Abstract
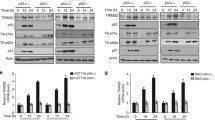
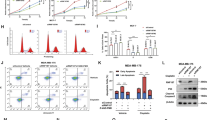
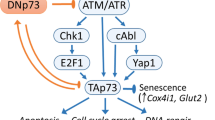
Tumor suppressor protein p63 is a transcription factor of the p53 protein family. It is known that the proteins of this family, namely, p53, p63, and p73, perform a number of common tumor suppressor functions related to cell cycle regulation, apoptosis, and malignant transformation of cells, in addition to specific functions in the development of tissues and organs. All the proteins of the p53 family are regulated at post-translational level due to ubiquitination by E3 ligases and subsequent proteasomal degradation. The expression of the majority of E3 ubiquitin ligases, which modify the proteins of the p53 family, is regulated by the same proteins, thus forming negative feedback with them. In the present study, we first showed that the expression of the PIRH2 (RCHY1) gene, the product of which ubiquitinates all three members of the p53 family, is also regulated by p63. Regulation of the PIRH2 expression occurs at the transcription level by only fulllength TAp63 isoform, but not ΔNp63 isoform, which lacks a transactivation domain.
Similar content being viewed by others
References
Cam, M., Bid, H.K., Xiao, L., Zambetti, G.P., Houghton, P.J., and Cam, H., p53/TAp63 and AKT regulate mammalian target of rapamycin complex 1 (mTORC1) signaling through two independent parallel pathways in the presence of DNA damage, J. Biol. Chem., 2014, vol. 289, pp. 4083–4094.
Daks, A.A., Melino, D., and Barlev, N.A., The role of different E3 ubiquitin ligases in regulation of the P53 tumor suppressor protein, Tsitologiia, 2013, vol. 55, no. 10, pp. 673–687.
Gallegos, J.R., Litersky, J., Lee, H., Sun, Y., Nakayama, K., Nakayama, K., and Lu, H., SCFβTrCP1 activates and ubiquitylates TAp63γ, J. Biol. Chem., 2008, vol. 283, pp. 66–75.
Gressner, O., Schilling, T., Lorenz, K., Schleithoff, E.S., Koch, A., Schulze-Bergkamen, H., and Müller, M., TAp63α induces apoptosis by activating signaling via death receptors and mitochondria, EMBO J., 2005, vol. 24, pp. 2458–2471.
Jung, Y.S., Qian, Y., and Chen, X., The p73 tumor suppressor is targeted by Pirh2 RING finger E3 ubiquitin ligase for the proteasome-dependent degradation, J. Biol. Chem., 2011, vol. 286, pp. 35388–35395.
Jung, Y.S., Qian, Y., Yan, W., and Chen, X., Pirh2 E3 ubiquitin ligase modulates keratinocyte differentiation through p63, J. Invest. Dermatol., 2013, vol. 133, pp. 1178–1187.
Kadakia, M., Slader, C., and Berberich, S.J., Regulation of p63 function by Mdm2 and MdmX, DNA Cell Biol., 2001, vol. 20, pp. 321–330.
Kerr, J.B., Hutt, K.J., Michalak, E.M., Cook, M., Vandenberg, C.J., Liew, S.H., and Strasser, A., DNA damageinduced primordial follicle oocyte apoptosis and loss of fertility require TAp63-mediated induction of Puma and Noxa, Mol. Cell., 2012, vol. 48, pp. 343–352.
Lee, C.-W. and La Thangue, N.B., Promoter specificity and stability control of the P53-related protein p73, Oncogene, 1999, vol. 18, pp. 4171–4181.
Leng, R.P., Lin, Y., Ma, W., Wu, H., Lemmers, B., Chung, S., Parant, J.M., Lozano, G., Hakem, R., and Benchimol, S., Pirh2, a p53-induced ubiquitin-protein ligase, promotes p53 degradation, Cell, 2003, vol. 112, pp. 779–791.
Logan, I.R., Gaughan, L., McCracken, S.R., Sapountzi, V., Leung, H.Y., and Robson, C.N., Human PIRH2 enhances androgen receptor signaling through inhibition of histone deacetylase 1 and is overexpressed in prostate cancer, Mol. Cell. Biol., 2006, vol. 26, pp. 6502–6510.
Marouco, D., Garabadgiu, A.V., Melino, G., and Barlev, N.A., Lysine-specific modifications of p53: a matter of life and death?, Oncotarget, 2013, vol. 4, pp. 1556–1571.
Mills, A.A., Zheng, B., Wang, X.-J., Vogel, H., Roop, D.R., and Bradley, A., p63 is a p53 homologue required for limb and epidermal morphogenesis, Nature, 1999, vol. 398, pp. 708–713.
Ongkeko, W.M., Wang, X.Q., Siu, W.Y., Lau, A.W., Yamashita, K., Harris, A.L., Cox, L.S., and Poon, R.Y., MDM2 and MDMX bind and stabilize the p53-related protein p73, Curr. Biol., 1999, vol. 9, pp. 829–832.
Shimada, M., Kitagawa, K., Dobashi, Y., Isobe, T., Hattori, T., Uchida, C., Abe, K., Kotake, Y., Oda, T., Suzuki, H., Hashimoto, K., and Kitagawa, M., High expression of Pirh2, an E3 ligase for p27, is associated with low expression of p27 and poor prognosis in head and neck cancers, Cancer Sci., 2009, vol. 100, pp. 866–872.
Suh, E.K., Yang, A., Kettenbach, A., Bamberger, C., Michaelis, A.H., Zhu, Z., Elvin, J.A., Bronson, R.T., Crum, C.P., and McKeon, F., p63 protects the female germ line during meiotic arrest, Nature, 2006, vol. 444, pp. 624–628.
Yan, W., Chen, X., Zhang, Y., Zhang, J., Jung, Y.S., and Chen, X., Arsenic suppresses cell survival via Pirh2-mediated proteasomal degradation of ΔNp63 protein, J. Biol. Chem., 2013, vol. 288, pp. 2907–2913.
Yang, A., Kaghad, M., Wang, Y., Gillett, E., Fleming, M.D., Dötsch, V., Andrews, N.C., Caput, D., and McKeon, F., p63, a p53 homolog at 3q27–29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities, Mol. Cell., 1998, vol. 2, pp. 305–316.
Yang, A., Schweitzer, R., Sun, D., Kaghad, M., Walker, N., Bronson, R.T., Tabin, C., Sharpe, A., Caput, D., and Crum, C., p63 is essential for regenerative proliferation in limb, craniofacial and epithelial development, Nature, 1999, vol. 398, pp. 714–718.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Daks, A.V. Petukhov, O.Yu. Shuvalov, E.A. Vasil’eva, G. Melino, N.A. Barlev, O.A. Fedorova, 2015, published in Tsitologiya, 2015, Vol. 57, No. 12, pp. 876–879.
Rights and permissions
About this article
Cite this article
Daks, A.A., Petukhov, A.V., Shuvalov, O.Y. et al. Tumor suppressor p63 regulates Pirh2 ubiquitin ligase expression. Cell Tiss. Biol. 10, 202–205 (2016). https://doi.org/10.1134/S1990519X16030044
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X16030044