Abstract
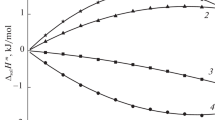
Integral enthalpy of dissolution of L-alanine, D,L-alanine, and D,L-valine in aqueous propanediol-1,3 (the diol molar fraction 0.26) has been determined. Standard enthalpies of the amino acids dissolution and transfer from water into the mixed solvent have been calculated. Influence of the interaction types and structural features of the amino acids and organic cosolvent on the enthalpies have been analyzed.
Similar content being viewed by others
References
Franks, F. and Smith, T., Trans. Faraday Soc., 1968, vol. 64, p. 2962.
Saleh, M., Begum, S, Begum, S.K., and Begum, B.A., Phys. Chem. Liq., 1999, vol. 37, p. 785.
Cabani, S., Conti, G., and Lepori, L., J. Phys. Chem., 1974, vol. 78, p. 1030.
Smirnov, V.I., Mezhevoi, I.N., and Badelin, V.G., Russ. J. Phys. Chem., A, 2004, vol. 78, no. 2, p. 214.
Friedman, H.L. and Krishnan, C.V., Water. A Comprehensive Treatise, Franks, F., New York: Plenum Press, 1973, vol. 3, ch. 1.
Santoro, M.M., Liu, Y., Khan, S.M.A., Hou, L.X., and Bolen, D.W., Biochem., 1992, vol. 31, p. 5278.
Mezhevoi, I.N. and Badelin, V.G., Russ. J. Phys. Chem., A, 2010, no. 4, vol. 84, p. 607.
Lilley, T.H., Pure Appl. Chem., 1993, vol. 65, p. 2551.
Lilley, T.H., Moses, E., and Tasker, I.R., J. Chem. Soc., Faraday Trans., 1980, vol. 76, p. 906.
Mezhevoi, I.N. and Badelin, V.G., Biofizika, 2009, vol. 54, no. 6, p. 984.
McMillan, W.G. and Mayer, J.E., J. Chem. Phys., 1945, vol. 13, p. 276.
Matsumoto, M. and Amaya, K., Chem. Lett., 1978, vol. 1, p. 87.
Palecz, B., Thermochim. Acta, 2005, vol. 435, p. 99.
Zittle, C.A. and Schmidt, C.L.A., J. Biol. Chem., 1935, vol. 108, p. 161.
Desnoyers, J.E., Perron, G., Avedikian, L., and Morel, J.-P., J. Solution Chem., 1976, vol. 5, p. 631.
Mezhevoi I.N. and Badelin V.G., Russ. J. Gen. Chem., 2012, vol. 82, no. 11, p. 1801.
Nozaki, Y. and Tanford, C., J. Biol. Chem., 1971, vol. 246, p. 2211.
Devido, D.R., Dorsey, J.G., Chan, H.S., and Dill, K.A., J. Phys. Chem. (B), 1998, vol. 102, p. 7272.
Makhatadze, G.I. and Privalov, P.L., J. Mol. Biol., 1990, vol. 213, p. 375.
Ide, M., Maeda, Y., and Kitano, H., J. Phys. Chem. (B), 1997, vol. 101, p. 7022.
Romero, C.M. and Paez, M., J. Therm. Anal. Calorim., 2002, vol. 70, p. 263.
Sijpkes, A.H. and Somsen, G., J. Chem. Soc., Faraday Trans. 1, 1989, vol. 85, p. 2563.
Castronuovo, G., Elia, V., and Velleca, F., J. Solution Chem., 1996, vol. 25, p. 51.
Mezhevoi, I.N., Candidate Sci. (Chem.) Dissertation, Moscow, 2004.
Parker, V.B., Washington: NSRDS-NBS 2, 1965, p. 66.
Wadso, I. Mezhevoi, I.N. Goldberg, R.N., Pure Appl. Chem., 2001, vol. 73, p. 1625.
Archer, D.G., J. Phys. Chem. Ref. Data., 1999, vol. 28, p. 1.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © I.N. Mezhevoi, V.G. Badelin, 2014, published in Zhurnal Obshchei Khimii, 2014, Vol. 84, No. 2, pp. 229–233.
Rights and permissions
About this article
Cite this article
Mezhevoi, I.N., Badelin, V.G. Thermochemical analysis of intermolecular interaction of aliphatic amino acids with propanediol-1,3 in aqueous media. Russ J Gen Chem 84, 223–226 (2014). https://doi.org/10.1134/S107036321402011X
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S107036321402011X