Abstract
Introduction
Sentinel lymph node (SLN) biopsy is recommended for all patients with intermediate-thickness melanomas. We sought to identify such patients at low risk of SLN positivity.
Methods
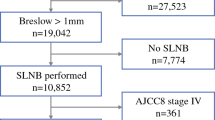
All patients with intermediate-thickness melanomas (1.01–4 mm) undergoing SLN biopsy at a single institution from 1995–2011 were included in this retrospective cohort study. Univariate and multivariate logistic regression determined factors associated with a low risk of SLN positivity. Classification and regression tree (CART) analysis was used to stratify groups based on risk of positivity.
Results
Of the 952 study patients, 157 (16.5 %) had a positive SLN. In the multivariate analysis, thickness <1.5 mm (odds ratio [OR] 0.29), age ≥60 (OR 0.69), present tumor-infiltrating lymphocytes (OR 0.60), absent lymphovascular invasion (OR 0.46), and absent satellitosis (OR 0.44) were significantly associated with a low risk of SLN positivity. CART analysis identified thickness of 1.5 mm as the primary cut point for risk of SLN metastasis. Patients with a thickness of <1.5 mm represented 36 % of the total cohort and had a SLN positivity rate of 6.6 % (95 % confidence interval 3.8–9.4 %). In patients with melanomas <1.5 mm in thickness, the presence of additional low risk factors identified 257 patients (75 % of patients with <1.5 mm melanomas) in which the rate of SLN positivity was <5 %.
Conclusions
Despite a SLN positivity rate of 16.5 % overall, substantial heterogeneity of risk exists among patients with intermediate-thickness melanoma. Most patients with melanoma between 1.01 and 1.5 mm have a risk of SLN positivity similar to that in patients with thin melanomas.

Similar content being viewed by others
References
Karakousis GC, Gimotty PA, Czerniecki BJ, et al. Regional nodal metastatic disease is the strongest predictor of survival in patients with thin vertical growth phase melanomas: a case for SLN staging biopsy in these patients. Ann Surg Oncol. 2007;14(5):1596–603.
Morton DL, Thompson JF, Cochran AJ, et al. Sentinel-node biopsy or nodal observation in melanoma. N Engl J Med. 2006;355(13):1307–17.
Mozzillo N, Caraco C, Chiofalo MG, et al. Sentinel lymph node biopsy in patients with cutaneous melanoma: outcome after 3-year follow-up. Eur J Surg Oncol. 2004;30(4):440–3.
Murali R, Haydu LE, Quinn MJ, et al. Sentinel lymph node biopsy in patients with thin primary cutaneous melanoma. Ann Surg. 2012;255(1):128–33.
Coit DG, Thompson JA, Andtbacka R, et al. Melanoma, version 4.2014. J Natl Compr Canc Netw. 2014;12(5):621–9.
Carlson GW, Murray DR, Hestley A, Staley CA, Lyles RH, Cohen C. Sentinel lymph node mapping for thick (≥4-mm) melanoma: should we be doing it? Ann Surg Oncol. 2003;10(4):408–15.
Gajdos C, Griffith KA, Wong SL, et al. Is there a benefit to sentinel lymph node biopsy in patients with T4 melanoma? Cancer. 2009;115(24):5752–60.
Bartlett EK, Gimotty PA, Sinnamon AJ, et al. Clark level risk stratifies patients with mitogenic thin melanomas for sentinel lymph node biopsy. Ann Surg Oncol. 2014;21(2):643–9.
Han D, Zager JS, Shyr Y, et al. Clinicopathologic predictors of sentinel lymph node metastasis in thin melanoma. J Clin Oncol. 2013;31(35):4387–93.
Karakousis GC, Gimotty PA, Botbyl JD, et al. Predictors of regional nodal disease in patients with thin melanomas. Ann Surg Oncol. 2006;13(4):533–41.
Warycha MA, Zakrzewski J, Ni Q, et al. Meta-analysis of sentinel lymph node positivity in thin melanoma (≤1 mm). Cancer. 2009;115(4):869–79.
McKinnon JG, Yu XQ, McCarthy WH, Thompson JF. Prognosis for patients with thin cutaneous melanoma: long-term survival data from New South Wales Central Cancer Registry and the Sydney Melanoma Unit. Cancer. 2003;98(6):1223–31.
Wright BE, Scheri RP, Ye X, et al. Importance of sentinel lymph node biopsy in patients with thin melanoma. Arch Surg. 2008;143(9):892–9; discussion 899–900.
Morton DL, Thompson JF, Essner R, et al. Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymphadenectomy for early-stage melanoma: a multicenter trial. Multicenter Selective Lymphadenectomy Trial Group. Ann Surg. 1999;230(4):453–63; discussion 463–5.
Gimotty PA, Guerry D, Ming ME, et al. Thin primary cutaneous malignant melanoma: a prognostic tree for 10-year metastasis is more accurate than American Joint Committee on cancer staging. J Clin Oncol. 2004;22(18):3668–76.
Kesmodel SB, Karakousis GC, Botbyl JD, et al. Mitotic rate as a predictor of sentinel lymph node positivity in patients with thin melanomas. Ann Surg Oncol. 2005;12(6):449–58.
Steinberg D. CART: tree-structured non-parametric data analysis. San Diego: Salford Systems; 1995.
Taylor RC, Patel A, Panageas KS, Busam KJ, Brady MS. Tumor-infiltrating lymphocytes predict sentinel lymph node positivity in patients with cutaneous melanoma. J Clin Oncol. 2007;25(7):869–75.
Mays MP, Martin RC, Burton A, et al. Should all patients with melanoma between 1 and 2 mm Breslow thickness undergo sentinel lymph node biopsy? Cancer. 2010;116(6):1535–44.
Balch CM, Thompson JF, Gershenwald JE, et al. Age as a predictor of sentinel node metastasis among patients with localized melanoma: an inverse correlation of melanoma mortality and incidence of sentinel node metastasis among young and old patients. Ann Surg Oncol. 2014;21(4):1075–81.
Cintolo JA, Gimotty P, Blair A, et al. Local immune response predicts survival in patients with thick (t4) melanomas. Ann Surg Oncol. 2013;20(11):3610–7.
Balch CM, Gershenwald JE, Soong SJ, et al. Final version of 2009 AJCC melanoma staging and classification. J Clin Oncol. 2009;27(36):6199–206.
Wong SL, Kattan MW, McMasters KM, Coit DG. A nomogram that predicts the presence of sentinel node metastasis in melanoma with better discrimination than the American Joint Committee on Cancer staging system. Ann Surg Oncol. 2005;12(4):282–8.
Acknowledgement
This study was supported in part by grants P50-CA093372, P30-CA016520, and P50-174523 from the National Cancer Institute. Content is solely the responsibility of the authors and does not necessarily represent the official view of the National Cancer Institute or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure
The authors declare no conflicts of interest.
Additional information
Edmund K. Bartlett and Madalyn G. Peters have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Bartlett, E.K., Peters, M.G., Blair, A. et al. Identification of Patients with Intermediate Thickness Melanoma at Low Risk for Sentinel Lymph Node Positivity. Ann Surg Oncol 23, 250–256 (2016). https://doi.org/10.1245/s10434-015-4766-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-015-4766-y