre study. J Allergy Clin Immunol 1997; 99: 13–21
Busse WW, Casale TB, Murray JJ, et al. Efficacy, safety, and impact on quality of life of salmeterol in patients with moderate persistent asthma. Am J Manag Care 1998; 4(11): 1579–87
Cheung D, Timmers MC, Zwinderman AH, et al. Long term effects of a long acting beta adrenoceptor agonist in patients with mild asthma. N Engl J Med 1992; 327(17): 1198–203
Kavuru M, Melamed J, Gross G, et al. Salmeterol and fluticasone propionate combined in a new powder inhalation device for the treatment of asthma: a randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol 2000; 105(6): 1108–16
Kemp J, DeGraff A, Pearlman D, et al. A 1-year study of salmeterol powder on pulmonary function and hyper responsiveness to methacholine. J Allergy Clin Immunol 1999; 104(6): 1189–97
Shapiro G, Lumry W, Wolfe J, et al. Combined salmeterol 50 mg and fluticasone propionate 250 in the Diskus device for the treatment of asthma. Am J Respir Crit Care Med 2000; 161 (2 Pt 1): 527–34
Wolfe J, Kreitzer S, Chervinsky P, et al. Comparison of powder and aerosol formulations of salmeterol in the treatment of asthma. Ann Allergy 2000; 84(3): 334–40
Booth H, Bish R, Walters J, et al. Salmeterol tachyphylaxis in steroid treated asthmatic subjects. Thorax 1996; 51: 1100–4
Boyd G. Salmeterol xinafoate in asthmatic patients under consideration for maintenance oral corticosteroid therapy: UK Study Group. Eur Respir J 1995; 8: 1494–8
Chuchalin AG, Ovcharenko SI, Goriachkina LA, et al. The safety and efficacy of formoterol (Oxis) turbohaler plus budesonide (Pulmicort) turbohaler in mild to moderate asthma: a comparison with budesonide Turbohaler alone and current non-corticosteroid therapy in Russia. Int J Clin Pract 2002; 56(1): 15–20
Gardiner PV, Ward C, Booth H, et al. Effect of eight weeks treatment with salmeterol on bronchoalveolar lavage inflammatory indices in asthmatics. Am J Respir Crit Care Med 1994; 150: 1006–11
Li X, Bamford T, Ward C, et al. Influence of salmeterol on eosinophil inflammation in bronchial biopsies from asthmatics on inhaled steroid [abstract]. Eur Respir J Suppl 1998; 28:157s
Pauwels RA, Lofdahl CG, Postma DS, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. N Engl J Med 1997; 337: 1404–11
Van der Molen T, Postma DS, Turner MO, et al. Effects of the long acting beta agonist formoterol on asthma control in asthmatic patients using inhaled corticosteroids. Thorax 1996; 52(6): 535–9
Wilding P, Clark M, Thompson J, et al. The effect of long-term treatment with salmeterol on asthma control: a double blind randomised crossover study. BMJ 1997; 314: 1441–6
D’Urzo AD, Chapman KR, Cartier A, et al. Effectiveness and safety of salmeterol in nonspecialist practice settings. Chest 2001; 119:714–9
Pauwels RA, Sears MR, Campbell M, et al. on behalf of the RELIEF Study investigators. Formoterol as relief medication in asthma: a worldwide safety and effectiveness trial. Eur Respir J 2003; 22: 787–94
Busse W, Levine B, Andriano K, et al. Efficacy, tolerability, and effect on asthma-related quality of life of formoterol bid via multidose dry powder inhaler and albuterol QID via metered dose inhaler in patients with persistent asthma: a multicenter, randomized, double-blind, double-dummy, placebo-controlled, parallel-group study. Clin Ther 2004 Oct; 26(10): 1587–98
Lockey RF, DuBuske LM, Friedman B, et al. Nocturnal asthma: effect of salmeterol on quality of life and clinical outcomes. Chest 1999; 115: 666–73
Wolfe J, LaForce C, Friedman B, et al. Formoterol, 24 (ig bid, and serious asthma exacerbations. Chest 2006; 129: 27–38
Westby M, Benson M, Gibson P. Anticholinergic agents for chronic asthma in adults. Cochrane Database Syst Rev 2004; (3): CD003269
Haahtela T, Ahokas I, Ahonen A, et al. Inhaled bronchodilators in asthma: low or high dose? Ann Allergy 1991; 66: 175–80
Philip-Joet F, Reynaud-Gaubert M, Jirou-Najou JL, et al. Comparison of Berodual and salbutamol in asthma: a multicenter evaluation. Respiration 1990; 57: 379–83
Tormey VJ, Pathmakanthan S, Faul J, et al. A comparison of the effects of inhaled salbutamol or oxitropium bromide on bronchial hyper responsiveness in asthmatic patients [abstract]. Irish J Med Sci 1995; 164: 172
Travers J, Marsh S, Williams M, et al. External validity of randomised controlled trials in asthma: to whom do the results of the trials apply? Thorax 2007; 62(3): 219–23
Herland K, Akselsen JP, Skjonsberg OH, et al. How representative are clinical study patients with asthma or COPD for a larger “real life” population of patients with obstructive lung disease? Respir Med 2005 Jan; 99(1): 11–9
Kobilka BK, Dixon RA, Frielle HG, et al. cDNA for the human beta2-adrenergic receptor: a protein with multiple spanning domains and encoded by a gene whose chromosomal location is shared with that of a receptor for platelet growth factor. Proc Natl Acad Sci U S A 1987; 84: 46–50
Frielle T, Daniel KW, Caron MG, et al. Structural basis of beta-adrenergic receptor subtype specificity studies with chimeric beta2/beta2-adrenergic receptors. Proc Natl Acad Sci U S A 1988; 85(24): 9494–8
Johnson M. The β-adrenoceptor. Am J Respir Crit Care Med 1998 Nov; 158 (5 Suppl.): S146–53
Brodde O. Beta 1- and beta 2-adrenoceptors in the human heart: properties, function, and alterations in chronic heart failure. Pharmacol Rev 1991 Jun; 43(2): 203–42
Kobilka B. Adrenergic receptors as models for G protein-coupled receptors. Annu Rev Neurosci 1992; 15: 87–114
Onaran HO, Costa T, Rodbard D. Subunits of guanine nucleotide-binding proteins and regulation of spontaneous receptor activity: thermodynamic model for the interaction between receptors and guanine nucleotide-binding protein subunits. Mol Pharmacol 1993; 43: 245–56
Daaka Y, Luttrell LM, Lefkowitz RJ. Switching of the coupling of the β2-adrenergic receptor to different G proteins by protein kinase A. Nature 1997 Nov 2006; 390: 88–91
Johnson M, Coleman RA. Mechanisms of action of beta2-adrenoceptor agonists. In: Busse WW, Holgate ST, editors. Asthma and rhinitis. Cambridge: Blackwell, 1995: 1278–95
Fujiwara T, Sumimoto K, Itoh T, et al. Relaxing actions of procaterol, a β2-adrenoceptor stimulant, on smooth muscle cells of the dog trachea. Br J Pharmacol 1988; 93: 199–209
Felbel J, Trockur B, Ecker T, et al. Regulation of cytosolic calcium by cAMP and cGMP in freshly isolated smooth muscle cells from bovine trachea. J Biol Chem 1988; 263: 16764–71
Takuwa Y, Takuwa N, Rasmussen H. The effects of isoproterenol on intracellular calcium concentration. J Biol Chem 1988; 263: 762–8
Cook SJ, Small RC, Berry JL, et al. Beta-adrenoceptor subtypes and plasmalemmal K+-channels in trachealis muscle. Br J Pharmacol 1993; 109: 1140–8
Chiu P, Cook SJ, Small RC. Beta-adrenoceptor subtypes and the opening of plasmalemmal K+-channels in bovine tracheal muscle: studies of mechanical activity and ion fluxes. Br J Pharmacol 1993; 109: 1149–56
Wiener CM, Dunn A, Sylvester JT. ATP-dependent K+ channels modulate vasoconstrictor responses to severe hypoxia in isolated ferret lungs. J Clin Invest 1991; 88: 500–4
Kidney JC, Lotvall JO, Lei Y, et al. The effect of inhaled K+ channel openers on bronchoconstriction and airway microvas-cular leakage in anaesthetized guinea pigs. Eur J Pharmacol 1996; 296: 81–7
Sheridan BC, Mclntyre RC, Meldrum DR, et al. KATP channels contribute to beta and adenosine receptor-mediated pulmonary vasorelaxation. Am J Physiol 1997; 273 (5 Pt 1): L950–6
Giembycz MA, Newton R. Beyond the dogma: novel β2-adrenoceptor signaling in the airways. Eur Respir J 2006; 27:1286–306
Goldstein DS. Plasma catecholamines and essential hypertension: an analytical review. Hypertension 1983; 5: 86–99
Ziegler MG, Lake CR, Kopin IJ. Plasma noradrenaline increases with age. Nature 1976; 261: 333–5
Preuss JM, Goldie RG. Age-related changes in muscarinic cholinoceptor function in guinea-pig and rat airways. Naunyn Schmiedeberg Arch Pharmacol 1999; 360: 179–86
Scarpace PJ, Baresi LA. Increased beta-adrenergic receptors in the light-density membrane fraction in lungs from senescent rats. J Gerontol 1988; 43: B163–7
Feldman RD, Limbird LE, Nadeau J, et al. Alterations in leukocyte beta-receptor affinity with aging: a potential explanation for altered beta-adrenergic sensitivity in the elderly. N Engl J Med 1984; 310: 815–9
White M, Roden R, Minobe W, et al. Age-related changes in beta-adrenergic neuroeffector systems in the human heart. Circulation 1994 Sep; 90: 1225–38
Schocken DD, Roth GS. Reduced beta-adrenergic receptor concentrations in aging man. Nature 1977; 267: 856–8
Abrass IB, Scarpace PJ. Human lymphocyte beta-adrenergic receptors are unaltered with age. J Gerontol 1981; 36: 298–301
Landmann R, Bittiger H, Buhler FR. High affinity beta-adrenergic receptors in mononuclear leucocytes: similar density in young and old normal subjects. Life Sci 1981; 29: 1761–71
Doyle V, O’Malley K, Kelly JG. Human lymphocyte beta-adrenoceptor density in relation to age and hypertension. J Cardiovasc Pharmacol 1982; 2: 738–40
Xiao RP, Tomhave ED, Wang D, et al. Age-associated reductions in cardiac β1- and β2-adrenergic responses without changes in inhibitory G proteins or receptor kinases. J Clin Invest 1998 Mar; 101(6): 1273–82
Fitzgerald D, Doyle V, Kelly JG, et al. Cardiac sensitivity to isoprenaline, lymphocyte beta-adrenoceptors and age. Clin Sci (Lond) 1984; 66(6): 697–9
Narayanan N, Derby JA. Alterations in the properties of β-adrenergic receptors of myocardial membranes in aging: impairments in agonist-receptor interactions and guanine nucleotide regulation accompany diminished catecholamine responsiveness of adenylate cyclase. Mech Aging Dev 1982; 19:127–39
Ericsson E, Lundholm L. Adrenergic beta-receptor activity and cyclic AMP metabolism in vascular smooth muscle: variations with age. Mech Aging Dev 1975; 4: 1–6
O’Connor SW, Scarpace PJ, Abrass IP. Age-associated decrease in the catalytic unit activity of rat myocardial adenylate cyclase. Mech Aging Dev 1983; 21: 357–63
Scarpace PJ, Abrass IP. Decreased beta-adrenergic agonist affinity and adenylate cyclase activity in senescent rat lung. J Gerontol 1983; 38: 143–7
Kilts JD, Akazawa T, Richardson MD, et al. Age increases cardiac Galpha i2 expression, resulting in enhanced coupling to G protein-coupled receptors. J Biol Chem 2002; 277(34): 31257–62
Schutzer WE, Xue H, Reed JF, et al. Effect of age on vascular β2 adrenergic receptor desensitization is not mediated by the receptor coupling to Gai proteins. J Gerontol (Series A) 2006; 61: 899–906
Small KM, McGraw DW, Liggett SB. Pharmacology and physiology of human adrenergic receptor polymorphisms. Annu Rev Pharmacol Toxicol 2003; 43: 381–411
Green SA, Cole G, Jacinto M, et al. A polymorphism of the human beta 2-adrenergic receptor within the fourth transmembrane domain alters ligand binding and functional properties of the receptor. J Biol Chem 1993; 268: 23116–21
Green SA, Turki J, Bejarano P, et al. Influence of beta 2-adrenergic receptor genotypes on signal transduction in human airway smooth muscle cells. Am J Respir Cell Mol Biol 1995; 13: 25–33
Moore PE, Laporte JD, Abraham JH, et al. Polymorphism of the beta (2)-adrenergic receptor gene and desensitization in human airway smooth muscle. Am J Respir Crit Care Med 2000; 162: 2117–24
McGraw DW, Forbes SL, Kramer LA, et al. Polymorphisms of the 5′ leader cistron of the human beta2-adrenergic receptor regulate receptor expression. J Clin Invest 1998; 102: 1927–32
Liggett SB. Pharmacogenetics of beta-1- and beta-2-adrenergic receptors. Pharmacology 2000; 61: 167–73
Tan S, Hall IP, Dewar J, et al. Association between beta 2-adrenoceptor polymorphism and susceptibility to bronchodilator desensitization in moderately severe stable asthmatics. Lancet 1997; 350: 995–9
Snyder EM, Kenneth CB, Dietz NM, et al. Influence of β2 adrenergic receptor genotype on airway function during exercise in healthy adults. Chest 2006; 129: 762–90
Martinez FD, Graves PE, Baldini M, et al. Association between genetic polymorphisms of the beta2-adrenoceptor and response to albuterol in children with and without a history of wheezing. J Clin Invest 1997; 100: 3184–8
Xie X, Ott J. Testing linkage disequilibrium between a disease gene and marker loci [abstract]. Am J Hum Genet 1993; 53 Suppl.: 1107
Lima JJ, Thomason DB, Mohamed MH, et al. Impact of genetic polymorphisms of the beta2-adrenergic receptor on albuterol bronchodilator pharmacodynamics. Clin Pharmacol Ther 1999; 65: 519–25
Chinchilli IE, Ford VM, Boushey JG, et al. Use of regularly scheduled albuterol treatment in asthma: genotype-stratified, randomised, placebo-controlled cross-over trial. Lancet 2004; 364: 1505–12
Taylor DR, Drazen JM, Herbison GP, et al. Asthma exacerbations during long term beta agonist use: influence of beta-2 adrenoceptor polymorphism. Thorax 2000; 55: 762–7
Litonjua AA, Silverman EA, Tantisira KG, et al. β2-Adrenergic receptor polymorphisms and haplotypes are associated with airways hyper responsiveness among nonsmoking men. Chest 2004; 126: 66–74
Veen AV, Wierenga EA, Westland R, et al. Limited β2-adrenoceptor haplotypes display different agonist mediated airway responses in asthmatics. Respir Res 2006; 7(1): 19
Gosens R, Zaagsma J, Meurs H, et al. Muscarinic receptor signaling in the pathophysiology of asthma and COPD. Respir Res 2006; 7(1): 73
Ishii M, Kurachi Y. Muscarinic acetylcholine receptor. Curr Pharm Des 2006; 12(28): 3573–81
Peng JY, Vaidehi N, Hall SE, et al. The predicted 3D structures of the human Ml muscarinic acetylcholine receptor with agonist or antagonist bound. Chem Med Chem 2006 Aug; 1(8): 878–90
Messer Jr WS. Cholinergic agonists and the treatment of Alzheimer’s disease. Curr Top Med Chem 2002; 2: 353–8
Bymaster FP, McKinzie DL, Felder CC, et al. Use of M1-M5 muscarinic receptor knockout mice as novel tools to delineate the physiological roles of the muscarinic cholinergic system. Neurochem Res 2003; 28: 437–42
Eglen RM, Choppin A, Watson N. Therapeutic opportunities from muscarinic receptor research. Trends Pharmacol Sci 2001; 22: 409–14
Eglen RM. Muscarinic receptor subtypes in neuronal and nonneuronal cholinergic function. Auton Autacoid Pharmacol 2006 Jul; 26(3): 219–33
Wang Y, Gu Q, Mao F, et al. Activity-dependent expression and distribution of Ml muscarinic ACh receptors in visual cortex neuronal cultures. J Neurosci 1994; 14(7): 4147–58
Garzon M, Pickel VM. Subcellular distribution of M2 muscarinic receptors in relation to dopaminergic neurons of the rat ventral tegmental area. J Comp Neurol 2006 Oct 20; 498(6): 821–39
Carden WB, Bickford ME. The location of muscarinic type 2 receptors within the synaptic circuitry of the cat lateral posterior nucleus. Neurosci Lett 1999 Dec 10; 276(3): 153–6
Wang S, Bickford ME, Van Horn SC, et al. Synaptic targets of thalamic reticular nucleus terminals in the visual thalamus of the cat. J Comp Neurol 2001 Nov 26; 440(4): 321–41
Wang Z, Shi H, Wang H. Functional M3 muscarinic acetylcholine receptors in mammalian hearts. Br J Pharmacol 2004 Jun; 142(3): 395–408
Matsui M, Motomura D, Fujikawa T, et al. Mice lacking M2 and M3 muscarinic acetylcholine receptors are devoid of cholinergic smooth muscle contractions but still viable. J Neurosci 2002 Dec 15; 22(24): 10627–32
Gautam D, Han SJ, Hamdan FF, et al. A critical role for beta cell/M3 muscarinic acetylcholine receptors in regulating insulin release and blood glucose homeostasis in vivo. Cell Metab 2006 Jun; 3(6): 449–61
Proctor GB, Carpenter GH. Regulation of salivary gland function by autonomic nerves. Auton Neurosci 2007 Apr 30; 133(1): 3–18
Roffel AF, Elzinga CR, Van Amsterdam RG, et al. Muscarinic M2 receptors in bovine tracheal smooth muscle: discrepancies between binding and function. Eur J Pharmacol 1988; 153: 73–82
Ten Berge RE, Roffel AF, Zaagsma J. The interaction of selective and non-selective antagonists with pre- and postjunctional muscarinic receptor subtypes in the guinea pig trachea. Eur J Pharmacol 1993; 233: 279–84
van Nieuwstadt RA, Henricks PA, Hajer R, et al. Characterization of muscarinic receptors in equine tracheal smooth muscle in vitro. Vet Q 1997; 19: 54–7
Roffel AF, Elzinga CR, Zaagsma J. Muscarinic M3 receptors mediate contraction of human central and peripheral airway smooth muscle. Pulm Pharmacol 1990; 3: 47–51
Fisher JT, Vincent SG, Gomeza J, et al. Loss of vagally mediated bradycardia and bronchoconstriction in mice lacking M2 or M3 muscarinic acetylcholine receptors. FASEB J 2004; 18: 711–3
Struckmann N, Schwering S, Wiegand S, et al. Role of muscarinic receptor subtypes in the constriction of peripheral airways: studies on receptor-deficient mice. Mol Pharmacol 2003; 64: 1444–51
Roffel AF, Elzinga CR, Zaagsma J. Cholinergic contraction of the guinea pig lung strip is mediated by muscarinic M2-like receptors. Eur J Pharmacol 1993; 250: 267–79
Deshpande DA, White TA, Dogan S, et al. CD38/cyclic ADPribose signaling: role in the regulation of calcium homeostasis in airway smooth muscle. Am J Physiol Lung Cell Mol Physiol 2005; 288: L773–88
Higashida H, Yokoyama S, Hashii M, et al. Muscarinic receptor-mediated dual regulation of ADP-ribosyl cyclase in NG108-15 neuronal cell membranes. J Biol Chem 1997; 272: 31272–7
Deshpande DA, Walseth TF, Panettieri RA, et al. CD38/cyclic ADP-ribose-mediated Ca2+ signaling contributes to airway smooth muscle hyper-responsiveness. FASEB J 2003; 17:452–4
Deshpande DA, Dogan S, Walseth TF, et al. Modulation of calcium signaling by interleukin-13 in human airway smooth muscle: role of CD38/cyclic adenosine diphosphate ribose pathway. Am J Respir Cell Mol Biol 2004; 31: 36–42
Tliba O, Panettieri Jr RA, Tliba S, et al. Tumor necrosis factor-alpha differentially regulates the expression of proinflammatory genes in human airway smooth muscle cells by activation of interferon-beta-dependent CD38 pathway. Mol Pharmacol 2004; 66: 322–9
Tliba O, Cidlowski J, Amrani Y. CD38 expression is insensitive to steroid action in cells treated with TNFα and IFNγ by a mechanism involving the upregulation of glucocorticoid receptor β isoform. Mol Pharmacol 2005 Feb; 69(2): 588–96
Gosens R, Schaafsma D, Nelemans SA, et al. Rho-kinase as a drug target for the treatment of airway hyperresponsiveness in asthma. Mini Rev Med Chem 2006; 6: 339–48
Hirshman CA, Lande B, Croxton TL. Role of M2 muscarinic receptors in airway smooth muscle contraction. Life Sci 1999; 64: 443–8
Lutz S, Freichel-Blomquist A, Yang Y, et al. The guanine nucleotide exchange factor p63RhoGEF, a specific link between Gq/11-coupled receptor signaling and RhoA. J Biol Chem 2005; 280: 11134–9
Somlyo AP, Somlyo AV. Ca2+ sensitivity of smooth muscle and nonmuscle myosin II: modulated by G proteins, kinases, and myosin phosphatase. Physiol Rev 2003; 83: 1325–58
Mak JC, Barnes PJ. Autoradiographic visualization of muscarinic receptor subtypes in human and guinea pig lung. Am Rev Respir Dis 1990; 141: 1559–68
Laitinen A, Partanen M, Hervonen A, et al. Electron microscopic study on the innervation of the human lower respiratory tract: evidence of adrenergic nerves. Eur J Respir Dis 1985; 67: 209–15
Ishihara H, Shimura S, Satoh M, et al. Muscarinic receptor subtypes in feline tracheal submucosal gland secretion. Am J Physiol 1992; 262: L223–8
Ramnarine SI, Haddad EB, Khawaja AM, et al. On muscarinic control of neurogenic mucus secretion in ferret trachea. J Physiol 1996; 494: 577–86
Jeffery PK. Remodeling and inflammation of bronchi in asthma and chronic obstructive pulmonary disease. Proc Am Thorac Soc 004; 1: 176–83
Giraldo E, Martos F, Gomez A, et al. Characterization of muscarinic receptor subtypes in human tissues. Life Sci 1988; 43: 1507–15
Deighton NM, Motomura S, Borquez D, et al. Muscarinic cholinoceptors in the human heart: demonstration, subclassification, and distribution. Naunyn Schmiedeberg Arch Pharmacol 1990; 341: 414–21
Poller U, Nedelka G, Radke J, et al. Age-dependent changes in cardiac muscarinic receptor function in healthy volunteers. J Am Coll Cardiol 1997; 29: 187–93
Kennedy HR, Seifen E. Aging effects on chronotropic actions of muscarinic agonists in isolated rat atria. Mech Aging Dev 1990 Jan; 51(1): 81–7
Brodde OE, Konschak U, Becker K, et al. Cardiac muscarinic receptors decrease with age. J Clin Invest 1998 Jan; 101(2): 471–8
Schneider T, Hein P, Micher-Reher MB. Effects of aging on muscarinic receptor subtypes and function in rat urinary bladder. Naunyn Schmiedeberg Arch Pharmacol 2005 Jul; 372(1): 71–8
Arakawa H, Mochizuki K, Tokuyama A, et al. Airway responsiveness to acetylcholine or capsaicin in immature and mature guinea pigs in vivo. Allergol Intern 2000; 49: 99–104
Preuss JM, Goldie RG. Age-related changes in airway responsiveness to phosphodiesterase inhibitors and activators of adenyl cyclase and guanylyl cyclase. Pulm Pharmacol Ther 1999; 12(4): 237–43
Barnes PJ. Theophylline: new perspectives for an old drug. Am J Respir Crit Care Med 2003; 167: 813–8
Finney MJB, Karlson JA, Persson CGA. Effects of bronchoconstriction and bronchodilation on a novel human small airway preparation. Br J Pharmacol 1985; 85: 29–36
Hansel TT, Tennant RC, Tan AJ, et al. Theophylline: mechanism of action and use in asthma and chronic obstructive pulmonary disease. Drugs Today 2004; 40: 55–69
Barnes PJ. Theophylline in COPD. Proc Am Thoracic Soc 2005; 2: 334–9
Rabe KF, Magnussen H, Dent G. Theophylline and selective PDE inhibitors as bronchodilators and smooth muscle relaxants. Eur Respir J 1995; 8: 637–42
Miura M, Belvisi MG, Stretton CD, et al. Role of potassium channels in bronchodilator responses in human airways. Am Rev Respir Dis 1992; 146: 132–6
Pauwels RA, Joos GF. Characterization of the adenosine receptors in the airways. Arch Int Pharmacodyn Ther 1995; 329: 151–60
Björck T, Gustafsson LE, Dahlen SE. Isolated bronchi from asthmatics are hyperresponsive to adenosine, which apparently acts indirectly by liberation of leukotrienes and histamine. Am Rev Respir Dis 1992; 145: 1087–91
Feoktistov I, Polosa R, Holgate ST, et al. Adenosine A2B receptors: a novel therapeutic target in asthma? Trends Pharmacol Sci 1998; 19: 148–53
Inbe H, Watanabe S, Miyawaki M, et al. Identification and characterization of a cell-surface receptor, P2Y15, for AMP and adenosine. J Biol Chem 2004; 279: 19790–9
Foukas LC, Daniele N, Ktori C, et al. Direct effects of caffeine and theophylline on p110δ and other phosphoinositide 3-kinases: differential effects on lipid kinase and protein kinase activities. J Biol Chem 2002; 277: 37124–30
Yamamori T, Inanami O, Nagahata H, et al. Phosphoinositide 3-kinase regulates the phosphorylation of NADPH oxidase component p47Phox by controlling cPKC/PKCδ but not Akt. Biochem Biophys Res Commun 2004; 316: 720–30
Yasui K, Hu B, Nakazawa T, et al. Theophylline accelerates human granulocyte apoptosis not via phosphodiesterase inhibition. J Clin Invest 1997; 100: 1677–84
Takanashi S, Hasegawa Y, Kanehira Y, et al. Interleukin-10 level in sputum is reduced in bronchial asthma, COPD and in smokers. Eur Respir J 1999; 14: 309–14
Tomita K, Chikumi H, Tokuyasu H, et al. Functional assay of NF-δB translocation into nuclei by laser scanning cytometry: inhibitory effect by dexamethasone or theophylline. Naunyn Schmiedeberg Arch Pharmacol 1999; 359: 249–55
Ito K, Lim S, Caramori G, et al. A molecular mechanism of action of theophylline: induction of histone deacetylase activity to decrease inflammatory gene expression. Proc Natl Acad Sci U S A 2002; 99: 8921–6
Retamales I, Elliott WM, Meshi B, et al. Amplification of inflammation in emphysema and its association with latent adenoviral infection. Am J Respir Crit Care Med 2001; 164: 469–73
Ito K, Barnes PJ, Adcock IM. Glucocorticoid receptor recruitment of histone deacetylase 2 inhibits IL-1 β-induced histone H4 acetylation on lysines 8 and 12. Mol Cell Biol 2000; 20: 6891–903
Cosio B, Tsarprouni L, Ito K, et al. Theophylline restores histone deacetylase activity and steroid responses in COPD macrophages. J Exp Med 2004 Sep 6; 200(5): 689–95
Barnes PJ, Ito K, Adcock IM. A mechanism of corticosteroid resistance in COPD: inactivation of histone deacetylase. Lancet 2004; 363: 731–3
Alberola J, Perez Y, Puigdemont A, et al. Effect of age on theophylline pharmacokinetics in dogs. Am J Vet Res 1993 Jul; 54(7): 1112–5
Peggins JO, McMohan TF, Weiner M, et al. The effects of age on the pharmacokinetics and biotransformation of theophylline in vivo and in vitro in the Mongolian gerbil (Meriones unguiculatus). Mech Aging Dev 1992 Nov; 66(2): 173–86
Jackson SH, Johnston A, Woolard R, et al. The relationship between theophylline clearance and age in adult life. Eur J Clin Pharmacol 1989; 36(1): 29–34
Bellia V, Battaglia S, Matera GM, et al. The use of bronchodilators in the treatment of airway obstruction in elderly patients. Pulmon Pharmacol Therap 2006 Oct; 19(5): 311–9
Fowler JB. Medication monitoring in the elderly. Clin Lab Sci 1995 Jan-Feb; 8(1): 34–8
Windom HH, Burgess CD, Sieber RW, et al. The pulmonary and extrapulmonary effects of inhaled beta-agonists in patients with asthma. Clin Pharmacol Ther 1990 Sep; 48(3): 296–301
Newnham DM. Asthma medications and their potential adverse effects in the elderly: recommendations for prescribing. Drug Saf 2001; 24(14): 1065–80
NAEPP Working Group. Considerations for diagnosing and managing asthma in the elderly. Bethesda (MD): National Institutes of Health; National Heart, Lung, and Blood Institute, 1996. NIH publication no. 96-3662
Bristow MR. Beta-adrenergic receptor blockade in chronic heart failure. Circulation 2000 Feb 8; 101(5): 558–69
Au DH, Curtis JR, Every NR, et al. Association between inhaled beta-agonists and the risk of unstable angina and myocardial infarction. Chest 2002 Mar; 121(3): 846–51
Cazzola M, Imperatore F, Salzillo A, et al. Cardiac effects of formoterol and salmeterol in patients suffering from COPD with preexisting cardiac arrhythmias and hypoxemia. Chest 1998; 114(2): 411–5
Newhouse M, Chapman K, McCallum A, et al. Cardiovascular safety of high doses of inhaled fenoterol and albuterol in acute severe asthma. Chest 1996; 110: 595–603
Tveskov C, Djurhuus MS, Klitgaard NAH. Potassium and magnesium distribution, ECG changes, and ventricular ectopic beats during beta-2-adrenergic stimulation with terbutaline in healthy subjects. Chest 1994 Dec; 106(6): 1654–9
Gregorevic P, Ryall JG, Plant DR, et al. Chronic beta-agonist administration affects cardiac function of adult but not old rats, independent of beta-adrenoceptor density. Am J Physiol Heart Circ Physiol 2005 Jul; 289(1): H344–9
Coughlin SS, Metayer C, McCarthy EP, et al. Respiratory illness, beta-agonists, and risk of idiopathic dilated cardiomyopathy. Am J Epidemiol 1995; 142: 395–403
Au D, Lemaitre RN, Curtis JR, et al. The risk of myocardial infarction associated with inhaled β-adrenoceptor agonists. Am J Respir Crit Care Med 2000; 161: 827–30
Montgomery HE, Gill J, Pumphrey CW. Unsuspected coronary artery disease revealed by administration of nebulised salbutamol. Br Heart J 1994; 72: 181
Neville E, Corris PA, Vivian J, et al. Nebulised salbutamol and angina. BMJ 1982; 285: 796–7
Fisher AA, Davis MW, McGill DA. Acute myocardial infarction associated with albuterol. Ann Pharmacother 2004 Dec; 38(12): 2045–9
Salpeter SR, Ormiston TM, Salpeter EE. Cardiovascular effects of β-agonists in patients with asthma and COPD: a metaanalysis. Chest 2004; 125: 2309–21
Au DH, Udris EM, Fan VS, et al. Risk of mortality and heart failure exacerbations associated with inhaled beta-adrenoceptor agonists among patients with known left ventricular systolic dysfunction. Chest 2003 Jun; 123(6): 1964–9
Calverley PMA, Anderson JA, Celli B, et al. The TORCH investigators: salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med 2007 Feb 22; 356(8): 775–89
Rabe KF. Treating COPD: the TORCH trial, P values and the dodo [editorial]. N Engl J Med 2007 Feb 22; 356(8): 851–3
Tesfamariam B, Waldron T, Seymour AA. Quantitation of tremor in response to β-adrenergic receptor stimulation in primates: relationship with hypokalemia. J Pharmacol Toxicol Meth 1998 Nov; 40(4): 201–5
Sovani MP, Whale CI, Tattersfield AE. A benefit-risk assessment of long acting β-2 agonists in the management of obstructive pulmonary disease. Drug Saf 2004; 27(10): 689–715
Wilkinson JRW, Roberts JA, Brackling P, et al. Paradoxical bronchoconstriction in asthmatic patients after salmeterol by MDI. BMJ 1992; 305: 931–2
Raghunathan K, Nagajothi N. Paradoxical bronchospasm: a potentially life threatening adverse effect of albuterol [case report]. South Med J 2006 Mar; 99(3): 288–9
Finnerty JP, Howarth PH. Paradoxical bronchoconstriction with nebulized albuterol but not with terbutaline. Am Rev Respir Dis 1993; 148: 512–3
O’Callaghan C, Milner AD, Swarbrick A. Paradoxical deterioration in lung function after nebulised salbutamol in wheezy infants. Lancet 1986; 2: 1424–5
Spooner L, Olin JL. Paradoxical bronchoconstriction with albuterol administered by metered-dose inhaler and nebulizer solution. Ann Pharmacother 2005; 39(11): 1924–7
Donohue JF, Menjoge S, Kesten S. Tolerance to bronchodilating effects of salmeterol in COPD. Respir Med 2003; 97: 1014–20
Salpeter SR, Ormiston TM, Salpeter EE. Meta-analysis: respiratory tolerance to regular β2 agonist use in patients with asthma. Ann Int Med 2004; 140: 802–13
Mann M, Chowdhury B, Sullivan E, et al. Serious asthma exacerbations in asthmatics treated with high-dose formoterol. Chest 2003; 124: 70–4
Salpeter SR, Buckley NS, Ormiston TM, et al. Meta-analysis: effect of long acting β agonists on severe asthma and asthma related deaths. Ann Int Med 2006; 144: 904–12
Wooltorton E. Salmeterol (Serevent) asthma trial halted early. CMAJ 2003 Mar 18; 168(6): 738
Takeda S, Elefteriou F, Levasseur R, et al. Leptin regulates bone formation via the sympathetic nervous system. Cell 2000; 111: 305–17
Bonnet N, Benhamou CL, Brunet-Imbault B, et al. Severe bone alterations under beta-2 agonist treatments: bone mass, microarchitecture and strength analyses in female rats. Bone 2005 Nov; 37(5): 622–33
Bonnet N, Brunet-Imbault B, Aarlettaz A, et al. Alteration of trabecular bone under chronic beta2 agonist treatment. Med Sci Sports Exerc 2005 Sep; 37(9): 1493–501
Pasco JA, Henry MJ, Sanders KM, et al. Beta-adrenergic blockers reduce the risk of fracture partly by increasing bone mineral density: Geelong Osteoporosis Study. J Bone Miner Res 2004; 19: 19–24
Schlienger RG, Kraenzlin ME, Jick SS, et al. Use of betablockers and risk of fractures. JAMA 2004 Sep 15; 292(11): 1326–32
Gross NJ, Skorodin MS. Anticholinergic, antimuscarinic bronchodilators. Am Rev Respir Dis 1984; 129: 856–70
Feinberg M. The problems of anticholinergic adverse effects in older patients. Drugs Aging 1993 Jul-Aug; 3(4): 335–48
Gross NJ. Anticholinergic agents in asthma and COPD. Eur J Pharmacol 2006 Mar 8; 533(1–3): 36–9
Hall SK. Acute angle-closure glaucoma as a complication of combined beta-agonist and ipratropium bromide therapy in the emergency department. Ann Emerg Med 1994 Apr; 23(4): 884–7
Reuser T, Flanagan DW, Borlan C, et al. Acute angle closure glaucoma occurring after nebulized bronchodilator treatment with ipratropium bromide and salbutamol. J R Soc Med 1992 Aug; 85(8): 499–500
Covelli H, Bhattacharya S, Cassino C, et al. Absence of electrocardiographic findings and improved function with once-daily tiotropium in patients with chronic obstructive pulmonary disease. Pharmacotherapy 2005 Dec; 25(12): 1708–18
Morganroth J, Golisch W, Kesten S. Electrocardiographic monitoring in COPD patients receiving tiotropium. COPD 2004; 1(2): 181–90
Salpeter SR, Buckley NS, Salpeter EE. Meta-analysis: anticholinergics, but not β-agonists, reduce severe exacerbations and respiratory mortality in COPD. J Gen Int Med 2006 Oct; 21(10): 1011–9
Ancelin ML, Artero S, Portet F, et al. Non-degenerative mild cognitive impairment in elderly people and use of anticholinergic drugs: longitudinal cohort study. BMJ 2006 Feb; 332: 455–9
Aizenberg D, Sigler M, Weizman A, et al. Anticholinergic burden and the risk of falls among elderly psychiatric inpatients: a 4 year case control study. Int Psychogeriat 2002 Mar; 14(3): 307–10
Ness J, Hoth A, Barnett MJ, et al. Anticholinergic medications in community-dwelling older veterans: prevalence of anticholinergic symptoms, symptom burden, and adverse drug events. Am J Geriatr Pharmacother 2006 Mar; 4(1): 42–51
Sessler CN. Theophylline toxicity: clinical features of 116 consecutive cases. Am J Med 1990 Jun; 88(6): 567–76
Shannon M. Life-threatening events after theophylline overdose: a 10-year prospective analysis. Arch Intern Med 1999 May 10; 159(9): 989–94
Howell RE, Meuhsam VT, Kinnier WJ. Mechanism for the emetic side effect of xanthine bronchodilators. Life Sci 1990; 46(8): 563–8
Barr RG, Rowe BH, Camargo Jr CA. Methylxanthines for exacerbations of chronic obstructive pulmonary disease: meta-analysis of randomised trials. BMJ 2003; 327: 643
Paggiaro PL, Giannini D, Di Franco A, et al. Comparison of inhaled salmeterol and individually dose-titrated slow-release theophylline in patients with reversible airway obstruction. European Study Group. Eur Respir J 1996 Aug; 9(8): 1689–95
Yurdakul AS, Calisir HC, Tunctan B, et al. Comparison of second controller medications in addition to inhaled corticosteroid in patients with moderate asthma. Respir Med 2002; 96(5): 322–9
Shi D, Daly JW. Chronic effects of xanthines on levels of central receptors in mice. Cell Mol Neurobio 1999 Dec; 19(6): 719–32
Fjellbirkeland L, Gulsvik A, Palmer JBD. The efficacy and tolerability of inhaled salmeterol and individually dose-titrated, sustained-release theophylline in patients with reversible airways disease. Respir Med 1994; 88(8): 599–607
Crowley JJ, Cusack BJ, Jue SG, et al. Aging and drug interactions: II. Effect of phenytoin and smoking on the oxidation of theophylline and cortisol in healthy men. J Pharmacol Exp Ther 1988; 245: 513–23
Sarkar M, Polk RE, Guzelian PS, et al. In vitro effect of fluoroquinolones on theophylline metabolism in human liver microsomes. Antimicrob Agents Chemother 1990; 34: 594–9
Loi CM, Parker BM, Cusack BJ, et al. Aging and drug interactions: III. Individual and combined effects of cimetidine and ciprofloxacin on theophylline metabolism in healthy male and female nonsmokers. Pharmacol Exp Ther 1997; 280(2): 627–37
Acknowledgements
No sources of funding were used to assist in the preparation of this review. The authors have no conflicts of interest that are directly relevant to the content of this review. The authors wish to thank Mrs Shirley Green for assistance in typing this manuscript and Mr Andrew Pierce for his help with the pharmacological tables.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gupta, P., O’Mahony, M.S. Potential Adverse Effects of Bronchodilators in the Treatment of Airways Obstruction in Older People. Drugs Aging 25, 415–443 (2008). https://doi.org/10.2165/00002512-200825050-00005
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002512-200825050-00005