Abstract
Background and objective
Rheumatoid arthritis (RA) is an autoimmune disease with an unknown aetiology that results in >9 million physician visits and >250 000 hospitalisations per year in the US. Tumour necrosis factor-a (TNFa) inhibitors are effective agents in treating RA; however, their cost effectiveness as first-line agents has not been investigated. This study aimed to examine the cost effectiveness of using TNFa inhibitors (both as monotherapy and in combination with methotrexate) as first-line agents versus methotrexate (monotherapy) from a payer perspective.
Methods
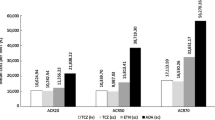
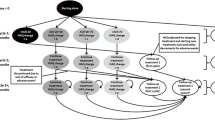
A Markov model was developed utilising a discount rate of 3% per annum, a cycle length of 1 year and a lifetime time-horizon for a hypothetical cohort of US females aged 55–60 years who had been diagnosed with RA. The source of data for predicted probabilities, expected mortality rates and treatment costs in year 2005 US dollars (drug, toxicity, monitoring and hospitalisation) was from the literature. These costs are assigned in 5-year cycles (calculated from initial 1-year estimates) along with the effect on quality-adjusted life-years (QALYs), which were calculated using the Health Assessment Questionnaire score. Univariate sensitivity analyses were conducted on all relevant parameters. Results: Adalimumab, etanercept, adalimumab plus methotrexate and infliximab plus methotrexate had incremental cost-effectiveness ratios (ICERs) versus methotrexate monotherapy of $US63 769, $US89 772, $US194 589 and $US409 523 per QALY, respectively. When taking into consideration age at diagnosis, the ICER for etanercept ranged from $US84 129 to $US96 225 per QALY. In considering males for the base-case age at diagnosis, the ICER for etanercept versus methotrexate was $US85 100 per QALY. The average lifetime cost across all treatment arms in a woman diagnosed between 55 and 60 years of age was $US211 702.
Conclusion
While these ICERs cannot be used to directly compare one biological agent with another since there are no comparative trials, they do provide a valid comparison versus methotrexate as first-line agents. Depending where the cost-effectiveness threshold is drawn (i.e. whether it is considered to be $US50 000 or $US100 000 per QALY), etanercept and adalimumab may be considered relatively cost-effective first-line treatments for RA compared with methotrexate monotherapy.





Similar content being viewed by others
References
American College of Rheumatology. Guidelines for the management of rheumatoid arthritis: 2002 update. Arthritis Rheum 2002; 46 (2): 328–346
Hochberg M. Adult and juvenile rheumatoid arthritis: current epidemiologic concepts. Epidemiol Rev 1981; 3: 27–44
Cooper N. Economic burden of rheumatoid arthritis: a systematic review. Rheumatology 2000; 39: 28–33
Jobanputra P, Barton P, Bryan S, et al. The clinical effectiveness and cost-effectiveness of new drug treatments for rheumatoid arthritis: etanercept and infliximab. 2001 Sep 3 [online]. Available from URL: http://www.nice.org.uk/page.aspx?o=29675 [Accessed 2006 Sep 6]
Arnett F, Edworthy S, Bloch D. The American Rheumatology Association 1987 revised criteria for classification or rheumatoid arthritis. Arthritis Rheum 1988 Mar; 31 (3): 315–324
MacGregor A, Schneider H, Rigby A, et al. Characterizing the quantitative genetic contribution to rheumatoid arthritis using data from twins. Arthritis Rheum 2000; 43: 30–37
Barton A, John S, Ollier W, et al. Association between rheumatoid arthritis and polymorphisms of tumor necrosis factor II, but not tumor necrosis factor in Caucasians. Arthritis Rheum 2001; 44: 61–65
Silman A. Epidemiology and rheumatic diseases. In: Maddison P, Isenburg D, Woo P, et al., editors. Oxford textbook of rheumatology. Oxford: Oxford University Press, 1998: 811–828
Maini R, Feldman M. Immunopathogenesis of rheumatoid arthritis. In: Maddison P, Isenburg D, Woo P, Glass D, editors. Oxford textbook of rheumatology. Oxford: Oxford University Press, 1998: 983–1004
Boyce E. Rheumatoid arthritis. In: Herfindal E, Gourley D, editors. Textbook of therapeutics: drug and disease management. 7th ed. Philadelphia (PA): Lippincott Williams & Wilkins, 2000: 641–666
Lotz M. Cytokines and their receptors. In: Koopman W, editor. Arthritis and allied conditions: a textbook of rheumatology. Philadelphia (PA): Lippincott Williams & Wilkins, 1996: 439–478
Van der Heide A, Jacobs J, Bijlsma W, et al. The effectiveness of early treatment with “second-line” anti-rheumatic drugs. Ann Intern Med 1996; 124: 699–707
Piascik P. New antibody approved for the treatment of rheumatoid arthritis. J Am Pharm Assoc 2003; 43 (2): 327–328
Breedveld FC, Weisman MH, Kavanaugh AF, et al. The efficacy and safety of adalimumab (Humira®) plus methotrexate vs adalimumab alone or methotrexate alone in the early treatment of rheumatoid arthritis (RA): 1- and 2-year results of the PREMIER study [OP0013]. The Annual European Congress of Rheumatology; 2005 Jun 8–11; Vienna
St Clair W, van der Heijde D, Smolen J, et al. Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: a randomized, controlled trial. Arthritis Rheum 2004; 50 (11): 3432–3443
Genovese MC. Etanercept vs MTX in patients with early RA. Arthritis Rheum 2002; 46 (8): 1443–1450
Cohen M, Keystone E, Weaver A. The role of biologics in optimizing RA treatment: a return to monotherapy? Rheumatology CME symposium; 2001 Nov 11–15; San Francisco (CA)
First DataBank’s national drug data file plus (NDDF Plus) [electronic database]. San Bruno (CA): First DataBank, 2005 Oct
Express scripts drug trend 2002 report. Maryland Heights (MO): Express Scripts, Inc., 2003 Jun
Physicians’s fee and coding guide. 16th ed. Duluth (GA): MAG Mutual Healthcare Solutions Inc., 2005
Jobanputra P, Barton P. The effectiveness of infliximab and etanercept for the treatment of rheumatoid arthritis: a systematic review and economic evaluation. Health Technol Assess 2002; 6 (21): 1–99
Healthcare Cost and Utilization Project (HCUP). 2003 inpatient data. Rockville (MD): Agency for Healthcare Research and Quality, 2003
Wolfe F, Michaud K. Lymphoma in rheumatoid arthritis: the effect of methotrexate and anti-tumor necrosis factor therapy in 18,572 patients. Arthritis Rheum 2004; 50 (6): 1740–1751
Michaud K, Messer J, Choi HK, et al. Direct medical costs and their predictors in patients with rheumatoid arthritis: a threeyear study of 7527 patients. Arthritis Rheum 2003; 48 (10): 2750–2762
Yelin E, Wanke L. An assessment of annual and long-term direct costs of RA. Arthritis Rheum 1999; 42: 1209–1218
Gold M, Russel L, Siegel J, et al. Cost-effectiveness in health and medicine. New York: Oxford Press, 1996
Kobelt G. Modeling the progression of rheumatoid arthritis: a two-country model to estimate costs and consequences of rheumatoid arthritis. Arthritis Rheum 2002; 46 (9): 2310–2319
Kobelt G, Eberhardt K, Jonsson L, et al. Economic consequences of the progression of rheumatoid arthritis in Sweden. Arthritis Rheum 1999; 42 (2): 347–356
Kobelt G, Jonsson L, Young A, et al. The cost-effectiveness of infliximab (Remicade) in the treatment of rheumatoid arthritis in Sweden and the United Kingdom based on the attract study. Rheumatology 2003; 42: 326–335
Clark W, Jobanputra P, Barton P, et al. The clinical and cost-effectiveness of anakinra for the treatment of rheumatoid arthritis in adults. 2003 Jan [online]. Available from URL: http://www.nice.org.uk/page.aspx?o=65574 [Accessed 2006 Sep 6?
Breedveld FC, Weismann MH, Kavanaugh AF, et al. A multicenter, randomized, double-blind clinical trial of combination therapy with adalimumab plus methotrexate versus methotrexate alone or adalimumab alone in patients with early, aggressive rheumatoid arthritis who had not had previous methotrexate treatment. Arthritis Rheum 2006; 54 (1): 26–37
Hay J. Evaluation and review of pharmacoeconomic models. Expert Opin Pharmacother 2004; 5 (9): 1867–1880
Keystone E, Kavanaugh A, Sharp J, et al. Radiographic, clinical, and functional outcomes of treatment with adalimumab (a human anti-tumor necrosis factor monoclonal antibody) in patients with active rheumatoid arthritis receiving concomitant methotrexate therapy: a randomized, placebo-controlled, 52-week trial. Arthritis Rheum 2004; 50 (5): 1400–1411
Klareskog L, Heijde DVD, Jager JPD, et al. Therapeutic effect of the combination of etanercept and methotrexate compared with each treatment alone in patients with rheumatoid arthritis: double-blind randomised controlled trial. Lancet 2004; 363: 675–681
Lipsky PE, van der Heijde D, St Clair EW, et al. Infliximab and methotrexate in the treatment of rheumatoid arthritis. N Engl J Med 2000; 343: 1594–1602
van de Putte LBA, Atkins C, Malaise M, et al. Efficacy and safety of adalimumab as monotherapy in patients with rheumatoid arthritis for whom previous disease modifying antirheumatic drug treatment has failed. Ann Rheum Dis 2005; 63: 508–516
Weinblatt M. Adalimumab, a fully human anti-tumor necrosis factor alpha monoclonal antibody, for the treatment of RA in patients taking concomitant MTX. Arthritis Rheum 2003; 48 (1): 35–45
Felson D, Anderson J, Meenen R. Use of short-term efficacy/toxicity tradeoffs to select second-line drugs in rheumatoid arthritis: a meta-analysis of published clinical trials. Arthritis Rheum 1992; 35: 1117–1125
Wolfe F. The mortality of rheumatoid arthritis. Arthritis Rheum 1994; 37 (4): 481–494
Young A, Dixey J, Kulinskaya E, et al. Which patients stop working because of rheumatoid arthritis? Results of five years’ follow up in 732 patients from the Early RA Study (ERAS). Ann Rheum Dis 2002; 61: 335–340
Employment and earnings report, January 2005. Washington, DC: Bureau of Labor Statistics, 2005 Sep 21 [online]. Available from URL: http://www.bls.gov/ncs/ect/home.htm [Accessed 2006 Sep 6]
Yelin E, Trupin L, Katz P, et al. Association between etanercept use and employment outcomes among patients with rheumatoid arthritis. Arthritis Rheum 2003; 48 (11): 3046–3054
Brennan A, Bansback N, Reynolds A, et al. Modelling the cost-effectiveness of etanercept in adults with rheumatoid arthritis in the UK. Rheumatology 2004; 42: 62–72
Bansback NJ, Regier DA, Ara R, et al. An overview of economic evaluations for drugs used in rheumatoid arthritis. Drugs 2005; 65 (4): 473–496
Viller F, Guillemin F. Compliance to drug treatment of patients with rheumatoid arthritis: a 3-year longitudinal study. J Rheumatol 1999; 26: 2114–2122
Ollendorf DA, Massarotti E, Birbara C, et al. Frequency, predictors, and economic impact of upward dose adjustment of infliximab in managed care patients with rheumatoid arthritis. J Manag Care Pharm 2005; 11 (5): 383–393
Wolfe F, Michaud K, Anderson J, et al. Tuberculosis infection in patients with rheumatoid arthritis and the effect of infliximab therapy. Arthritis Rheum 2004; 50 (2): 372–379
Gabriel S, Crowson C, Luthra H, et al. Modeling the lifetime costs of rheumatoid arthritis. J Rheumatol 1999; 26: 1269–1274
Wolfe F, Zwillich S. Long-term outcomes of rheumatoid arthritis: a 23-year prospective, longitudinal study of total joint replacement and its predictors in 1600 patients with rheumatoid arthritis. Arthritis Rheum 1998; 41 (6): 1072–1082
van Vollenhoven R. Switching between biological agents. Clin Exp Rheumatol 2004; 22 Suppl. 35: S115–S121
Wong J, Singh G, Kavanaugh A. Estimating the cost-effectiveness of 54 weeks of infliximab for rheumatoid arthritis. Am J Med 2002; 113 (5): 400–408
Acknowledgements
This study was conducted while Dr Spalding was a graduate student at the University of Southern California, Los Angeles. Funding for this study was provided solely by the University of Southern California. Both authors made significant contributions to the intellectual content of the manuscript. Dr Hay has previously consulted with Abbott Laboratories and Dr Spalding has previously worked for a company that awarded Abbott stock as part of its stock option package.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Spalding, J.R., Hay, J. Cost Effectiveness of Tumour Necrosis Factor-α Inhibitors as First-Line Agents in Rheumatoid Arthritis. Pharmacoeconomics 24, 1221–1232 (2006). https://doi.org/10.2165/00019053-200624120-00006
Published:
Issue Date:
DOI: https://doi.org/10.2165/00019053-200624120-00006