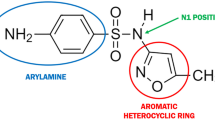
Abstract
Objective: To evaluate cross reactivity between sulfonamide antimicrobials and celecoxib in patients with histories of allergies to sulfonamide antimicrobials.
Methods: Immunocompetent patients with a history of sulfonamide antimicrobial allergy who were being considered for therapy with celecoxib were prospectively enrolled. Sulfamethoxazole and trimethoprim skin prick and intradermal testing and/or an in vitro lymphocyte toxicity assay were performed. If skin testing was negative, an oral challenge with sulfamethoxazole and trimethoprim was performed. Oral challenges with celecoxib were administered to all patients.
Results: Twenty-eight immunocompetent patients (26 female; mean age 60 years) were evaluated. History of sulfonamide antimicrobial allergy included urticaria (n = 7), cutaneous eruptions (n = 9), and other (n = 12). Four of the 28 patients who were skin prick tested were positive to sulfamethoxazole and two of the ten patients who underwent in vitro testing were positive to sulfamethoxazole. All 28 patients were administered celecoxib and tolerated the medication. Phone call follow up in 25 patients disclosed that 15 patients continued to take celecoxib, while five patients did not take celecoxib following the oral challenge, and five discontinued celecoxib due to adverse effects, lack of drug efficacy or physician preference.
Conclusions: Confusion exists regarding the potential for cross reactivity between sulfonamide antimicrobials and other sulfonamide-containing compounds. The six sulfonamide-allergic patients tolerated celecoxib uneventfully. This pilot study supports the hypothesis that the potential for cross-reactivity between celecoxib and sulfonamide antimicrobials appears to be low. However, further investigations are required to confirm this.






Similar content being viewed by others
References
Hawkey CJ. Cox-2 inhibitors. Lancet 1999; 353: 307–14
Koch WJ, Sidel VW, Dexter M, et al. Adverse reactions to sulfisoxazole, sulfamethoxazole and nitrofurantoin: manifestations and specific reaction rates during 2118 courses of therapy. Arch Intern Med 1971; 128: 399–404
Sullivan TJ. Cross-reactions among furosemide, hydrochlorothiazide and sulfonamides. JAMA 1991; 265: 120–1
Knowles S, Shapiro L, Shear NH. Should celecoxib be contraindicated in patients who are allergic to sulfonamides? Revisiting the meaning of ‘sulfa’ allergy. Drug Saf 2001; 24(4): 239–47
Hansbrough JR, Wedner HJ, Chaplin DD. Anaphylaxis to intravenous furosemide. J Allergy Clin Immunol 1987; 80: 538–41
Sullivan TJ. Cross-reactions among furosemide, hydrochlorothiazide and sulfonamides. JAMA 1991; 265: 120–1
Barrio M, Tornero P, Baeza M, et al. Cross-reactivity among the para-amine group in sulfonamide-induced urticaria and fixed drug eruption [abstract]. J Allergy Clin Immunol 1991; 87: 364A
Stock JG. Sulfonamide hypersensitivity and acetazolamide. Arch Ophthalmol 1990; 108: 634–5
Schnewweiss F. Cross-sensitivity between sulfonamides and furosemide [letter]. Clin Pharm 1983; 2: 510
Stock JG. Sulfonamide hypersensitivity and acetazolamide. Arch Ophthalmol 1990; 108: 634–5
Stricker B, Biriell C. Skin reactions and fever with indapamide. BMJ 1987; 295: 1313–4
De Barrio M, Tornero P, Zubeldia JM, et al. Fixed drug eruption induced by indapamide: cross-reactivity with sulfonamides. J Investig Allergol Clin Immunol 1998; 8: 253–5
Shenfield GM, Jacka J. Adverse drug reaction [letter]. Lancet 2001; 57: 561
Bretza J. Thrombocytopenia due to sulfonamide cross-sensitivity. Wis Med J 1982; 81: 21–3
Shenfield GM, Jacka J. Adverse drug reactions [letter]. Lancet 2001; 357: 561
Britschgi M, Steiner U, Schmid S, et al. T-cell involvement in drug-induced acute generalized exanthematous pustulosis. J Clin Invest 2001; 107: 1433–41
Neuman MG, Malkiewicz IM, Shear NH. A novel lymphocyte toxicity assay to assess drug hypersensitivity syndromes. Clin Biochem 2000 Oct; 33(7): 517–24
Weber EA, Uetrecht JP, Knowles SR, et al. Detection of sulfamethoxazole hypersensitivity: skin testing with sulfamethoxazoyl-poly-L-tyrosine [abstract]. J Allergy Clin Immunol 1994; 93: 623A
Gruchalla R, Sullivan T. Detection of human IgE to sulfamethoxazole by skin testing with sulfamethoxazoly-poly-L-tyrosine. J Allergy Clin Immunol 1991; 88: 784–92
Spielberg SP. Acetaminophen toxicity in human lymphocytes in vitro. J Pharmacol Exp Ther 1980; 213: 395–8
Harle DG, Baldo BA, Wells JV. Drugs as allergens: detection and combining site specificities of IgE antibodies to sulfamethoxazole. Mol Immunol 1988; 25: 1347–54
Shear NH, Spielberg SP. Anticonvulsant hypersensitivity syndrome: in vitro assessment of risk. J Clin Invest 1988; 82: 1826–32
Knowles SR, Shapiro L, Shear NH. Serious adverse drug reactions induced by minocycline: report of 13 patients and review of the literature. Arch Dermatol 1996; 132: 934–9
Rieder M, Uetrecht J, Shear NH, et al. Diagnosis of sulfonamide hypersensitivity reactions by in-vitro ‘rechallenge’ with hydroxylamine metabolites. Ann Intern Med 1989; 110: 286–9
Rieder M, Ueterecht J, Shear NH, et al. Diagnosis of sulfonamide hypersensitivity reactions by in vitro ‘rechallenge’ with hydroxylamine metabolites. Ann Intern Med 1989; 110: 286–9
Cribb A, Spielberg S. Hepatic microsomal metabolism of sulfamethoxazole to the hydroxylamine. Drug Metab Dispos 1990; 18: 784–7
Cribb A, Spielberg S, Griffin G. N4-hydroxylation of sulfamethoxazole by cytochrome P450 of the cytochrome P4502C subfamily and reduction of sulfamethoxazole hydroxylamine in human and rat hepatic microsomes. Drug Metab Dispos 1995; 23: 406–14
Cribb A, Miller M, Leeder J, et al. Reactions of the nitroso and hydroxylamine metabolites of sulfamethoxazole with reduced glutathione. Drug Metab Dispos 1991; 19: 900–6
Spielberg SP, Leeder JS, Cribb AE, et al. Is sulfamethoxazole hydroxylamine the proximal toxin for sulfamethoxazole toxicity? Eur J Clin Pharmacol 1989; A173: 04:37
Rieder MJ, Uetrecht J, Shear NH, et al. Diagnosis of sulfonamide hypersensitivity reactions by in-vitro ‘rechallenge’ with hydroxylamine metabolites. Ann Intern Med 1989; 110(4): 286–9
Naisbitt DJ, Hough SJ, Gill HJ, et al. Cellular disposition of sulfamethoxazole and its metabolites: implications for hypersensitivity. Br J Pharmacol 1999 Mar; 12: 1393–407
Cribb AE, Lee BL, Trepanier LA, et al. Adverse reactions to sulfonamide and sulfonamide-trimethoprim antimicrobials: clinical syndromes and pathogenesis. Adverse Drug React Toxicol Rev 1996 Mar; 1: 9–50
Shapiro LE, Neuman MG, Malkiewicz I, et al. Assessment of sulfonamide-induced hypersensitivity syndrome reactions with the use of a novel lymphocyte toxicity assay: preliminary results. Clin Invest Med 1998; 21:Suppl. 15: A571
Neuman MG, Shapiro LE, Phillips E, et al. Lymphocyte toxicity test as a predictor of hypersensitivity reactions in immunocompetent and immunocompromised populations [abstract]. Ther Drug Monit 1999; 21: 453
Neuman MG, Malkiewicz IM, Shear NH. A novel lymphocyte toxicity assay to assess drug hypersensitivity syndromes. Clin Biochem 2000 Oct; 33(7): 517–24
Newman LC, Lay CL, O’Connor KA, et al. Lack of cross-reactivity to sumatriptan in patients allergic to sulfonamides: a retrospective chart review [letter]. Headache 1999; 39: 372
Patterson R, Bello AE, Lefkowith J. Immunologic tolerability profile of celecoxib. Clin Ther 1999 Dec; 21: 2065–79
Wilholm BE. Identification of sulfonamide-like adverse drug reactions to celecoxib in the World Health Organizaion database. Curr Med Res Opin 2001; 17: 210–6
Weber EA, Knight A. Testing for allergy to antibiotics. Semin Dermatol 1989; 8: 204–12
Acknowledgements
No sources of funding were used to assist in the preparation of this manuscript. Dr S. Knowles and Dr N. Shear have acted as paid consultants for Pharmacia and Pfizer.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shapiro, L.E., Knowles, S.R., Weber, E. et al. Safety of Celecoxib in Individuals Allergic to Sulfonamide. Drug-Safety 26, 187–195 (2003). https://doi.org/10.2165/00002018-200326030-00004
Published:
Issue Date:
DOI: https://doi.org/10.2165/00002018-200326030-00004