Abstract
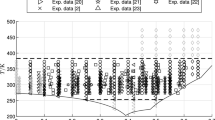
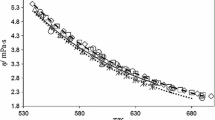
An equation for describing the dynamic viscosity of aqueous solutions of sodium chloride in the concentration range of 0 to 6m at temperatures from 0 to 325°C and pressures up to 40 MPa is presented.
Similar content being viewed by others
References
Stokes, R.H. and Mills, R., Viscosity of Strong Electrolytes, London: Pergamon, 1965, p. 118.
Ezrokhi, L.L., Zh. Prikl. Khim. (Leningrad), 1952, vol. 25, no. 8, p. 838.
Kaminscky, M., Z. Phys. Chem. (Leipzig), 1955, vol. 5, p. 154.
Kaminsky, M., Z. Phys. Chem. (Leipzig), 1956, vol. 8, p. 173.
Suryanarayana, C.V. and Venkatesan, V.K., Trans. Faraday Soc., 1958, vol. 54, p. 1709.
Korosi, A. and Fabuss, B.M., J. Chem. Eng. Data, 1968, vol. 13, no 4, p. 548.
Semenyuk, E.N., Zarembo, V.I., and Fedorov, M.K., Zh. Prikl. Khim. (Leningrad), 1977, vol. 50, no. 2, p. 215.
Goncalves, F.K. and Kestin, J., Ber. Bunsen-Ges., 1977, vol. 81, p. 1156.
Kestin, J., Khalifa, H.E., Ro, S.T., and Wakeham, W.A., J. Chem. Eng. Data, 1977, vol. 22, p. 207.
Kestin, J., Khalifa, H.E., Abe, Y., and Grimes, C.E., J. Chem. Eng. Data, 1978, vol. 23, p. 328.
Penonov, R.I., Yusufova, V.D., Lobkova, N.V., and Panakhov, I.A., High Temp., 1978, vol. 16, no. 5, p. 816.
Out, D.J.P. and Los, J.M., J. Solution Chem., 1980, vol. 9, p. 19.
Kestin, J. and Shankland, J., Int. J. Thermophys., 1984, vol. 5, no. 3, p. 241.
Afzal, M., Saleem, M., and Mahmood, M.T., J. Chem. Eng. Data, 1989, vol. 34, p. 339.
Agaev, N.A., in Teplofizicheskie svoistva veshchestv. Trudy VIII Vsesoyuznoi konferentsii (Proceedings of the VIII All-Union Conference on Thermophysical Properties of Substances, Novosibirsk, Russia, 1989), Novosibirsk: Institute of Thermophysics, Siberian Branch of the Academy of Sciences of the Soviet Union, 1989, part 1, p. 119.
Akhundov, T.S., Guseinov, A.G., Ishkhanov, Yu.B., Akhundov, R.T., and Iskanderov, A.I., Izv. Vyssh. Uchebn. Zaved., Neft Gaz, 1990, no. 7, p. 65.
Sawamura, S., Yoshimura, Y., Kitamura, K., and Taniguchi, Y., J. Phys. Chem., 1992, vol. 96, p. 5526.
Zhang, H.-L. and Han, S.-J., J. Chem. Eng. Data, 1996, vol. 41, p. 516.
Motin, M.A., J. Chem. Eng. Data, 2004, vol. 49, p. 49.
Liu, Y.S., Hu, Y.F., Hao, Q.C., Zhang, X.M., Liu, Z.C., and Li, J.G., J. Chem. Eng. Data, 2009, vol. 54, p. 739.
International Association for the Properties of Water and Steam. Release on the IAPWS Formulation 2008 for the Viscosity of Ordinary Water Substance. http://www. iapws.org.
Falkenhagen, H., Theorie der Elektrolite, Leipzig: S. Hirzel, 1971.
Viscosity of Water, Geneva: International Organization for Standardization (ISO), ISO/TR Technical Report 3666 (E), 1998.
Valyashko, V.M., Kravchuk, K.G., Urusova, M.A., et al., Obz. Teplofiz. Svoistvam Veshchestv, 1986, no. 4.
Pepinov, R.I., Yusufova, V.D., and Lobkova, N.V., GSSSD (State Service of Standard Reference Data) R 43-82, Moscow, 1982.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.A. Aleksandrov, E.V. Dzhuraeva, V.F. Utenkov, 2012, published in Teplofizika Vysokikh Temperatur, 2012, Vol. 50, No. 3, pp. 378–383.
Rights and permissions
About this article
Cite this article
Aleksandrov, A.A., Dzhuraeva, E.V. & Utenkov, V.F. Viscosity of aqueous solutions of sodium chloride. High Temp 50, 354–358 (2012). https://doi.org/10.1134/S0018151X12030029
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0018151X12030029