Abstract
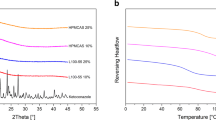
Colloidal solid dispersion is an innovative breakthrough in the pharmaceutical industry that overcomes the solubility-related issue of poorly soluble drugs by using an amorphous approach and also the stability-related issue by means of a complex formation phenomenon using different carrier materials. In the present study, a newly developed adsorption method is introduced to incorporate a high-energy sulfathiazole–polyvinylpyrrolidone (Plasdone® K-29/32) solid dispersion on porous silicon dioxide (Syloid® 244FP). Different ternary systems of sulfathiazole–Plasdone® K-29/32–Syloid® 244FP were prepared (1:1:2, 1:1:3, and 1:2:2) and categorized depending on the mechanism by which Syloid® 244FP was incorporated. Modulated differential scanning calorimetry (MDSC), X-ray diffraction, Fourier transform infrared spectroscopy, and in vitro dissolution studies were conducted to characterize the ternary systems. The X-ray diffraction and MDSC data showed a lack of crystallinity in all internal and external ternary systems, suggesting a loss of the crystallinity of sulfathiazole compared to the physical mixtures. USP apparatus II was used to measure the in vitro dissolution rate of the prepared systems at 75 rpm in different media. The dissolution rate of the optimum ratio (1:2:2) containing an internal ternary solid dispersion system was found to be three times higher than that of the external and physical systems. Thus, the porous silicon dioxide incorporated into the conventional binary solid dispersion acted as a carrier to disperse the complex and increase the dissolution rate.











Similar content being viewed by others
REFERENCES
Arnum P. Solubilizing the insoluble. Pharm Technol. 2010;34(11):50–6.
Dare J. Particle size in relation to formulation. Australas J Pharm. 1964;45:S58–65.
Kai T, Akiyama Y, Nomura S, Sato M. Oral absorption improvement of poorly soluble drug using solid dispersion technique. Chem Pharm Bull. 1996;44:568–71.
Serajuddin A. Solid dispersion of poorly water-soluble drugs: early promises, subsequent problems, and recent breakthroughs. J Pharm Sci. 1999;88:1058–66.
Chiou W, Riegelman S. Pharmaceutical application of solid dispersion systems. J Pharm Sci. 1971;60:1281–302.
Noyes A, Whitney W. The rate of solution of solid substances in their own solutions. J Am Chem Soc. 1897;19:930–4.
Sekiguchi K, Obi N. Studies on absorption of eutectic mixture. I. A comparison of the behavior of eutectic mixture of sulfathiazole and that of ordinary sulfathiazole in man. Chem Pharm Bull. 1961;9:866–72.
Vasconcelos T, Sarmento B, Costa P. Solid dispersion as the strategy to improve oral bioavailability of poor water soluble drugs. Drug Discov Today. 2010;12:1068–75.
Leuner C, Dressman J. Improving drug solubility using solid dispersion. Eur J Pharm Biopharm. 2000;50:47–60.
Gupta M, Goldman D, Bogner R, Tseng Y. Enhanced drug dissolution and bulk properties of solid dispersions granulated with a surface adsorbent. Pharm Dev Technol. 2001;6:563–72.
Prabhu S, Brocks D, Betageri G. Enhancement of dissolution of ethopropazine using solid dispersions prepared with phospholipid and/or polyethylene glycol. Drug Dev Ind Pharm. 2001;27:413–8.
Hirasawa N, Ishise S, Miyata H, Danjo K. An attempt to stabilize nilvadipine solid dispersion by the use of ternary systems. Drug Dev Ind Pharm. 2003;29:997–1004.
Okonogi S, Puttipipatkhachorn S. Dissolution improvement of high drug-loaded solid dispersion. AAPS PharmSciTech. 2006;7:E1–6.
Chutimaworapan S, Ritthidej G, Yonemochi E, Oguchi T, Yamamoto K. Effect of water-soluble carriers on dissolution characteristics of nifedipine solid dispersions. Drug Dev Ind Pharm. 2000;26(11):1141–50.
Lalitha Y, Lakshmi P. Enhancement of dissolution of nifedipine by surface solid dispersion technique. Int J Pharm Pharm Sci. 2011;3 Suppl 3:41–6.
Elbary A, Salem H, Maher M. In vitro and in vivo evaluation of glibenclamide using surface solid dispersion (SSD) approach. Br J Pharmacol Toxicol. 2011;2(1):51–62.
Wang L, Cui F, Sunada H. Preparation and evaluation of solid dispersions of nitrendipine prepared with fine silica particles using the melt-mixing method. Chem Pharm Bull. 2006;54:37–43.
Kim K, Frank M, Henderson N. Application of differential scanning calorimetry to the study of solid drug dispersions. J Pharm Sci. 1985;74(3):283–9.
Takeuchi H, Nagira S, Yamamoto H, Kawashima Y. Solid dispersion particles of amorphous indomethacin with fine porous silica particles prepared by using spray-drying method. Int J Pharm. 2005;293:155–64.
Takeuchi H, Nagira S, Yamamoto H, Kawashima Y. Solid dispersion particles of tolbutamide prepared with fine silica particles by the spray-drying method. Powder Technol. 2004;141:187–95.
Simonell A, Mehta S, Higuchi W. Inhibition of sulfathiazole crystal growth by polyvinylpyrrolidone. J Pharm Sci. 1970;59:633–44.
Simonelli A, Mehta S, Higuchi W. Dissolution rate of high energy polyvinylpyrrolidone (PVP)–sulfathiazole coprecipitates. J Pharm Sci. 1969;58:538–49.
Sethia S, Squillante E. Solid dispersion of carbamazepine in PVP K30 by conventional solvent evaporation and supercritical methods. Int J Pharm. 2004;272:1–10.
Bakara M, Nagya Z, Rielly C, Dann S. Investigation of the riddle of sulfathiazole polymorphism. Int J Pharm. 2011;414:86–103.
Tantishaiyakul V, Kaewnopparat N, Ingkatawornwong S. Properties of solid dispersions of piroxicam in polyvinylpyrrolidone K-30. Int J Pharm. 1996;143:59–66.
Karavas E, Ktistis G, Xenakis A, Georgarakis E. Miscibility behavior and formation mechanism of stabilized felodipine–polyvinylpyrrolidone amorphous solid dispersions. Drug Dev Ind Pharm. 2005;31(6):473–89.
Drooge D, Hinrichs W, Visser M, Frijlink H. Characterization of the molecular distribution of drugs in glassy solid dispersions at the nano-meter scale, using differential scanning calorimetry and gravimetric water vapour sorption techniques. Int J Pharm. 2006;310:220–9.
Feng T, Pinal R, Carvjal MT. Process induced disorder in crystalline materials: differentiating defective crystals from the amorphous form of griseofulvin. J Pharm Sci. 2008;88:1058–66.
Bahl D, Bogner RH. Amorphization alone does not account for the enhancement of solubility of drug co-ground with silicate: the case of indomethacin. AAPS PharmSciTech. 2008;9(1):146–53.
Benchaabane H, Ozer Y, Ozalp M, Kilic E, Polat M, Korkmaz M. Gamma radiation studies on sulfathiazole (powder and model-ophthalmic solution). FABAD J Pharm Sci. 2003;28(2):93–106.
Mooter G, Augustijns P, Blaton N, Kinget R. Physico-chemical characterization of solid dispersions of temazepam with polyethylene glycol 6000 and PVP K30. Int J Pharm. 1998;164:67–80.
Boldyrev V, Shakhtshneider T, Burleva L, Severtsev V. Preparation of the disperse system of sulfathiazole–polyvinylpyrrolidone by mechanical activation. Drug Dev Ind Pharm. 1994;20(6):1103–14.
Hirasawa N, Ishise S, Miyata H, Danjo K. Physicochemical characterization and drug release studies of nilvadipine solid dispersions using water-insoluble polymer as a carrier. Drug Dev Ind Pharm. 2003;29(3):339–44.
Chauhan B, Shimpi S, Paradkar A. Preparation and evaluation of glibenclamide-polyglycolized glycerides solid dispersions with silicon dioxide by spray drying technique. Eur J Pharm Sci. 2005;26:219–30.
Zhu L, Wong L, Yu L. Surface-enhanced crystallization of amorphous nifedipine. Mol Pharm. 2008;5(6):921–6.
Author information
Authors and Affiliations
Corresponding author
ELECTRONIC SUPPLEMENTARY MATERIAL
Below is the link to the electronic supplementary material.
ESM 1
(PDF 9113 kb)
Rights and permissions
About this article
Cite this article
Patel, V.I., Dave, R.H. Evaluation of Colloidal Solid Dispersions: Physiochemical Considerations and In Vitro Release Profile. AAPS PharmSciTech 14, 620–628 (2013). https://doi.org/10.1208/s12249-013-9947-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1208/s12249-013-9947-z