Summary
Synopsis
l-Carnitine 1 occurs naturally as an essential cofactor of fatty acid metabolism which is synthesised endogenously or obtained from dietary sources. In patients with primary carnitine deficiencies, which may be life-threatening, and some secondary deficiencies such as organic acidurias, the exogenously administered compound is clearly beneficial: by abolishing hypotonia, motor skills are improved, as are muscle weakness and wasting. In preliminary clinical trials in patients with ischaemic cardiac disease, therapy with l-carnitine has shown beneficial effects on myocardial function and metabolism and has improved exercise tolerance in patients with angina pectoris — findings which require further substantiation in larger controlled studies. Moreover, while some interesting evidence suggests that l-carnitine may find potential use in such diverse conditions as carnitine deficiencies secondary to prolonged total parenteral nutrition supplementation or chronic haemodialysis, hyperlipidaemias and the prevention of toxicity induced by anthracyclines and valproate, such findings must be regarded as preliminary. Exogenously administered l-carnitinine is very well tolerated.
Thus, while its role in primary deficiencies is established, with its profile of negligible toxicity l-carnitine is worthy of further investigation to more clearly define its therapeutic applications in a variety of conditions which may be indirectly related to alterations in fatty acid metabolism.
Role of l-Carnitine in Fatty Acid Metabolism
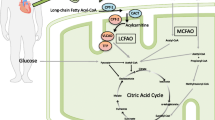
l-Carnitine is a cofactor of several enzymes (carnitine translocase, acylcarnitine transferases I and II) necessary for the transformation of free long chain fatty acids to acylcarnitines, and their transport into the mitochondrial matrix. β-Oxidation of these compounds precedes their entry into the Krebs cycle, where energy production occurs. In the absence of l-carnitine, the accumulation of free fatty acids in the cytoplasm produces a toxic effect on the cell, and an energy deficit arises from the unavailability of fatty acids within the mitochondria.
Pharmacokinetic Studies
The pharmacokinetic properties of exogenously administered l-carnitine have not been thoroughly described. Peak plasma concentrations of free carnitine of 48.5 and 69 µmol/L have been attained 3.5 to 5 hours following single oral 500mg and 2g doses, respectively. l-Carnitine is actively transported into tissues via a saturable system, although passive diffusion also occurs. The volume of distribution is about 26% of bodyweight. The compound is likely not metabolised in humans (other than by partial conversion to acylcarnitine esters) and thus is eliminated via the kidneys. The portion of a dose of l-carnitine excreted in the urine within 24 hours varies depending on the route of administration. Thus, following an intravenous dose 80% has been recovered, in contrast to 7% of a dose recovered within 24 hours following an oral dose. Faecal elimination accounts for less than 2% of a dose. In healthy volunteers, the half-life of l-carnitine ranges from 2 to 15 hours.
Use in Patients with Ischaemic Cardiac Disease
The rationale for the use of l-carnitine in patients with ischaemic heart disease relates to the premise that increased lactate production and decreased energy output of cardiac muscle in such patients result in part from decreased l-carnitine concentrations in heart muscle, which has been demonstrated at necropsy in the necrotic area and the infarct border tissue of patients who died of myocardial infarction. In single dose studies in patients with ischaemic heart disease, l-carnitine 40 or 140 mg/kg intravenously or 1g daily orally, and dl-carnitine 20 or 40 mg/kg intravenously, decreased lactate production and increased the free fatty acid extraction ratio in the myocardium. Orally administered l-carnitine decreased the release of creatine kinase isoenzyme MB fraction in patients with acute myocardial infarction.
In patients with moderately impaired left ventricular function, intravenously administered l-carnitine 40 mg/kg exerted a positive inotropic effect as evidenced by a decrease in the left ventricular diastolic pressure and the pre-ejection period/left ventricular ejection time ratio. These effects were confirmed in patients treated with l-carnitine 2g intravenously daily for 10 days.
Preliminary trials in patients with chronic stable angina pectoris have indicated that orally administered l-carnitine 900 to 2000mg daily improved exercise tolerance, lessened the extent of ST segment depression, decreased the frequency of anginal attacks and reduced glyceryl trinitrate (nitroglycerin) consumption. These findings are supported by the results of an open multicentre study in more than 1000 patients with angina pectoris, which demonstrated that a reduction was obtained in the consumption of traditional antianginal drug therapies and the frequency of anginal attacks during the 12-month period of supplementation with oral l-carnitine 2g daily.
While some interesting evidence suggests that l-carnitine may be of benefit in the treatment of patients with acute myocardial infarction or arrhythmias, its role as an adjuvant in therapy of these conditions awaits definition in further studies.
Effects of l-Carnitine in Primary Deficiency
Concentrations of l-carnitine are below normal in the plasma, liver and muscle of patients with systemic carnitine deficiencies (SCD), and in the muscle of those with the myopathic form, possibly because of impaired transport or biosynthesis of l-carnitine. Replacement therapy with the drug in regimens of up to 4g daily reverses such clinical symptoms as severe muscular weakness, hypoglycaemia and intellectual retardation seen in patients more severely afflicted with SCD, and can be life-saving in some instances. Similarly, in patients with the less debilitating myopathic deficiency there has been objective and subjective evidence of improved muscle strength within 1 week of the commencement of l-carnitine therapy.
Use in Secondary Deficiencies
l-Carnitine has been shown to be of benefit in children with defects of intermediary metabolism (organic acidurias) who display impaired motor skills and low plasma concentrations of carnitine.
There is some evidence to indicate that the elevated plasma concentrations of triglycerides and total cholesterol in patients undergoing chronic intermittent haemodialysis can be decreased by l-carnitine supplementation, whether administered orally, intravenously or added to the dialysate. However, results of those studies that have included a placebo control have varied, and in view of the paradoxical increase in plasma triglycerides associated with high oral doses of l-carnitine (3g daily) in 1 trial, further studies are required to determine the optimum dose in these patients.
A few blind and non-blind studies in small numbers of patients with symptoms of the ‘post-dialysis syndrome’ have demonstrated symptomatic improvement after 2 or more months’ treatment with l-carnitine 990 to 2000mg daily, although objective measurements in one study did not alter significantly.
The addition of l-carnitine to total parenteral nutrition regimens in neonates has increased plasma concentrations of total and free carnitine which are lower than in infants fed by enterai methods. Some evidence indicates that carnitine supplementation results in better metabolism of intravenously administered fat emulsion, especially in premature infants.
A few uncontrolled studies in patients who had received anthracycline therapy in cumulative doses of less than 500 mg/m2, suggest that orally or intravenously administered l-carnitine may decrease the severity of cardiotoxicity associated with daunorubicin or doxorubicin administration.
Side Effects
l-Carnitine is very well tolerated; at doses of up to 15g daily no side effects have occurred other than infrequent diarrhoea, gastralgia and nausea. A symptom similar to myasthenia gravis which has been reported with the administration of dl-carnitine has not been seen with l-carnitine therapy.
Dosage
The usual dose of l-carnitine in adults with primary or secondary carnitine deficiencies or ischaemic cardiomyopathies is 1g given orally or parenterally 1 to 3 times daily. Children should receive 50 to 100 mg/kg daily in 2 to 3 divided doses.
Similar content being viewed by others
References
Adamo S, Tonon M, Carrara M, Sartori G, Ghidini O. Effect of carnitine on serum triglycerides in alcoholism: a double-blind placebo controlled trial. In Borum (Ed.) Clinical aspects of human carnitine deficiency, p.244, Pergamon Press, New York, 1986
Albertazzi A, Capelli P, Di Paolo B, Pola P, Tondi P, et al. Endocrine-metabolic effects of l-carnitine in patients on regular dialysis treatment. Proceedings of the EDTA 19: 302–307, 1982
Alberts DS, Peng Y-M, Moon TE. Carnitine prevention of adri-amycin in toxicity in mice. Biomedicine 29: 265–267, 1978
Almog C, Fried K, Reif R, Zieghelboim J, Lewinsohn G. Autosomal recessive lipid storage myopathy (probable carnitine deficiency). Journal of Medical Genetics 16: 435–438, 1979
Angelini C, Lucke S, Cantarutti F. Carnitine deficiency of skeletal muscle: report of a treated case. Neurology 26: 633–637, 1976
Angelini C, Vergani L, Costa L, Martinuzzi A, Dünner E, et al. Clinical study of efficacy of l-carnitine and metabolic observations in exercise physiology. In Borum (Ed.) Clinical aspects of human carnitine deficiency, p. 38, Pergamon Press, New York, 1986
Ashbrook DW. Carnitine supplementation in human carnitine deficiency. In Borum (Eds) Clinical aspects of human carnitine deficiency, pp. 120–134, Pergamon Press, New York, 1986
Aubia J, Masramon J, Lhoveras J, Llorach M, Andolz P. Carnitine in haemodialysis patients. Lancet 2: 1028, 1980
Bach AC. Carnitine in human nutrition. Zeitschrift für Ernährungswissenschaft 21: 257–265, 1982
Bach AC, Schirardin H, Sihr M-O, Storck D. Free and total carnitine in human serum after oral ingestion of l-carnitine. Diabete and Metabolisme (Paris) 9: 121–124, 1983
Balogh D, Hackl JM, Legenstein E, Musil HE. Erfahrungen mit 1-camitin in der post-aggressions phase. Infusion therapie 13: 204–208, 1986
Bartel LL. Hussey JL, Shrago E. Perturbation of serum carnitine levels in human adults by chronic renal disease and dialysis therapy. American Journal of Clinical Nutrition 34: 1314–1320, 1981
Barth PG, Scholte HR, Berden JA, van der Klei-van Moorsei JM, Luyt-Houwen IE, et al. An X-linked mitochondrial disease affecting cardiac muscle, skeletal muscle and neutrophil leucocytes. Journal of the Neurological Sciences 62: 327–355, 1983
Bazzato G, Coli U, Landini S, Mezzina C, Ciman M. Myasthenialike syndrome after dl- but not l-carnitine. Lancet 1: 1209, 1981
Bellinghieri G, Savica V, Mallama A, Di Stefano C, Consolo F, et al. Correlation between increased serum and tissue l-carnitine levels and improved muscle symptoms in hemodialyzed physiokinesiotherapy in chronic respiratory insufficiency. Clinical Trials Journal 4: 353–360, 1985
Dal Negro R, Zoccatelli O, Pomari G, Turco P. Changes in physical performance of untrained volunteers: effects of l-carnitine. Clinical Trials Journal 23: 242–248, 1986b
De Grandis D, Mezzina C, Fiaschi A, Pinelli P, Bazzato G, et al. Myasthenia due to carnitine treatment. Journal of the Neurological Sciences 46: 365–371, 1980
De Leonardis V, Neri B, Baculli S, Cinelli P. Reduction of cardiac toxicity of anthracyclines by l-carnitine: preliminary overview of clinical data. International Journal of Clinical Pharmacology Research 5: 137–142, 1985
Del-Valle JA, Martinez-Pardo M, Ludena C, Camarero C, Del-Olmo R, et al. A new patient with dicarboxylic aciduria presenting as Reye’s syndrome. 21st Annual Symposium of the Society for the Study of Inborn Errors of Metabolism, Lyon, September 6–9, 1983
Di Donato S, Rimoldi M, Comelio F, Bottacchi E, Giunta A. Evidence for autosomal recessive inheritance in systemic carnitine deficiency. Annals of Neurology 11: 190–192, 1982
Di Donato S, Pelucchetti P, Rimoldi M, Mora M, Garavaglia B, et al. Systemic carnitine deficiency: clinical, biochemical, and morphological cure with l-carnitine. Neurology 34: 157–162, 1984a
Di Donato S, Rimoldi M, Garavaglia B, Uziel G. Propionylcarnitine excretion in propionic acid and methylmalonic acidurias: a cause of carnitine deficiency. Clinica Chimica Acta 139: 13–21, 1984b
Di Marco V, Ginardi V, Dell’aira E, Abbate M, Costanza MG, et al. La camitina nel trattamento dei crampi muscolari nei pazienti in emodialisi periodica. Policlinico Sez. Med. 89: 104–110, 1982
Donatelli M, Terrizzi C, Zummo G, Russo V, Bucalo ML, et al. Effects of l-carnitine on chronic anaemia and erythrocyte adenosine triphosphate (ATP) concentration in haemodialysed patients. Current Therapeutic Research, in press, 1987
Engel AG, Angelini C. Carnitine deficiency of human muscle with associated lipid storage myopathy — a new syndrome. Science 179: 899–902, 1973
Engel AG, Rebouche CJ, Wilson DM, Glasgow AM, Romshe CA et al. Primary systemic carnitine deficiency. II. Renal handling of carnitine. Neurology 31: 819–825, 1981
Etzioni A, Levy J, Nitzan M, Erde P, Benderly A. Systemic carnitine deficiency exacerbated by a strict vegetarian diet. Archives of Disease in Childhood 59: 177–179, 1984
Fagher B, Cederblad G, Monti M, Olsson L, Rasmussen B, et al. Carnitine and left ventricular function in haemodialysis patients. Scandinavian Journal of Clinical and Laboratory Investigation 45: 193–198, 1985a
Fagher B, Cederblad G, Eriksson M, Monti M, Moritz U, et al. l-Carnitine and haemodialysis: double-blind study on muscle function and metabolism and peripheral nerve function. Scandanavian Journal of Clinical and Laboratory Investigation 45: 169–178, 1985b
Fernandez C, La Menza B, Pola P. Trials clinici difase IV e di farmacovigilanza-nuove ipotesi metodologiche-il terzo trial Ance. Journal of the American Medical Association (Italian Edition) 2: Number 9, 1985
Ferrari R, Cucchini F, DiLisa F, Raddino R, Bolognesi R, et al. The effect of l-carnitine (camitene) on myocardial metabolism of patients with coronary artery disease. Clinical Trials Journal 21: 40–58, 1984
Frohlich J, Seccombe DW, Hahn P, Dodek P, Hynie I. Effect of fasting on free and esterified carnitine levels in human serum and urine: correlation with serum levels of free fatty acids and β-hydroxybutyrate. Metabolism 27: 555–561, 1978
Furitano G, Patema S, Perricone R, Barbarino C, Palumbo FP, et al. Polygraphic evaluation of effects of carnitine in patients on adriamycin treatment. Drugs Under Experimental and Clinical Research 10: 107–111, 1984
Garzya G, Amico RM. Comparative study on the activity of racemic and laevorotatory carnitine in stable angina pectoris. International Journal of Tissue Reactions 2: 175–180, 1980
Geber N, Dickinson RG, Harland RC, Lynn RK, Houghton D, et al. Reye-like syndrome associated with valproic acid. Journal of Pediatrics 95: 142, 1979
Gilly R, Carrier H, Lamit J. Myopathie congénitale avec surcharge de la fibre musculaire en lipids et glycogene et déficit partiel en carnitine. Annals de Pédiatre 27: 319–324, 1980
Giordano MP, Corsi M, Roncarolo P, Falcone M, Gabasio C. Effect of l-carnitine on systolic time intervals in coronary artery disease. Current Therapeutic Research 33: 305–311, 1983
Glasgow AM, Eng G, Engel AG. Systemic carnitine deficiency simulating recurrent Reye syndrome. Journal of Pediatrics 96: 889–891, 1980
Glasgow AM, Engel AG, Bier DM, Perry LW, Dickie M, et al. Hypoglycemia, hepatic dysfunction, muscle weakness, cardiomyopathy, free carnitine deficiency and long-chain acylcamitine excess responsive to medium chain triglyceride diet. Pediatric Research 17: 319–326, 1983
Gross CJ, Henderson LM. Absorption of D and L-camitine by the intestine and kidney tubule in the rat. Biochimica et Biophysica Acta 772: 209–219, 1984
Hahn P, Allardyce DB, Frohlich J. Plasma carnitine levels during total parenteral nutrition of adult surgical patients. American Journal of Clinical Nutrition 36: 569–572, 1982
Hamilton JW, Li BU, Shug AL, Olsen WA. Carnitine transport in human intestinal biopsy specimens. Gastroenterology 91: 10–16, 1986
Helms RA, Whitington PF, Mauer EC, Catarou EM, Christensen ML, et al. Enhanced lipid utilization in infants receiving oral l-carnitine during long-term parenteral nutrition. Journal of Pediatrics 109: 984–988, 1986
Howard LJ, Beckerman AH. A case history of myopathic carnitine deficiency benefited by glucocorticoids and l-carnitine supplementation. Drug-Nutrient Interactions 3: 191–196, 1985
Kamikawa T, Suzuki Y, Kobayashi A, Hayashi H, Masumura Y, et al. Effects of l-carnitine on exercise tolerance in patients with stable angina pectoris. Japanese Heart Journal 25: 587–597, 1984
Kerr P, Shurin S, Tsemg K-Y, Hoppel C. Metabolic effects of treatment of systemic carnitine deficiency. Pediatric Research 15: 633, 1981
Kudoh Y, Shoji T, Oimatsu H, Yoshida S, Kikuchi K, et al. The role of l-carnitine in the pathogenesis of cardiomegaly in patients with chronic hemodialysis. Japanese Circulation Journal 47: 1391–1397, 1983
Kudoh Y, Shoji T, Oimatsu H, Kikuchi K, Iimura O, et al. Plasma l-carnitine in patients with chronic hemodialysis — pharmacokinetics of l-carnitine and its replacement therapy in these patients. Nippon Jimzo Gakki Shi 26: 57–64, 1984
Labonia WD, Morelli Jr OH, Gimenez ML, Freuler PV, Morelli OH. Effects of l-carnitine on sodium transport in erythrocytes from dialyzed uremic patients. VI Argentinian Congress of Nephrology, Buenos Aires, November 12–15, 1986
Leschke M, Rumpf W, Eisenhauer T, Fuchs C, Becker K, et al. Quantitative assessment of carnitine loss during hemodialysis and hemofiltration. Kidney International 24(Suppl. 16): S143–S146, 1983
Liedtke AJ. Metabolism of the ischemic heart: alterations in fatty acid intermediates and role of carnitine. In Kaiser et al. (Eds) Carnitine — its role in lung and heart disorders, pp. 100–111, Karger, Munich, 1987
Logue RB. Metabolic regulation and myocardial function. In Logue et al. (Eds) The heart, 6th ed., pp.91–94, McGraw-Hill, New York, 1985a
Logue RB. Metabolic regulation and myocardial function. In Logue et al. (Eds) The heart, 6th ed., pp.568–569, McGraw-Hill, New York, 1985b
Logue RB. Metabolic regulation and myocardial function. In Logue et al. (Eds) The heart, 6th ed., pp.653–654, McGraw-Hill, New York, 1985c
Maccari F, Ramacci MT. Antagonism of doxorubicin cardiotoxicity by camidine is specific of the 1-diasteriosomer. Biomedicine 35: 65–67, 1981
Maebashi M, Kawamura N, Sato M, Yoshinaga K, Suzuki M. Urinary excretion of carnitine in man. Journal of Laboratory and Clinical Medicine 87: 760–766, 1976
Maebashi M, Imamura A, Yoshinaga K. Effect of aging on lipid and carnitine metabolism. Tohoku Journal of Experimental Medicine 138: 231–236, 1982
Maingourd Y, Goldfarb A, Bieth R, Caron FM, Gontier MF, et al. Myocardiopathie familiale par déficit en carnitine. Archives des maladies du Coeur et des Vaisseaux 79: 1650–1654, 1986
Marconi C, Sassi G, Carpinelli A, Cerretelli P. Effects of l-carnitine loading on the aerobic and anaerobic performance of endurance athletes. European Journal of Applied Physiology 54: 131–135, 1985
Markesberry WR, McQuillen MP, Protopia PG, Harrison AR, Engel AG. Muscle carnitine deficiency. Archives of Neurology 31: 320–324, 1974
Martin P, Carrier H, Renoud JF, Kullmann B, Delpont E, et al. Un cas de myopathie avec déficit en carnitine. Revue Neurologique (Paris) 142: 625–634, 1986
Matsuishi T, Hirata K, Terasawa K, Kato H, Yoshino M, et al. Successful carnitine treatment in two siblings having lipid storage myopathy with hypertrophic cardiomyopathy. Neuropediatrics 16: 6–12, 1985
McGarry JD, Foster DW. An improved and simplified radioisotopic assay for the determination of free and esterified carnitine. Journal of Lipid Research 17: 277, 1976
Mehlman MA, Kader MM, Therriault DG. Metabolism, turnover time, half-life, body pool of carnitine-14C in normal, alloxan diabetic and insulin treated rats. Life Sciences 8: 465–472, 1969
Mioli V, Tarchini R, Boggi R. Use of dl- and l-carnitine in uraemic patients on intermittent haemodialysis. International Journal of Clinical Pharmacology Research 2: 143–148, 1982
Nanni G, Pittiruti M, Castagneto M. Carnitine plasma levels during total parenteral nutrition. American Journal of Clinical Nutrition 38: 339–341, 1983
Neri B, Cini-Neri G, Bartalucci S, Bandinelli M. Protective effect of l-carnitine on cardiac metabolic damage induced by doxorubicin in vitro. Anticancer Research 6: 659–662, 1986
Neri B, Torcia MG, Comparini T, Guidi S, Miliani A, et al. Creatin kinase-MB: a non-invasive test monitoring acute adriamycin and daunomycin cardiotoxicity. Journal of Experimental and Clinical Cancer Research 2: 41–45, 1983a
Neri B, Comparini T, Miliani A, Torcia M. Protective effects of l-carnitine (camitene) on acute adriamycin and daunomycin cardiotoxicity in cancer patients. Clinical Trials Journal 20: 98–103, 1983b
Nilsson-Ehle P, Cederblad G, Fagher B, Monti M, Thysell H. Plasma lipoproteins, liver function and glucose metabolism in haemodialysis patients: lack of effect of 1-carnitine supplementation. Scandanavian Journal of Clinical and Laboratory Investigation 45: 179–184, 1985
Noto R, Maugeri A, Grasso R, Sfogliano L. Free fatty acids and carnitine in patients with liver disease. Current Therapeutic Research 40: 35–39, 1986
Novak M, Monkus EF, Chung D, Buch M. Carnitine in the perinatal metabolism of lipids 1. Relationship between maternal and fetal plasma levels of carnitine and arylcamitines. Pediatrics 67: 95–100, 1981
Ohtani Y, Endo F, Matsuda I. Carnitine deficiency and hyperammonemia associated with valproic acid therapy. Journal of Pediatrics 101: 782–785, 1982
Opie LH. Role of carnitine in fatty acid metabolism of normal and ischemic myocardium. American Heart Journal 97: 375–388, 1979
Orzali A, Maetzke G, Donzelli F, Rubaltelli FF. Effect of carnitine on lipid metabolism in the neonate. II. Carnitine addition to lipid infusion during prolonged total parenteral nutrition. Journal of Pediatrics 104: 436–440, 1984
Parker D, Root AW, Schimmel S, Andriola M, DiMauro S. Encephalopathy and fatal myopathy in two siblings. American Journal of Diseases of Children 136: 598–601, 1982
Pelligrini G, Scarlato G, Moggio M. A hereditary case of lipid storage myopathy with carnitine deficiency. Journal of Neurology 223: 73–84, 1980
Penn D, Schmidt-Sommerfeld E, Pascu F. Decreased tissue carnitine concentrations in newborn infants receiving total parenteral nutrition. Journal of Pediatrics 98: 976–978, 1981
Penn D, Wolf H, Schmidt-Sommerfeld E. Possible carnitine deficiency in parenterally alimented newborn infants. Acta Paediatrica Scandinavica Suppl. 296: 113–114, 1982
Phillips GD, Odgers CL. Parenteral Nutrition: current status and concepts. Drugs 23: 276–323, 1982
Pola P, Savi L, Grilli M, Flore R, Serricchio M. Carnitine in the therapy of dyslipidemie patients. Current Therapeutic Research 27: 208–216, 1979
Pola P, Tondi P, Dal Lago A, Sericchio M, Flore R. Statistical evaluation of long-term l-carnitine therapy in hyperlipoproteinaemias. Drugs under Experimental and Clinical Research 12: 925–935, 1983
Pongratz D, Hübner G, Deufel T, Wieland O, Pongratz E, et al. Klinische, morphologische und biochemische befunde bei carnitinmangelmyopathien. Klinische Wochenschrift 57: 927–936, 1979
Rebouche CJ, Engel AG. Carnitine metabolism and deficiency syndromes. Mayo Clinic Proceedings 58: 533–540, 1983
Rebouche CJ, Engel AG. Kinetic compartmental analysis of carnitine metabolism in the human carnitine deficiency syndromes. Journal of Clinical Investigation 73: 857–867, 1984
Rebouche CJ, Mack DL, Edmonson PF. L-camitine dissimilation in the gastrointestinal tract of the rat. Biochemistry 23: 6422–6426, 1984
Rebuzzi AG, Schiavoni G, Amico CM, Montenero AS, Meo F, et al. Beneficial effects of l-carnitine in the reduction of the necrotic area in acute myocardial infarction. Drugs Under Experimental and Clinical Research 10: 219–223, 1984
Reforzo G, De Andreis Bessone PL, Rebaudo F, Tibaldi M. Evaluation of the effects of high doses of l-carnitine on the myocardial lactate balance during pacing-induced ischemia in ageing subjects. Current Therapeutic Research, in press, 1986
Rocchi L, Feola I, Calvani M, D’ladio S, Alfarone C, Frascarelli M. Effects of carnitine administration in patients with chronic renal failure undergoing periodic dialysis, evaluated by computerized electromyography. Drugs Under Experimental and Clinical Research 12: 707–711, 1986
Rodriguez-Segade S, Alonso de la Peña C, Paz JM, Novoa D, et al. Carnitine deficiency in haemodialysed patients. Clinica Chimica Acta 159: 249–256, 1986
Roe CR, Millington DS, Maitby DA, Bohan TP, Hoppel CL. L-camitine enhances excretion of propionyl coenzyme A as propionylcamitine in propionic acidemia. Journal of Clinical Investigation 73: 1785–1788, 1984
Roe CR, Hoppel CL, Stacey TE, Chalmers RA, Tracey BM, et al. Metabolic response to carnitine in methylmalonic aciduria. Archives of Disease in Childhood 58: 916–920, 1983
Rossi CS, Siliprandi N. Effect of carnitine on serum HDL-cholesterol: report of two cases. Johns Hopkins Medical Journal 150: 51–54, 1982
Rovamo L. Postheparin plasma lipases and carnitine in infants during parenteral nutrition. Pediatric Research 19: 292–297, 1985
Rubecz I, Sándor A, Hamar A, Mestyán J. Blood levels of total carnitine and lipid utilization with and without carnitine supplementation in newborn infants. Acta Paediatrica Hungarica 25: 165–171, 1984
Sachan DS, Rhew TH, Ruark RA. Ameliorating effects of carnitine and its precursors on alcohol-induced fatty liver. American Journal of Clinical Nutrition 39: 738–744, 1984
Sandstedt S, Larsson J, Cederblad G. Carnitine levels in skeletal muscle of malnourished patients before and after total parenteral nutrition. Clinical Nutrition 5: 227–230, 1986
Sawicka E, Ketelsen U-P, Petrykowski W, Schmidt-Sommerfeld E. Mitochondrial abnormalities in systemic carnitine deficiency before and after successful treatment with l-carnitine. Fifth International Congress on Neuromuscular Diseases, Marseille, September, 1982
Scarlato G, Pellegrini G, Moggio M, Meola G, Frattola L. Carnitine deficiency and lipid storage myopathy in three patients: analysis of some differential features. Perspectives in Inherited Metabolic Diseases 3: 109–128, 1979
Schiavoni G, Pennestri F, Mongiardo R, Mazzari M, Manzoli V. Cardiodynamic effects of l-carnitine in ischaemic cardiopathy. Drugs Under Experimental and Clinical Research 9: 171–185, 1983
Schiff D, Chan G, Seccombe D, Hahn P. Plasma carnitine levels during intravenous feeding of the neonate. Journal of Pediatrics 95: 1043–1046, 1979
Schmidt-Sommerfeld E, Penn D. Carnitine and neonatal lipid metabolism. Journal of Pediatrics 105: 848, 1984
Schmidt-Sommerfeld E, Penn D, Sodha RJ, Prögler M, Schneider H. Transfer and metabolism of carnitine and carnitine esters in the in vitro perfused placenta. Contraception Gynecology and Obstetrics 13: 141–143, 1985
Schmidt-Sommerfeld E, Penn D, Wolf H. Carnitine deficiency in premature infants receiving total parenteral nutrition: effect of l-carnitine supplementation. Journal of Pediatrics 102: 931–935, 1983
Sciveres G. Esperienza con 1-camitina nel trattamento della angina stabile da sforzo. Archivio di Medicina Interna 36: 61–66, 1984
Scholte HR, Meijer AE, Van Wijngaarden GK, Leenders KL. Familial carnitine deficiency. Journal of the Neurological Sciences 42: 87–101, 1979
Seccombe DW, Snyder F, Parsons HG. L-camitine for methyl-malonicaciduria. Lancet 2: 1401, 1982
Seccombe DW, James L, Booth F. L-camitine treatment in glutaric aciduria type I. Neurology 36: 264–267, 1986
Siliprandi N, DiLisa F, Toninello A. Biochemical derangements in ischemic myocardium: the role of carnitine. G. Ital. Cardiol. 14: 804–808, 1984
Slonim AE, Borum PR, Mrak RE, Najjar J, Richardson D, et al. Nonketotic hypoglycemia: an early indicator of systemic carnitine deficiency. Neurology 33: 29–33, 1983
Snyder TM, Little BW, Roman-Campos G, McQuillen JB. Successful treatment of familial idiopathic lipid storage myopathy with l-carnitine and modified lipid diet Neurology 32: 1106–1115, 1982
Spagnoli LG, Corsi M, Villaschi S, Palmieri G, Maccari F. Myocardial carnitine deficiency in acute myocardial infarction. Lancet 1: 1419–1420, 1982
Stumpf DA, Parker WD, Haas R. Carnitine deficiency with valproate therapy. Journal of Pediatrics 103: 175–176, 1983
Sugimoto T, Nishida N, Woo M, Takeuchi T, Yasuhara A, et al. Serum and urinary carnitine and organic acids in Reye syndrome and Reye-like syndrome. Brain & Development 8: 257–261, 1986
Suzuki M, Kanaya M, Muramatsu S, Takahashi T. Effects of carnitine administration, fasting, and exercise on urinary carnitine excretion in man. Journal of Nutritional Science and Vitaminology 22: 169–174, 1976
Takahashi M, Sawaguchi S. Lipid metabolism in parenterally alimented neonates: carnitine blood concentrations and fat utilization. Indian Journal of Pediatrics 50: 161–168, 1983
Tanphaichitr V, Lerdvuthisopon N. Urinary carnitine excretion in surgical patients on total parenteral nutrition. Journal of Parenteral and Enteral Nutrition 5: 505–509, 1981
Tao RC, Yoshimura NN. Carnitine metabolism and its application in parenteral nutrition. Journal of Parenteral and Enteral Nutrition 4: 469–486, 1980
Thomsen JH, Shug AL, Yap VU, Patel AK, Karras TJ, et al. Improved pacing tolerance of the ischemic human myocardium after administration of carnitine. American Journal of Cardiology 43: 300–306, 1979
Tripp ME, Katcher ML, Peters HA, Gilbert EF, Arya S, et al. Systemic carnitine deficiency presenting as familial endocardial fibroelastosis. New England Journal of Medicine 305: 385–390, 1981
Trovato GM, Ginardi V, Di Marco V, Dell’aira A, Corsi M. Longterm l-carnitine treatment of chronic anaemia of patients with end-stage renal failure. Current Therapeutic Research 31: 1042–1049, 1982
Vacha GM, Giorcelli G, Siliprandi N, Corsi M. Favorable effects of l-carnitine treatment on hypertriglyceridemia in hemodialysis patients: decisive role of low levels of high-density lipoprotein-cholesterol. American Journal of Clinical Nutrition 38: 532–540, 1983
Vacha GM, Corsi M, Giorcelli G, D’iddio S, Maccari F. Serum and muscle l-carnitine levels in hemodialyzed patients during and after long-term l-carnitine treatment. Current Therapeutic Research 37: 505–516, 1985
Vandyke DH, Griggs RC, Markesbery W, DiMauro S. Hereditary carnitine deficiency of muscle. Neurology 25: 154–159, 1975
Vergani L, Bertoli M, Ruffatti A, Romagnoli GF, Angelini C. Treatment with l-carnitine in hemodialysis patients. Abstract no. 168, Third International Congress on Nutrition and Metabolism in Renal Disease, Marseille, September 1–4, 1982
Waber LJ, Valle D, Neill C, DiMauro S, Shug A. Carnitine deficiency presenting as familial cardiomyopathy: a treatable defect in carnitine transport. Journal of Pediatrics 101: 700–705, 1982
Wanner C, Hörl WH. Potential role of carnitine in patients with renal insufficiency. Klinische Wochenschrift 64: 579–586, 1986
Ware AJ, Burton WC, McGarry JD, Marks JF, Weinberg AG. Systemic carnitine deficiency. Report of a fatal case with multisystemic manifestations. Journal of Pediatrics 93: 959–964, 1978
Welling PG, Thomsen JH, Shug AL, Tse FL. Pharmacokinetics of l-carnitine in man following intravenous infusion of dl carnitine. International Journal of Clinical Pharmacology and Biopharmacy 17: 56–60, 1979
Weschler A, Aviram M, Levin M, Better OS, Brook JG. High dose of l-carnitine increases platelet aggregation and plasma triglyceride levels in uremic patients on hemodialysis. Nephron 38: 120–124, 1984
Willner JH, Chutorian AM, DiMauro S. Tissue Carnitine in Reye syndrome. Annals of Neurology 4: 468–469, 1978
Worthley LI, Fishlock RC, Snoswell AM. Carnitine balance and effects of intravenous l-carnitine in two patients receiving long-term total parenteral nutrition. Journal of Parenteral and Enteral Nutrition 8: 717–719, 1984
Young RS, Bergman I, Gang DL. Fetal Reye-like syndrome associated with valproic acid. Annals of Neurology 7: 389, 1980
Author information
Authors and Affiliations
Additional information
Various sections of the manuscript reviewed by: C. Angelini, Universita degli Studi di Padova, Istituto di Clinica delle Malattie Nervose e Mentali, Padova, Italy; P.R. Borum, Institute of Food and Agricultural Sciences, University of Florida, Gainesville, Florida, USA; G. Cederblad, Department of Clinical Chemistry, Karolinska Institutet, Danderyd, Sweden; S. Di Donato, Istituto Neurologico ‘C. Besta’, Milano, Italy; A.G. Engel, Department of Neurology, Mayo Clinic, Rochester, Minnesota, USA; O. Iimura and Y. Kudoh, Second Department of Internal Medicine, Sapporo Medical College, Sapporo, Hokkaido, Japan; B. Neri, Istituto di Clinica Medica Generale e Teripica Medica II, Universita degli Studi di Firenze, Firenze, Italy; E. Schmidt-Sommerfeld, The International Institute for Infant Nutrition and Gastrointestinal Disease, Buffalo, New York, USA; P. Welling, School of Pharmacy, University of Michigan, Ann Arbor, Michigan, USA.
‘Carnitene’, ‘Carnicor’ (Sigma Tau); ‘Bicarnesine’ (Sanofi); ‘Flatistine’ (Sauba); ‘Lefcar’ (Glaxo); ‘Disocor’ (Medicamenta); ‘Carrier’ (Chiesi); ‘Carnitan’ (Kaken Seiyaku); ‘Monocamin’ (Tanabe Seiyaku); and various others worldwide.
Rights and permissions
About this article
Cite this article
Goa, K.L., Brogden, R.N. l-Carnitine. Drugs 34, 1–24 (1987). https://doi.org/10.2165/00003495-198734010-00001
Published:
Issue Date:
DOI: https://doi.org/10.2165/00003495-198734010-00001