Abstract
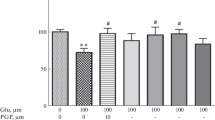
Activation of extrasynaptic NMDA receptors by high glutamate concentrations is one of the key pathogenic factors following a stroke. For this reason, the search for efficient neuroprotective agents that could reduce glutamate toxicity is a pressing need. Ca2+ overload in response to glutamate leads to activation of signaling cascades in the cell and the development of oxidative stress, which ultimately leads to apoptosis. Using a model system of acute excitotoxicity caused by 50 μM NMDA, which was used as a specific NMDA receptor activator, we demonstrated that during 2 hours of incubation the viability of the primary neuronal culture decreased by 30–50%. To demonstrate that the observed effect is associated not only with the Ca2+ influx into the cytoplasm through the activated NMDA receptors, we decreased the Ca2+ concentration in the medium. The lowered Ca2+ concentration, as well as its complete absence, did not affect NMDA toxicity. We tested carnosine, a naturally occurring dipeptide and promising antioxidant, as a neuroprotective agent. The addition of 2 mM carnosine prevented the decrease in cell viability caused by a 2-hour incubation with 50 μM NMDA, while it showed no effect on the viability of the cell culture in the control. Based on the results, we consider the further study of carnosine, its complexes, and analogues as neuroprotectors in cerebral ischemia promising.
Similar content being viewed by others
References
Parsons, M.P. and Raymond, L.A., Neuron, 2014, vol. 82, no. 2, pp. 279–293.
Davydova, O.N. and Boldyrev, A.A., Annaly Klin. Eksp. Nevrol., 2007, vol. 4, pp. 28–34.
Babot, Z., Cristofol, R., and Sunol, C., Eur. J. Neurosci., 2005, vol. 21, no. 1, pp. 103–112.
Dutta, R. and Trapp, B.D., Prog. Neurobiol., 2011, vol. 93, no. 1, pp. 1–12.
Lu, Y.M., Yin, H.Z., Chiang, J., and Weiss, J.H., J. Neurosci., 1996, vol. 16, no. 17, pp. 5457–5465.
Boldyrev, A.A. and Kuklei, M.L., Neirokhimiya, 1996, vol. 13, no. 4, pp. 271–278.
Tymianski, M., Wallace, M.C., Spigelman, I., Uno, M., Carlen, P.L., Tator, C.H., and Charlton, M.P., Neuron, 1993, vol. 11, no. 2, pp. 221–235.
Wie, M.B., Koh, J.Y., Won, M.H., Lee, J.C., Shin, T.K., Moon, C.J., Ha, H.J., Park, S.M., and Kim, H.C., Prog. Neuropsychopharmacol. Biol. Psychiatry, 2001, vol. 25, no. 8, pp. 1641–1659.
Hetman, M. and Gozdz, A., Eur. J. Biochem., 2004, vol. 271, no. 11, pp. 2050–2055.
Coffey, E.T., Nat. Rev. Neurosci., 2014, vol. 15, no. 5, pp. 285–299.
Lopachev, A.V., Lopacheva, O.M., Osipova, E.A., Vladychenskaya, E.A., Smolyaninova, L.V., Fedorova, T.N., Koroleva, O.V., and Akkuratov, E.E., Cell Biochem. Funct., 2016, vol. 34, no. 5, pp. 367–377.
Suslina, Z.A., Illarioshkin, S.N., and Piradov, M.A., Annaly Klin. Eksp. Nevrol., 2007, vol. 1, no. 1, pp. 5–9.
Reynolds, I.J. and Hastings, T.G., J. Neurosci., 1995, vol. 15, no. 5, pt. 1, pp. 3318–3327.
Boldyrev, A.A., Stvolinskii, S.L., and Fedorova, T.N., Usp. Fiziol. Nauk, 2007, vol. 38, no. 3, pp. 57–71.
Boldyrev, A.A., Aldini, G., and Derave, W., Physiol. Rev., 2013, vol. 93, no. 4, pp. 1803–1845.
Lopachev, A.V., Lopacheva, O.M., Abaimov, D.A., Koroleva, O.V., Vladychenskaya, E.A., Erukhimovich, A.A., and Fedorova, T.N., Biochemistry (Moscow), 2016, vol. 81, no. 5, pp. 511–520.
Abaimov, D.A., Sariev, A.K., Tankevich, M.V., Pantyukhova, E.Yu., Prokhorov, D.I., Fedorova, T.N., Lopachev, A.V., Stvolinskii, S.L., Konovalova, E.V., and Seifulla, R.D., Eksp. Klin. Farmakol., 2015, vol. 78, no. 3, pp. 30–35.
Dobrota, D., Fedorova, T., Stvolinsky, S., Babusikova, E., Likavcanova, K., Drgova, A., Strapkova, A., and Boldyrev, A., Neurochem. Res., 2005, vol. 30, no. 10, pp. 1283–1288.
Boldyrev, A., Song, R., Lawrence, D., and Carpenter, D.O., Neuroscience, 1999, vol. 94, no. 2, pp. 571–577.
Bilimoria, P.M. and Bonni, A., Cold Spring Harb. Protoc., 2008, vol. 3, no.12.
Antonov, S.M. and Johnson, J.W., Proc. Natl. Acad. Sci. USA, 1999, vol. 96, no. 25, pp. 14571–14576.
Davis, S. and Donnan, G.A., Cerebrovasc. Dis., 2014, vol. 38, no. 1, pp. 59–72.
Akkuratov, E.E., Lopacheva, O.M., Kruusmagi, M., Lopachev, A.V., Shah, Z.A., Boldyrev, A.A., and Liu, L., Mol. Neurobiol., 2015, vol. 52, no. 3, pp. 1726–1734.
Ruiz, A., Matute, C., and Alberdi, E., Cell Calcium, 2009, vol. 46, no. 4, pp. 273–281.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.V. Lopachev, O.M. Lopacheva, E.E. Akkuratov, S.L. Stvolinskii, T.N. Fedorova, 2017, published in Neirokhimiya, 2017, Vol. 34, No. 1, pp. 49–53.
Rights and permissions
About this article
Cite this article
Lopachev, A.V., Lopacheva, O.M., Akkuratov, E.E. et al. Carnosine protects a primary cerebellar cell culture from acute NMDA toxicity. Neurochem. J. 11, 38–42 (2017). https://doi.org/10.1134/S1819712417010068
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1819712417010068