Abstract
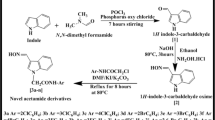
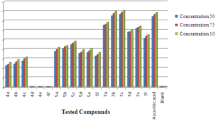
2,5-Disubstituted 1,3,4-oxadiazole compounds are one of the most attractive heterocyclic compounds for researchers due to their biological activities. In the undertaken research, a number of potential 2,5-disubstituted 1,3,4-oxadiazole analogues were synthesized through multi step reaction and characterized by FT-IR, 1H NMR, mass spectra, and also by elemental analysis. Further benzophenone tagged indole acetohydrazides and 2,5-disubstituted 1,3,4-oxadiazoles were evaluated for antioxidant potential, through different in vitro models such as DPPH, nitric oxide and hydrogen peroxide methods. In the series of compounds some of them had shown good to moderate in vitro antioxidant potential compare to the standard drug ascorbic acid in all the above three methods.
Similar content being viewed by others
References
Kramerff, J.B., Boschelli, D.H, Connoti, D.T., Kostlanw, C.R., Kuiperss, P.J., Kennedy, J.A., Bioorg. Med. Chem. Lett., 1993, vol. 3, no.12, pp. 2827–2835.
Chakraborty, P., Kumar, S., Dutta, D., and Gupta, V., Res. J. Pharm. Tech., 2009, vol. 2, no. 2, pp. 238–244.
Lopez-Gresa, M.P., Torres, C., Campos, L., Lison, P., Rodrigo, I., Belles, J.M., and Conejero, V., Environ. Exp. Bot., 2011, vol. 74, pp. 216–228.
Samshuddin, S., Narayana, B., Sarojini, B.K., and Madhu, N.L., Med. Chem. Res., 2012, doi: 10.1007/s00044-012-.0304-7
Rakesh Somani, R. and Prabhakar Shirodkar, Y., Der Pharma Chemica, 2009, vol. 1. pp. 130–140.
Karthikeyan, M.S., Prasad, D.J., Mahalinga, M., Holla, B.S., and Kumari, N.S., Eur. J. Med. Chem., 2008, vol. 43, pp. 25–31.
Undavia, N.K., Trivedi, P.B., Shanishchara, A.P., and Trivedi, V.P., J. Ind. Chem. Soc., 2005, vol. 82, no. 8, pp. 746–752.
Khanum, S.A., Shashikanth, S., Sudha, B.S., Deepak, S.A., and Shetty, H.S., Pest Manag. Sci., 2004, vol. 60, pp. 1119–1124.
Macaev, F., Rusu, G., Pogrebnoi, S., Gudima, A., Stingaci, E., and Vlad, L., Bioorg. Med. Chem., 2005, vol. 13, pp. 4842–4850.
Palaska, E., Sahin, G., Kelicen, P., Durlu, N.T., and Altinok, G., Il Farmaco, 2002, vol. 57, pp. 101–107.
Won, C., Shen, X.L., Mashiguchib, K., Zheng, Z., Dai, X.H., Cheng, Y.F., Kasahara, H., Kamiya, Y., Chory, J., and Zhao, Y.D., Proc. Natl. Acad. Sci. U.S.A., 2011, vol. 108, pp. 18518–18523.
Abdel-Motaleb, R.M., Makhloof, A.M.A., Ibrahim, H.M., and Elnagdi, M.H., J. Heterocycl. Chem., 2007, vol. 44, pp. 109–114.
Moth, C.W., Prusakiewicz, J.J., Marnett, L.J., and Lybrand, T.P., J. Med. Chem., 2005, vol. 48, pp. 3613–3620.
Khanum, S.A., Girish, V., Suparshwa, S.S, and Khanum, N.F., Bioorg. Med. Chem. Lett., 2009, vol. 19, pp. 1887–1891.
Khanum, S.A., Shashikanth, S., and Deepak, A.V., Bio. Org. Chem., 2004, vol. 32, pp. 211–222.
Lakshmi Ranganatha, V., Vinutha, N., Khanum, S.A., Anthal, S., Revannasiddaiah, D., Rajnikant, K., and Vivek Gupta, K., X-ray Structure Analysis Online, 2012, vol. 28, pp. 27–28.
Khanum, S.A., Shashikanth, S., Umesha, S., and Kavitha, R., 2Eur. J. Med. Chem., 2005, vol. 40, pp. 1156–1162.
Burits, M. and Bucar, F., Phytother. Res., 2000, vol. 14, pp. 323–328.
Marcocci, L., Maguire, J.J., Droy-Lefaix, M.T., and Packer, L., Biochem. Biophys. Res. Commun., 1994, vol. 201, pp. 748–755.
Ruch, R.J., Cheng, S.J., and Klaunig, J.E., Carcinogenesis, 1989, vol. 10, pp. 1003–1008.
Author information
Authors and Affiliations
Corresponding author
Additional information
The article is published in the original.
Rights and permissions
About this article
Cite this article
Lakshmi Ranganatha, V., Khanum, S.A. Synthesis and evaluation of in vitro antioxidant properties of novel 2,5-disubstituted 1,3,4-oxadiazoles. Russ J Bioorg Chem 40, 206–213 (2014). https://doi.org/10.1134/S1068162014020083
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1068162014020083