Abstract
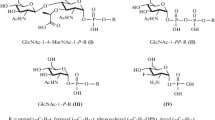
Phenyl, p-tolyl, and p-tert-butylphenyl β-1-thio-N-acetylglucosaminides were synthesized by the treatment of thiophenols with peracetate of α-D-glucosaminyl chloride in the presence of triethylamine or under the conditions of phase-transfer catalysis with quaternary ammonium salts. The compounds synthesized were used for obtaining of glycosides of 4,6-O-isopropylidene-N-acetylmuramic acid, which were coupled with L-Ala-D-Glu(NH2)-OBzl and then deprotected to obtain the target aryl β-thioglycosides of N-acetylmuramyl-L-analyl-D-isoglutamine (MDP). The aryl β-thioglycosides of MDP were found to stimulate an antibacterial resistance toward Staphylococcus aureus in mice. The reliable induction of the spontaneous activity of natural killers in the population of blood mononuclear cells was observed only for phenyl β-thio-MDP at a dose of 200 µg/ml.
Similar content being viewed by others
Abbreviations
- MDP:
-
N-acetylmuramyl-L-alanyl-D-isoglutamine (muramyl dipeptide)
- MNC:
-
mononuclear blood cells
- NK:
-
natural killers
References
Zemlyakov, A.E., Tsikalov, V.V., Kalyuzhin, O.V., Kur’yanov, V.O., and Chirva V.Ya., Bioorg. Khim., 2003, vol. 29, pp. 316–322; Rus. J. Bioorg. Chem., 2003, vol. 29, pp. 286–292.
Karaulov, A.V., Kalyuzhin, O.V., and Zemlyakov, A.E., Ross. Bioterapevt. Zh., 2002, vol. 1, pp. 35–39.
Zemlyakov, A.E., Tsikalova, V.N., Tsikalov, V.V., Chirva, V.Ya., Mulik, E.L., and Kalyuzhin, O.V., Bioorg. Khim., 2005, vol. 31, pp. 637–644; Rus. J. Bioorg. Chem., 2005, vol. 31, pp. 576–582.
Hasegawa, A., Hioki, Y., Kiso, M., Okumura, H., and Azuma, I., J. Carbohydr. Chem., 1982–1983, vol. 1, pp. 317–323.
Ishida, H., Kigawa, K., Kitagawa, M., Kiso, M., Hasegawa, A., and Azuma, I., Agric. Biol. Chem., 1991, vol. 55, pp. 585–587.
Zemlyakov, A.E., Tsikalova, V.N., Tsikalov, V.V., and Chirva, V.Ya., Zh. Inst. Farm. Khim., 2003, vol. 2, pp. 17–20.
Kleine, H.P., Weinberg, D.V., Kaufman, R.J., and Sidhu, R.S., Carbohydr. Res., 1985, vol. 142, pp. 333–337.
Kur’yanov, V.O., Chupakhina, T.A., Zemlyakov, A.E., Kotlyar, S.A., Kamalob, G.L., and Chirva, V.Ya., Bioorg. Khim., 2001, vol. 27, pp. 434–438; Rus. J. Bioorg. Chem., 2001, vol. 27, pp. 385–389.
US Patent 5874548, 1999 (httr://patft.usrto.gov/netahtml/searsh-bool.html).
Fermand-Jian, S., Perly, B., Level, M., and Lefrancier, P., Carbohydr. Res., 1987, vol. 162, pp. 23–32.
Kalyuzhin, O.V., Mulik, E.L., Sergeev, V.V., Kalina, N.G., Elkina, S.I., Kalyuzhina, M.I., Kuzovlev, F.N., and Karaulov, A.V., Immunopathologiya, Allergologiya, Infektologiya, 2000, no. 4, pp. 73–77.
Khaitov, R.M., Gushchin, I.S., Pinegin, B.F., and Zebrev, A.I., Rukovodstvo po eksperimental’nomu (doklinicheskomu) izucheniya novykh farmakologicheskikh veshchestv (Handbook on the Experimental (Preclinical) Study of New Pharmacological Substances), Moscow: IIA Remedium, 2000, p. 257–263.
Zemluakov, A.E., Tsikalova, V.N., Tsikalov, V.V., Chirva, V.Ya., Mulik, E.L., Kuzovlev, F.N., Kalyuzhin, O.V., and Kiselevsky, M.V., Bioorg. Khim., 2008, vol. 34, pp. 114–120, Rus. J. Bioorg. Chem., 2008, vol. 34.
Shimadate, T., Ghitoa, S., Inouye, K., Iino, T., and Hosoyama, Y., Bull. Chem. Soc. Jpn., 1982, vol. 55, pp. 3552–3554.
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © A.E. Zemlyakov, V.N. Tsikalova, L.R. Azizova, V.Ya. Chirva, E.L. Mulik, M.V. Shkalev, O.V. Kalyuzhin, M.V. Kiselevsky, 2008, published in Bioorganicheskaya Khimiya, 2008, Vol. 34, No. 2, pp. 245–251.
Rights and permissions
About this article
Cite this article
Zemlyakov, A.E., Tsikalova, V.N., Azizova, L.R. et al. Synthesis and biological activity of aryl S-β-glycosides of 1-thio-N-acetylmuramyl-L-alanyl-D-isoglutamine. Russ J Bioorg Chem 34, 223–229 (2008). https://doi.org/10.1134/S106816200802012X
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S106816200802012X