Abstract
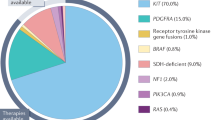
Gastrointestinal stromal tumours (GIST) are the most common mesenchymal tumours of the gastrointestinal tract, previously classified as leiomyosarcomas. Most GIST express KIT and the majority have mutations in the KIT gene. The most common KIT mutation occurs in the juxtamembrane domain of exon 11. These mutations lead to cellular proliferation and survival. GIST with exon 11 mutations respond better to tyrosine kinase inhibitors (TKIs) than those with exon 9 mutations. Most KIT-negative GIST express platelet-derived growth factor receptor (PDGFR)-α; however, a small percentage of GIST are negative for both KIT and PDGFRα. Imatinib and other TKIs have dramatically improved the outcome of patients with metastatic GIST. Newer and more advanced TKIs are under intense investigation as eventually all GIST patients develop resistant tumours. In addition, these drugs can be utilized in the pre-operative setting for patients with unresectable localized tumours or those at high risk for surgical morbidity. TKIs have been given in the adjuvant setting for patients with resected tumours at high risk for recurrence. The duration of adjuvant therapy is currently under evaluation; however, it is possible that these patients would need to continue therapy indefinitely.


Similar content being viewed by others
References
Mazur MT, Clark HB. Gastric stromal tumors: reappraisal of histogenesis. Am J Surg Pathol 1983 Sep; 7(6): 507–19
Hirota S, Isozaki K, Moriyama Y, et al. Gain of function mutations of c-kit in human gastrointestinal stromal tumors. Science 1998 Jan; 279: 577–80
Hirota S, Ohashi A, Nishida T, et al. Gain-of-function mutations of platelet-derived growth factor receptor alpha gene in gastrointestinal stromal tumors. Gastroenterology 2003 Sep; 125(3): 660–7
Heinrich MC, Corless CL, Duensing A, et al. PDGFRA activating mutations in gastrointestinal stromal tumors. Science 2003 Jan; 299(5607): 708–10
West RB, Corless CL, Chen X, et al. The novel marker, DOG 1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutation status. Am J Pathol 2004 Jul; 165: 107–13
Espinosa I, Lee CH, Kim MK, et al. A novel monoclonal antibody against DOG1 is a sensitive and specific marker for gastrointestinal stromal tumors. Am J Surg Pathol 2008 Feb; 32: 210–8
Lasota J, Miettinen M. Clinical significance of oncogenic KIT and PDGFRA mutations in gastrointestinal stromal tumours. Histopathology 2008 Sep; 53(3): 245–66
Lasota J, Corless CL, Heinrich MC, et al. Clinicopathologic profile of gastrointestinal stromal tumors (GISTs) with primary KIT exon 13 or exon 17 mutations: a multicenter study on 54 cases. Mod Pathol 2008 Apr; 21: 476–84
Lasota J, Wozniak A, Sarlomo-Rikala M, et al. Mutations in exons 9 and 13 of KIT gene are rare events in gastrointestinal stromal tumors: a study of two hundred cases. Am J Pathol 2000 Oct; 157: 1091–5
Mussi C, Schildhaus HU, Gronchi A, et al. Therapeutic consequences from molecular biology for gastrointestinal stromal tumor patients affected by neurofibromatosis type 1. Clin Cancer Res 2008 Jul 15; 14(14): 4550–5
Kawanowa K, Sakuma Y, Sakurai S, et al. High incidence of microscopic gastrointestinal stromal tumors in the stomach. Hum Pathol 2006 Dec; 37: 1527–35
Agaimy A, Wunsch PH, Dirnhofer S, et al. Microscopic gastrointestinal stromal tumors in esophageal and intestinal surgical resection specimens: a clinicopathologic, immunohistochemical, and molecular study of 19 lesions. Am J Surg Pathol 2008 Jun; 32: 867–73
Lok KH, Lai L, Yiu HL, et al. Endosonographic surveillance of small gastrointestinal tumors originating from muscularis propria. J Gastrointestin Liv Dis 2009 Jun; 18: 177–80
Sun S, Ge N, Wang C, et al. Endoscopic band ligation of small gastric stromal tumors and follow-up by endoscopic ultrasonography. Surg Endosc 2007; 21: 574–8
Choi SM, Kim MC, Jung GJ, et al. Laparoscopic wedge resection for gastric GIST: long-term follow-up results. Eur J Surg Oncol 2007 May; 33(4): 444–7
Eisenberg BL, Harris J, Blanke CD, et al. Phase II trial of neoadjuvant/adjuvant imatinib mesylate (IM) for advance primary and metastatic/recurrent operable gastrointestinal stromal tumor (GIST): early results of RTOG 0132/ACRIN 6665. J Surg Oncol 2009 Jan; 99(1): 42–7
McAuliffe JC, Hunt KK, Lazar AJ, et al. A randomized phase II study of preoperative plus postoperative imatinib in GIST: evidence of rapid radiographic response and temporal induction of tumor cell apoptosis. Ann Surg Oncol 2009 APR; 16: 910–9
Fiore M, Palassini E, Fumagalli E, et al. Preoperative imatinib mesylate for unresectable or locally advanced primary gastrointestinal stromal tumours (GIST). Eur J Surg Oncol 2009; 35(7): 739–45
Rutkowski P, Nowecki Z, Nyckowski P, et al. Surgical treatment of patients with initially inoperable and/or metastatic gastrointestinal stromal tumors (GIST) during therapy with imatinib mesylate. J Surg Oncol 2006; 93(4): 304–11
Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol 2006 May; 23: 70–83
Nillson B, Sjolund K, Kindblom LG, et al. Adjuvant imatinib treatment improves recurrence-free survival in patients with high-risk gastrointestinal stromal tumours (GIST). Br J Cancer 2007 Jun; 96: 1656–8
DeMatteo RP, Owzar K, Antonescu CR, et al. Efficacy of adjuvant imatinib mesylate following complete resection of localized primary gastrointestinal stromal tumor (GIST) at high risk of recurrence: the U.S. Intergroup phase II trial ACOSOG Z9000 [abstract no. 8]. 2008 Gastrointestinal Cancer Symposium — ASCO; 2008 Jan 25–27; Orlando (FL)
DeMatteo RP, Owzar K, Maki R, et al. Adjuvant imatinib mesylate increased recurrence free survival (RFS) in patients with completely resected localized primary gastrointestinal stromal tumor (GIST): North America Intergroup Phase III trial ACOSOG Z9001 [abstract]. J Clin Oncol 2007 May; 25: 10079
DeMatteo RP, Ballman KV, Antonescu CR, et al. Adjuvant imatinib mesylate after resection of localized, primary gastrointestinal stromal tumor (GIST): a randomized, double-blind study, placebo-controlled trial. Lancet 2009 Mar; 373(9669): 1097–104
Scandinavian Sarcoma Group. Study comparing 12 months versus 36 months of imatinib in the treatment of gastrointestinal stromal tumor (GIST) [ClinicalTrials.gov identifier NCT00116935]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Jan 14]
European Organization for Research and Treatment of Cancer. Imatinib mesylate or observation only in treating patients who have undergone surgery for localized gastrointestinal stromal tumor [ClinicalTrials.gov identifier NCT00103168]. US National Institutes of Health, Clinical Trials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Jan 14]
Joensuu H, Roberts PJ, Sarlomo-Rikala M, et al. Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor. N Engl J Med 2001 Apr; 344(14): 1052–6
vanOosterom AT, Judson IR, Verweij J, et al. Update of phase I study of imatinib (STI571) in advanced soft tissue sarcomas and gastrointestinal stromal tumors: a report of the EORTC Soft Tissue and Bone Sarcoma Group. Eur J Cancer 2002 Sep; 38 Suppl. 5: S83–7
Demetri GD, von Mehren M, Blanke CD, et al. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med 2002 Aug; 347: 472–80
Blanke CD, Demetri GD, von Mehren M, et al. Long-term results from a randomized phase II trial of standard-dose versus higher-dose imatinib mesylate for patients with unresectable or metastatic gastrointestinal stromal tumors expressing KIT. J Clin Oncol 2008 Feb; 26(4): 620–5
Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet 2004 Sep; 364: 1127–34
Casali PG, Verweij J, Kotasek D, et al. Imatinib mesylate in advanced gastrointestinal stromal tumors (GIST): survival analysis of the Intergroup EORTC ISG AGITG randomized trial in 946 patients [abstract]. Eur J Cancer 2005; Suppl. 3: 711
Blanke CD, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S 0033. J Clin Oncol 2008 Feb; 26(4): 626–32
MetaGIST. Comparison of two doses of imatinib for the treatment of unresectable or metastatic gastrointestinal stromal tumors: a meta-analysis of 1,640 patients. Gastrointestinal Stromal Tumor Meta-Analysis Group (Meta-GIST). J Clin Oncol 2010 Mar; 28: 1247–53
Dagher R, Cohen M, Williams G, et al. Approval summary: imatinib mesylate in the treatment of metastatic and/or unresectable malignant gastrointestinal stromal tumors. Clin Cancer Res 2002 Oct; 8: 3034–8
Ferrero D, Pogliani EM, Rege-Cambrin G, et al. Corticosteroids can reverse severe imatinib-induced hepatotoxicity. Haematologica 2006 Jun; 91 (6 Suppl.): ECR27
Quek R, Morgan JA, George S, et al. Small molecule tyrosine kinase inhibitor and depression. J Clin Oncol 2009 Aug; 27: 312–3
Demetri GD, van Oosterom AT, Garrett CR, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet 2006 Oct; 368: 1329–38
Demetri GD, Huang X, Garrett CR, et al. Novel statistical analysis of long-term survival to account for crossover in a phase III trial of sunitinib (SU) vs. placebo (PL) in advanced GIST after imatinib (IM) failure [abstract]. J Clin Oncol 2008 May; 26: 10524
George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer 2009 Jul; 45: 1959–68
Gajiwala KS, Wu JC, Christensen J, et al. Kit kinase mutants show unique mechanisms of drug resistance to imatinib and sunitinib in gastrointestinal stromal tumor patients. Proc Natl Acad Sci U S A 2009 Feb; 106(5): 1542–7
Goodman VL, Rock EP, Dagher R, et al. Approval summary: sunitinib for the treatment of imatinib refractory or intolerant gastrointestinal tumors and advanced renal cell carcinoma. Clin Cancer Res 2007 Mar; 13(5): 1367–73
Desai J, Yassa L, Marqusee E, et al. Hypothyroidism after sunitinib treatment for patients with gastrointestinal stromal tumors. Ann Intern Med 2006 Nov; 145: 660–4
Schittenhelm MM, Shiraga S, Schroeder A, et al. Dasatinib (BMS-354825), a dual SRC/ABL kinase inhibitor, inhibits the kinase activity of wild-type, juxtamembrane, and activation loop mutant kit isoforms associated with human malignancies. Cancer Res 2006 Jan; 66(1): 473–81
Shor AC, Kescheman EA, Lee FY, et al. Dasatinib inhibits migration and invasion in diverse human sarcoma cell lines and induces apoptosis in bone sarcoma cells dependent on Src kinase for survival. Cancer Res 2007 Mar; 67: 2800–8
Sarcoma Alliance for Research through Collaboration. Trial of dasatinib in advanced sarcomas [ClinicalTrials.gov identifier NCT00464620]. US National Institutes of Health, ClinicalTrials.gov [online]. Available from URL: http://www.clinicaltrials.gov [Accessed 2011 Jan 14]
Wiebe L, Kasza KE, Maki RG, et al. Activity of sorafenib (SOR) in patients (pts) with imatinib (IM) and sunitinib (SU)-resistant (RES) gastrointestinal stromal tumors (GIST): a phase II trial of the University of Chicago phase II consortium [abstract]. J Clin Oncol 2008 May; 26 (15 Suppl.): 10502
Gelderbloom HJ, Montemurro M, Schutte J, et al. Sorafenib fourth-line treatment in imatinib, sunitinib, and nilotinib resistant metastatic GIST: a retrospective analysis [abstract no. 51]. ASCO Gastrointestinal Cancers Symposium 2009; 2009 Jan 15–17; San Francisco (CA)
Sleijfer S, Ray-Coquard I, Papai Z, et al. Pazopanib, a multikinase angiogenesis inhibitor, in patients with relapsed or refractory advanced soft tissue sarcoma: a phase II study from the European organisation for research and treatment of cancer-soft tissue and bone sarcoma group (EORTC study 62043). J Clin Oncol 2009 Jul; 27(19): 3126–32
Demetri GD, Casali PG, Blay JY, et al. A phase I study of single-agent nilotinib or in combination with imatinib in patients with imatinib-resistant gastrointestinal stromal tumors. Clin Cancer Res 2009 Sep; 15: 5910–6
Schoffski P, Reichardt P, Blay JY, et al. A phase I–II study of everolimus (RAD001) in combination with imatinib in patients with imatinib-resistant gastrointestinal stromal tumors. Ann Oncol 2010 May; 21(10): 1990–8
Pouyssegur J, Dayan F, Mazure NM, et al. Hypoxia signaling in cancer and approaches to enforce tumor regression. Nature 2006 May; 441: 437–43
Semenza GL. Targeting HIF-1 for cancer therapy. Nat Rev Cancer 2003 Oct; 3: 721–31
Faivre S, Kroemer G, Raymond E. Current development of mTOR inhibitors as anticancer agents. Nat Rev Drug Discov 2006 Aug; 5: 671–88
Braconi C, Bracci R, Bearzi I, et al. Insulin-like growth factor (IGF) 1 and 2 help to predict disease outcome in GIST patients. Ann Oncol 2008 Mar; 19(7): 1293–8
Tarn C, Rink L, Merkel E, et al. Insulin-like growth factor 1 receptor is a potential therapeutic target for gastrointestinal stromal tumors. Proc Natl Acad Sci U S A 2008 Jun; 105(24): 8387–92
Janeway KA, Zhu MJ, Barretina J, et al. Strong expression of IGF1R in pediatric gastrointestinal tumors without IGF1R genomic amplification. Int J Cancer Feb 2010 Dec 1; 127(11): 2718–22
Therasse P, Arbuck SG, Eisenhauer EA, et al. New guidelines to evaluate the response to treatment in solid tumors. J Natl Cancer Inst 2000 Feb; 92: 205–16
Prior JO, Montemurro M, Orcuto MV, et al. Early prediction of response to sunitinib after imatinib failure by 18F-fluorodeoxyglucose positron emission tomography in patients with gastrointestinal stromal tumor. J Clin Oncol 2009 Jan; 27(3): 439–45
Choi H, Charnsanavej C, Faria S, et al. Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol 2007 May; 25: 1753–9
Benjamin RS, Choi H, Macapinlac HA, et al. We should desist using RECIST, at least in GIST. J Clin Oncol 2007 May; 25: 1760–4
Acknowledgements
No sources of funding were used to assist in the preparation of this review. Dr Patel has received honoraria from Novartis for speaking and advising. Dr Ganjoo has no conflicts of interest that are directly relevant to the content of this review.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ganjoo, K.N., Patel, S. Current and Emerging Pharmacological Treatments for Gastrointestinal Stromal Tumour. Drugs 71, 321–330 (2011). https://doi.org/10.2165/11585370-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11585370-000000000-00000