ABSTRACT
Background
The National Comprehensive Cancer Network guidelines recommend axillary imaging prior to neoadjuvant chemotherapy (NAC) in breast cancer patients who are clinically node negative (cN0) by physical examination. However, the benefit of this approach remains uncertain. The purpose of this study was to determine whether abnormal axillary imaging pre-NAC predicts nodal metastases post-NAC (ypN+) in cN0 patients.
Methods
cN0 patients undergoing NAC followed by axillary surgery were identified. Rates of ypN+ were compared among patients with abnormal pre-treatment axillary imaging vs. normal or no pre-treatment imaging using Fisher’s exact test.
Results
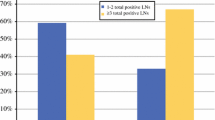
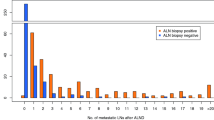
From May 2008 to March 2016, 402 eligible cN0 patients were identified. The median age of the patients was 49.5 years, and the median tumor size was 4 cm. Of these patients, 38% were estrogen receptor-positive (ER+) and human epidermal growth factor receptor 2-negative (HER2−), 30% were HER2+ , and 32% were triple negative. All had pre-NAC mammograms, 40% axillary ultrasound, 83% MRI, and 51% PET. Abnormal nodes on imaging were seen in 208 patients (52%); 128 had pre-NAC node biopsy, and 75 were positive. Overall, 28% of the patients (n = 111) were ypN+ post-NAC. Although the incidence of ypN+ was significantly higher in patients with abnormal nodes on pre-NAC imaging (p = 0.001), 54% did not require axillary lymph node dissection (ALND) post-NAC. Among the patients with normal nodes on pre-NAC imaging, 20% were ypN+ post-NAC.
Conclusions
Half of patients with abnormal nodes on pre-NAC imaging did not require ALND post-NAC, while 20% of those with normal pre-NAC nodes had disease post-NAC, indicating that in cN0 patients already selected for NAC, axillary imaging pre-NAC does not predict the need for axillary surgery post-NAC with sufficient accuracy to be clinically useful.
Similar content being viewed by others
REFERENCES
Houssami N, Ciatto S, Turner RM, Cody HS III, Macaskill P. Preoperative ultrasound-guided needle biopsy of axillary nodes in invasive breast cancer: meta-analysis of its accuracy and utility in staging the axilla. Ann Surg. 2011; 254:243–51.
Holwitt DM, Gillanders WE, Aft RL, Eberlein TJ, Margenthaler JA. Sentinel lymph node biopsy in patients with multicentric/multifocal breast cancer: low false-negative rate and lack of axillary recurrence. Am J Surg. 2008;196:562–5.
Pilewskie M, Jochelson M, Gooch JC, Patil S, Stempel M, Morrow M. Is preoperative axillary imaging beneficial in identifying clinically node-negative patients requiring axillary lymph node dissection? J Am Coll Surg. 2016;222:138–45.
Giuliano AE, Hunt KK, Ballman KV, Beitsch PD, Whitworth PW, Blumencranz PW, et al. Axillary dissection vs no axillary dissection in women with invasive breast cancer and sentinel node metastasis: a randomized clinical trial. JAMA. 2011;305:569–75.
Gradishar WJ, Anderson BO, Blair SL, Burstein HJ, Cyr A, Elias AD, et al. Breast cancer version 3.2014. J Natl Compr Cancer Netw. 2014;12:542–90.
Boughey JC, McCall LM, Ballman KV, Mittendorf EA, Ahrendt GM, Wilke LG, et al. Tumor biology correlates with rates of breast-conserving surgery and pathologic complete response after neoadjuvant chemotherapy for breast cancer: findings from the ACOSOG Z1071 (Alliance) Prospective Multicenter Clinical Trial. Ann Surg. 2014; 260: 608–14.
Mamtani A, Barrio AV, King TA, Van Zee KJ, Plitas G, Pilewskie M, et al. How often does neoadjuvant chemotherapy avoid axillary dissection in patients with histologically confirmed nodal metastases? Results of a prospective study. Ann Surg Oncol. 2016, 23: 3467–3474
Bevilacqua JL, Kattan MW, Fey JV, Cody HS III, Borgen PI, Van Zee KJ. Doctor, what are my chances of having a positive sentinel node? A validated nomogram for risk estimation. J Clin Oncol. 2007;25:3670–9.
Rastogi P, Anderson SJ, Bear HD, Geyer CE, Kahlenberg MS, Robidoux A, et al. Preoperative chemotherapy: updates of National Surgical Adjuvant Breast and Bowel Project Protocols B-18 and B-27. J Clin Oncol. 2008;26:778–85.
Hunt KK, Yi M, Mittendorf EA, Guerrero C, Babiera GV, Bedrosian I, et al. Sentinel lymph node surgery after neoadjuvant chemotherapy is accurate and reduces the need for axillary dissection in breast cancer patients. Ann Surg. 2009;250:558–66.
Diepstraten SC, Sever AR, Buckens CF, Veldhuis WB, van Dalen T, van den Bosch MA, et al. Value of preoperative ultrasound-guided axillary lymph node biopsy for preventing completion axillary lymph node dissection in breast cancer: a systematic review and meta-analysis. Ann Surg Oncol. 2014;21:51–9.
Javid S, Segara D, Lotfi P, Raza S, Golshan M. Can breast MRI predict axillary lymph node metastasis in women undergoing neoadjuvant chemotherapy. Ann Surg Oncol. 2010;17:1841–6.
Jeong YJ, Kang DY, Yoon HJ, Son HJ. Additional value of F-18 FDG PET/CT for initial staging in breast cancer with clinically negative axillary nodes. Breast Cancer Res Treat. 2014;145:137–42.
Cristofanilli M, Gonzalez-Angulo A, Sneige N, Kau SW, Broglio K, Theriault RL, et al. Invasive lobular carcinoma classic type: response to primary chemotherapy and survival outcomes. J Clin Oncol. 2005;23:41–8.
Lips EH, Mulder L, de Ronde JJ, Mandjes IA, Vincent A, Vrancken Peeters MT, et al. Neoadjuvant chemotherapy in ER+HER2− breast cancer: response prediction based on immunohistochemical and molecular characteristics. Breast Cancer Res Treat. 2012;131:827–36.
Wiechmann L, Sampson M, Stempel M, Jacks LM, Patil SM, King T, et al. Presenting features of breast cancer differ by molecular subtype. Ann Surg Oncol. 2009;16:2705–10.
Clarke M, Collins R, Darby S, Davies C, Elphinstone P, Evans V, et al. Effects of radiotherapy and of differences in the extent of surgery for early breast cancer on local recurrence and 15-year survival: an overview of the randomised trials. Lancet. 2005;366:2087–106.
Mamounas EP, Anderson SJ, Dignam JJ, Bear HD, Julian TB, Geyer CE Jr, et al. Predictors of locoregional recurrence after neoadjuvant chemotherapy: results from combined analysis of National Surgical Adjuvant Breast and Bowel Project B-18 and B-27. J Clin Oncol. 2012;30:3960–6.
ACKNOWLEDGEMENTS
A National Institutes of Health/National Cancer Institute Cancer Center Support Grant (No. P30 CA008748) supported the preparation of this report.
Conflict of interest
There are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
Andrea V. Barrio and Anita Mamtani contributed equally to this study and share first authorship.
Rights and permissions
About this article
Cite this article
Barrio, A.V., Mamtani, A., Eaton, A. et al. Is Routine Axillary Imaging Necessary in Clinically Node-Negative Patients Undergoing Neoadjuvant Chemotherapy?. Ann Surg Oncol 24, 645–651 (2017). https://doi.org/10.1245/s10434-017-5765-y
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1245/s10434-017-5765-y