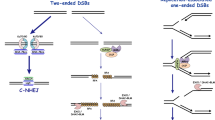
Abstract
Rejoining of broken chromosomes is crucial for cell survival and prevention of malignant transformation. Most mammalian cells rely primarily on the non-homologous end-joining pathway of DNA double-strand break (DSB) repair to accomplish this task. This review focuses both on the core non-homologous end-joining machinery, which consists of DNA-dependent protein kinase and the ligase IV/XRCC4 complex, and on accessory factors that facilitate rejoining of a subset of the DSBs. We discuss how the ATM protein kinase and the Mre11/Rad50/Nbs1 complex might function in DSB repair and what role ionizing radiation-induced foci may play in this process.


Similar content being viewed by others
References
Ahnesorg P, Smith P, Jackson SP . (2006). XLF interacts with the XRCC4-DNA ligase IV complex to promote DNA nonhomologous end-joining. Cell 124: 301–313.
Aravind L, Koonin EV . (2000). SAP—a putative DNA-binding motif involved in chromosomal organization. Trends Biochem Sci 25: 112–114.
Bailey SM, Meyne J, Chen DJ, Kurimasa A, Li GC, Lehnert BE et al. (1999). DNA double-strand break repair proteins are required to cap the ends of mammalian chromosomes. Proc Natl Acad Sci USA 96: 14899–14904.
Barnes DE, Stamp G, Rosewell I, Denzel A, Lindahl T . (1998). Targeted disruption of the gene encoding DNA ligase IV leads to lethality in embryonic mice. Curr Biol 8: 1395–1398.
Bartek J, Lukas J . (2007). DNA damage checkpoints: from initiation to recovery or adaptation. Curr Opin Cell Biol 19: 238–245.
Block WD, Yu Y, Merkle D, Gifford JL, Ding Q, Meek K et al. (2004). Autophosphorylation-dependent remodeling of the DNA-dependent protein kinase catalytic subunit regulates ligation of DNA ends. Nucleic Acids Res 32: 4351–4357.
Bryans M, Valenzano MC, Stamato TD . (1999). Absence of DNA ligase IV protein in XR-1 cells: evidence for stabilization by XRCC4. Mutat Res 433: 53–58.
Buck D, Malivert L, de Chasseval R, Barraud A, Fondaneche MC, Sanal O et al. (2006). Cernunnos, a novel nonhomologous end-joining factor, is mutated in human immunodeficiency with microcephaly. Cell 124: 287–299.
Burma S, Chen BP, Chen DJ . (2006). Role of non-homologous end joining (NHEJ) in maintaining genomic integrity. DNA Repair (Amst) 5: 1042–1048.
Capp JP, Boudsocq F, Bertrand P, Laroche-Clary A, Pourquier P, Lopez BS et al. (2006). The DNA polymerase lambda is required for the repair of non-compatible DNA double strand breaks by NHEJ in mammalian cells. Nucleic Acids Res 34: 2998–3007.
Capp JP, Boudsocq F, Besnard AG, Lopez BS, Cazaux C, Hoffmann JS et al. (2007). Involvement of DNA polymerase mu in the repair of a specific subset of DNA double-strand breaks in mammalian cells. Nucleic Acids Res 35: 3551–3560.
Celeste A, Fernandez-Capetillo O, Kruhlak MJ, Pilch DR, Staudt DW, Lee A et al. (2003). Histone H2AX phosphorylation is dispensable for the initial recognition of DNA breaks. Nat Cell Biol 5: 675–679.
Chan DW, Chen BP, Prithivirajsingh S, Kurimasa A, Story MD, Qin J et al. (2002). Autophosphorylation of the DNA-dependent protein kinase catalytic subunit is required for rejoining of DNA double-strand breaks. Genes Dev 16: 2333–2338.
Chen BP, Chan DW, Kobayashi J, Burma S, Asaithamby A, Morotomi-Yano K et al. (2005). Cell cycle dependence of DNA-dependent protein kinase phosphorylation in response to DNA double strand breaks. J Biol Chem 280: 14709–14715.
Chen L, Trujillo K, Sung P, Tomkinson AE . (2000). Interactions of the DNA ligase IV-XRCC4 complex with DNA ends and the DNA-dependent protein kinase. J Biol Chem 275: 26196–26205.
Chun HH, Gatti RA . (2004). Ataxia-telangiectasia, an evolving phenotype. DNA Repair (Amst) 3: 1187–1196.
Costantini S, Woodbine L, Andreoli L, Jeggo PA, Vindigni A . (2007). Interaction of the Ku heterodimer with the DNA ligase IV/Xrcc4 complex and its regulation by DNA-PK. DNA Repair (Amst) 6: 712–722.
Cui X, Yu Y, Gupta S, Cho YM, Lees-Miller SP, Meek K . (2005). Autophosphorylation of DNA-dependent protein kinase regulates DNA end processing and may also alter double-strand break repair pathway choice. Mol Cell Biol 25: 10842–10852.
Darroudi F, Wiegant W, Meijers M, Friedl AA, van der Burg M, Fomina J et al. (2007). Role of Artemis in DSB repair and guarding chromosomal stability following exposure to ionizing radiation at different stages of cell cycle. Mutat Res 615: 111–124.
de Jager M, van Noort J, van Gent DC, Dekker C, Kanaar R, Wyman C . (2001). Human Rad50/Mre11 is a flexible complex that can tether DNA ends. Mol Cell 8: 1129–1135.
de Vries E, van Driel W, Bergsma WG, Arnberg AC, van der Vliet PC . (1989). HeLa nuclear protein recognizing DNA termini and translocating on DNA forming a regular DNA-multimeric protein complex. J Mol Biol 208: 65–78.
Deckbar D, Birraux J, Krempler A, Tchouandong L, Beucher A, Walker S et al. (2007). Chromosome breakage after G2 checkpoint release. J Cell Biol 176: 749–755.
DeFazio LG, Stansel RM, Griffith JD, Chu G . (2002). Synapsis of DNA ends by DNA-dependent protein kinase. EMBO J 21: 3192–3200.
Digweed M, Reis A, Sperling K . (1999). Nijmegen breakage syndrome: consequences of defective DNA double strand break repair. Bioessays 21: 649–656.
Ding Q, Reddy YV, Wang W, Woods T, Douglas P, Ramsden DA et al. (2003). Autophosphorylation of the catalytic subunit of the DNA-dependent protein kinase is required for efficient end processing during DNA double-strand break repair. Mol Cell Biol 23: 5836–5848.
Douglas P, Cui X, Block WD, Yu Y, Gupta S, Ding Q et al. (2007). The DNA-dependent protein kinase catalytic subunit is phosphorylated in vivo on threonine 3950, a highly conserved amino acid in the protein kinase domain. Mol Cell Biol 27: 1581–1591.
Drouet J, Delteil C, Lefrancois J, Concannon P, Salles B, Calsou P . (2005). DNA-dependent protein kinase and XRCC4-DNA ligase IV mobilization in the cell in response to DNA double strand breaks. J Biol Chem 280: 7060–7069.
Ege M, Ma Y, Manfras B, Kalwak K, Lu H, Lieber MR et al. (2005). Omenn syndrome due to ARTEMIS mutations. Blood 105: 4179–4186.
Falck J, Coates J, Jackson SP . (2005). Conserved modes of recruitment of ATM, ATR and DNA-PKcs to sites of DNA damage. Nature 434: 605–611.
Franco S, Gostissa M, Zha S, Lombard DB, Murphy MM, Zarrin AA et al. (2006). H2AX prevents DNA breaks from progressing to chromosome breaks and translocations. Mol Cell 21: 201–214.
Frank KM, Sekiguchi JM, Seidl KJ, Swat W, Rathbun GA, Cheng HL et al. (1998). Late embryonic lethality and impaired V(D)J recombination in mice lacking DNA ligase IV. Nature 396: 173–177.
Frank KM, Sharpless NE, Gao Y, Sekiguchi JM, Ferguson DO, Zhu C et al. (2000). DNA ligase IV deficiency in mice leads to defective neurogenesis and embryonic lethality via the p53 pathway. Mol Cell 5: 993–1002.
Gao Y, Chaudhuri J, Zhu C, Davidson L, Weaver DT, Alt FW . (1998a). A targeted DNA-PKcs-null mutation reveals DNA-PK-independent functions for KU in V(D)J recombination. Immunity 9: 367–376.
Gao Y, Sun Y, Frank KM, Dikkes P, Fujiwara Y, Seidl KJ et al. (1998b). A critical role for DNA end-joining proteins in both lymphogenesis and neurogenesis. Cell 95: 891–902.
Gell D, Jackson SP . (1999). Mapping of protein–protein interactions within the DNA-dependent protein kinase complex. Nucleic Acids Res 27: 3494–3502.
Goodarzi AA, Yu Y, Riballo E, Douglas P, Walker SA, Ye R et al. (2006). DNA-PK autophosphorylation facilitates Artemis endonuclease activity. EMBO J 25: 3880–3889.
Grawunder U, Wilm M, Wu X, Kulesza P, Wilson TE, Mann M et al. (1997). Activity of DNA ligase IV stimulated by complex formation with XRCC4 protein in mammalian cells. Nature 388: 492–495.
Grawunder U, Zimmer D, Leiber MR . (1998). DNA ligase IV binds to XRCC4 via a motif located between rather than within its BRCT domains. Curr Biol 8: 873–876.
Gu J, Lu H, Tsai AG, Schwarz K, Lieber MR . (2007). Single-stranded DNA ligation and XLF-stimulated incompatible DNA end ligation by the XRCC4-DNA ligase IV complex: influence of terminal DNA sequence. Nucleic Acids Res 35: 5755–5762.
Gu Y, Seidl KJ, Rathbun GA, Zhu C, Manis JP, van der Stoep N et al. (1997). Growth retardation and leaky SCID phenotype of Ku70-deficient mice. Immunity 7: 653–665.
Helleday T, Lo J, van Gent DC, Engelward BP . (2007). DNA double-strand break repair: from mechanistic understanding to cancer treatment. DNA Repair (Amst) 6: 923–935.
Hopfner KP, Craig L, Moncalian G, Zinkel RA, Usui T, Owen BA et al. (2002). The Rad50 zinc-hook is a structure joining Mre11 complexes in DNA recombination and repair. Nature 418: 562–566.
Hsu HL, Yannone SM, Chen DJ . (2002). Defining interactions between DNA-PK and ligase IV/XRCC4. DNA Repair (Amst) 1: 225–235.
Iliakis G, Wang H, Perrault AR, Boecker W, Rosidi B, Windhofer F et al. (2004). Mechanisms of DNA double strand break repair and chromosome aberration formation. Cytogenet Genome Res 104: 14–20.
Jung D, Giallourakis C, Mostoslavsky R, Alt FW . (2006). Mechanism and control of V(D)J recombination at the immunoglobulin heavy chain locus. Annu Rev Immunol 24: 541–570.
Junop MS, Modesti M, Guarne A, Ghirlando R, Gellert M, Yang W . (2000). Crystal structure of the xrcc4 DNA repair protein and implications for end joining. EMBO J 19: 5962–5970.
Kabotyanski EB, Gomelsky L, Han JO, Stamato TD, Roth DB . (1998). Double-strand break repair in Ku86- and XRCC4-deficient cells. Nucleic Acids Res 26: 5333–5342.
Karanjawala ZE, Grawunder U, Hsieh CL, Lieber MR . (1999). The nonhomologous DNA end joining pathway is important for chromosome stability in primary fibroblasts. Curr Biol 9: 1501–1504.
Kobayashi N, Agematsu K, Sugita K, Sako M, Nonoyama S, Yachie A et al. (2003). Novel Artemis gene mutations of radiosensitive severe combined immunodeficiency in Japanese families. Hum Genet 112: 348–352.
Koch CA, Agyei R, Galicia S, Metalnikov P, O'Donnell P, Starostine A et al. (2004). Xrcc4 physically links DNA end processing by polynucleotide kinase to DNA ligation by DNA ligase IV. EMBO J 23: 3874–3885.
Kurimasa A, Kumano S, Boubnov NV, Story MD, Tung CS, Peterson SR et al. (1999). Requirement for the kinase activity of human DNA-dependent protein kinase catalytic subunit in DNA strand break rejoining. Mol Cell Biol 19: 3877–3884.
Lavin MF, Kozlov S . (2007). ATM activation and DNA damage response. Cell Cycle 6: 931–942.
Lee AC, Fernandez-Capetillo O, Pisupati V, Jackson SP, Nussenzweig A . (2005). Specific association of mouse MDC1/NFBD1 with NBS1 at sites of DNA-damage. Cell Cycle 4: 177–182.
Lee JW, Blanco L, Zhou T, Garcia-Diaz M, Bebenek K, Kunkel TA et al. (2004). Implication of DNA polymerase lambda in alignment-based gap filling for nonhomologous DNA end joining in human nuclear extracts. J Biol Chem 279: 805–811.
Lee Y, McKinnon PJ . (2007). Responding to DNA double-strand breaks in the nervous system. Neuroscience 145: 1365–1374.
Li L, Moshous D, Zhou Y, Wang J, Xie G, Salido E et al. (2002). A founder mutation in Artemis, an SNM1-like protein, causes SCID in Athabascan-speaking Native Americans. J Immunol 168: 6323–6329.
Lieber MR, Hesse JE, Lewis S, Bosma GC, Rosenberg N, Mizuuchi K et al. (1988). The defect in murine severe combined immune deficiency: joining of signal sequences but not coding segments in V(D)J recombination. Cell 55: 7–16.
Lou Z, Minter-Dykhouse K, Franco S, Gostissa M, Rivera MA, Celeste A et al. (2006). MDC1 maintains genomic stability by participating in the amplification of ATM-dependent DNA damage signals. Mol Cell 21: 187–200.
Ma Y, Pannicke U, Lu H, Niewolik D, Schwarz K, Lieber MR . (2005). The DNA-dependent protein kinase catalytic subunit phosphorylation sites in human Artemis. J Biol Chem 280: 33839–33846.
Ma Y, Pannicke U, Schwarz K, Lieber MR . (2002). Hairpin opening and overhang processing by an Artemis/DNA-dependent protein kinase complex in nonhomologous end joining and V(D)J recombination. Cell 108: 781–794.
Mari PO, Florea BI, Persengiev SP, Verkaik NS, Bruggenwirth HT, Modesti M et al. (2006). Dynamic assembly of end-joining complexes requires interaction between Ku70/80 and XRCC4. Proc Natl Acad Sci USA 103: 18597–18602.
Maser RS, Monsen KJ, Nelms BE, Petrini JH . (1997). hMre11 and hRad50 nuclear foci are induced during the normal cellular response to DNA double-strand breaks. Mol Cell Biol 17: 6087–6096.
McBlane JF, van Gent DC, Ramsden DA, Romeo C, Cuomo CA, Gellert M et al. (1995). Cleavage at a V(D)J recombination signal requires only RAG1 and RAG2 proteins and occurs in two steps. Cell 83: 387–395.
McKinnon PJ . (2004). ATM and ataxia telangiectasia. EMBO Rep 5: 772–776.
Meek K, Douglas P, Cui X, Ding Q, Lees-Miller SP . (2007). Trans Autophosphorylation at DNA-dependent protein kinase's two major autophosphorylation site clusters facilitates end processing but not end joining. Mol Cell Biol 27: 3881–3890.
Modesti M, Junop MS, Ghirlando R, van de Rakt M, Gellert M, Yang W et al. (2003). Tetramerization and DNA ligase IV interaction of the DNA double-strand break repair protein XRCC4 are mutually exclusive. J Mol Biol 334: 215–228.
Moreno-Herrero F, de Jager M, Dekker NH, Kanaar R, Wyman C, Dekker C . (2005). Mesoscale conformational changes in the DNA-repair complex Rad50/Mre11/Nbs1 upon binding DNA. Nature 437: 440–443.
Moshous D, Callebaut I, de Chasseval R, Corneo B, Cavazzana-Calvo M, Le Deist F et al. (2001). Artemis, a novel DNA double-strand break repair/V(D)J recombination protein, is mutated in human severe combined immune deficiency. Cell 105: 177–186.
Moshous D, Pannetier C, Chasseval Rd R, Deist Fl F, Cavazzana-Calvo M, Romana S et al. (2003). Partial T and B lymphocyte immunodeficiency and predisposition to lymphoma in patients with hypomorphic mutations in Artemis. J Clin Invest 111: 381–387.
Nick McElhinny SA, Havener JM, Garcia-Diaz M, Juarez R, Bebenek K, Kee BL et al. (2005). A gradient of template dependence defines distinct biological roles for family x polymerases in nonhomologous end joining. Mol Cell 19: 357–366.
Nick McElhinny SA, Snowden CM, McCarville J, Ramsden DA . (2000). Ku recruits the XRCC4-ligase IV complex to DNA ends. Mol Cell Biol 20: 2996–3003.
Nikjoo H, Uehara S, Wilson WE, Hoshi M, Goodhead DT . (1998). Track structure in radiation biology: theory and applications. Int J Radiat Biol 73: 355–364.
Noordzij JG, Verkaik NS, Van der Burg M, Van Veelen LR, De Bruin-Versteeg S, Wiegant W et al. (2003). Radiosensitive SCID patients with Artemis gene mutations show a complete B-cell differentiation arrest at the pre-B-cell receptor checkpoint in bone marrow. Blood 101: 1446–1452.
Nussenzweig A, Chen C, da Costa Soares V, Sanchez M, Sokol K, Nussenzweig MC et al. (1996). Requirement for Ku80 in growth and immunoglobulin V(D)J recombination. Nature 382: 551–555.
O'Driscoll M, Cerosaletti KM, Girard PM, Dai Y, Stumm M, Kysela B et al. (2001). DNA ligase IV mutations identified in patients exhibiting developmental delay and immunodeficiency. Mol Cell 8: 1175–1185.
Ouyang H, Nussenzweig A, Kurimasa A, Soares VC, Li X, Cordon-Cardo C et al. (1997). Ku70 is required for DNA repair but not for T cell antigen receptor gene recombination In vivo. J Exp Med 186: 921–929.
Pannicke U, Ma Y, Hopfner KP, Niewolik D, Lieber MR, Schwarz K . (2004). Functional and biochemical dissection of the structure-specific nuclease ARTEMIS. EMBO J 23: 1987–1997.
Poinsignon C, Moshous D, Callebaut I, de Chasseval R, Villey I, de Villartay JP . (2004). The metallo-beta-lactamase/beta-CASP domain of Artemis constitutes the catalytic core for V(D)J recombination. J Exp Med 199: 315–321.
Reddy YV, Ding Q, Lees-Miller SP, Meek K, Ramsden DA . (2004). Non-homologous end joining requires that the DNA-PK complex undergo an autophosphorylation-dependent rearrangement at DNA ends. J Biol Chem 279: 39408–39413.
Riballo E, Critchlow SE, Teo SH, Doherty AJ, Priestley A, Broughton B et al. (1999). Identification of a defect in DNA ligase IV in a radiosensitive leukaemia patient. Curr Biol 9: 699–702.
Riballo E, Kuhne M, Rief N, Doherty A, Smith GC, Recio MJ et al. (2004). A pathway of double-strand break rejoining dependent upon ATM, Artemis, and proteins locating to gamma-H2AX foci. Mol Cell 16: 715–724.
Ristic D, Modesti M, Kanaar R, Wyman C . (2003). Rad52 and Ku bind to different DNA structures produced early in double-strand break repair. Nucleic Acids Res 31: 5229–5237.
Rooney S, Alt FW, Lombard D, Whitlow S, Eckersdorff M, Fleming J et al. (2003). Defective DNA repair and increased genomic instability in Artemis-deficient murine cells. J Exp Med 197: 553–565.
Rooney S, Sekiguchi J, Zhu C, Cheng HL, Manis J, Whitlow S et al. (2002). Leaky SCID phenotype associated with defective V(D)J coding end processing in Artemis-deficient mice. Mol Cell 10: 1379–1390.
Sawada M, Sun W, Hayes P, Leskov K, Boothman DA, Matsuyama S . (2003). Ku70 suppresses the apoptotic translocation of Bax to mitochondria. Nat Cell Biol 5: 320–329.
Schatz DG . (2004). V(D)J recombination. Immunol Rev 200: 5–11.
Schwarz K, Gauss GH, Ludwig L, Pannicke U, Li Z, Lindner D et al. (1996). RAG mutations in human B cell-negative SCID. Science 274: 97–99.
Sharpless NE, Ferguson DO, O'Hagan RC, Castrillon DH, Lee C, Farazi PA et al. (2001). Impaired nonhomologous end-joining provokes soft tissue sarcomas harboring chromosomal translocations, amplifications, and deletions. Mol Cell 8: 1187–1196.
Sibanda BL, Critchlow SE, Begun J, Pei XY, Jackson SP, Blundell TL et al. (2001). Crystal structure of an Xrcc4-DNA ligase IV complex. Nat Struct Biol 8: 1015–1019.
Singleton BK, Torres-Arzayus MI, Rottinghaus ST, Taccioli GE, Jeggo PA . (1999). The C terminus of Ku80 activates the DNA-dependent protein kinase catalytic subunit. Mol Cell Biol 19: 3267–3277.
Slijepcevic P . (2006). The role of DNA damage response proteins at telomeres—an ‘integrative’ model. DNA Repair (Amst) 5: 1299–1306.
Smith GC, Jackson SP . (1999). The DNA-dependent protein kinase. Genes Dev 13: 916–934.
Spagnolo L, Rivera-Calzada A, Pearl LH, Llorca O . (2006). Three-dimensional structure of the human DNA-PKcs/Ku70/Ku80 complex assembled on DNA and its implications for DNA DSB repair. Mol Cell 22: 511–519.
Taccioli GE, Amatucci AG, Beamish HJ, Gell D, Xiang XH, Torres Arzayus MI et al. (1998). Targeted disruption of the catalytic subunit of the DNA-PK gene in mice confers severe combined immunodeficiency and radiosensitivity. Immunity 9: 355–366.
Taylor AM, Groom A, Byrd PJ . (2004). Ataxia-telangiectasia-like disorder (ATLD)-its clinical presentation and molecular basis. DNA Repair (Amst) 3: 1219–1225.
Teo SH, Jackson SP . (2000). Lif1p targets the DNA ligase Lig4p to sites of DNA double-strand breaks. Curr Biol 10: 165–168.
Tsai CJ, Kim SA, Chu G . (2007). Cernunnos/XLF promotes the ligation of mismatched and noncohesive DNA ends. Proc Natl Acad Sci USA 104: 7851–7856.
Uematsu N, Weterings E, Yano K, Morotomi-Yano K, Jakob B, Taucher-Scholz G et al. (2007). Autophosphorylation of DNA-PKCS regulates its dynamics at DNA double-strand breaks. J Cell Biol 177: 219–229.
van der Burg M, van Veelen LR, Verkaik NS, Wiegant WW, Hartwig NG, Barendregt BH et al. (2006). A new type of radiosensitive T-B-NK+ severe combined immunodeficiency caused by a LIG4 mutation. J Clin Invest 116: 137–145.
van der Burg M, Verkaik NS, den Dekker AT, Barendregt BH, Pico-Knijnenburg I, Tezcan I et al. (2007). Defective Artemis nuclease is characterized by coding joints with microhomology in long P-nucleotide stretches. Eur J Immunol (in press).
van Gent DC, Hoeijmakers JH, Kanaar R . (2001). Chromosomal stability and the DNA double-stranded break connection. Nat Rev Genet 2: 196–206.
van Gent DC, Ramsden DA, Gellert M . (1996). The RAG1 and RAG2 proteins establish the 12/23 rule in V(D)J recombination. Cell 85: 107–113.
van Heemst D, Brugmans L, Verkaik NS, van Gent DC . (2004). End-joining of blunt DNA double-strand breaks in mammalian fibroblasts is precise and requires DNA-PK and XRCC4. DNA Repair (Amst) 3: 43–50.
Verkaik NS, Esveldt-van Lange RE, van Heemst D, Bruggenwirth HT, Hoeijmakers JH, Zdzienicka MZ et al. (2002). Different types of V(D)J recombination and end-joining defects in DNA double-strand break repair mutant mammalian cells. Eur J Immunol 32: 701–709.
Villa A, Santagata S, Bozzi F, Giliani S, Frattini A, Imberti L et al. (1998). Partial V(D)J recombination activity leads to Omenn syndrome. Cell 93: 885–896.
Walker JR, Corpina RA, Goldberg J . (2001). Structure of the Ku heterodimer bound to DNA and its implications for double-strand break repair. Nature 412: 607–614.
Wang J, Pluth JM, Cooper PK, Cowan MJ, Chen DJ, Yannone SM . (2005). Artemis deficiency confers a DNA double-strand break repair defect and Artemis phosphorylation status is altered by DNA damage and cell cycle progression. DNA Repair (Amst) 4: 556–570.
Ward IM, Minn K, Jorda KG, Chen J . (2003). Accumulation of checkpoint protein 53BP1 at DNA breaks involves its binding to phosphorylated histone H2AX. J Biol Chem 278: 19579–19582.
Weterings E, van Gent DC . (2004). The mechanism of non-homologous end-joining: a synopsis of synapsis. DNA Repair (Amst) 3: 1425–1435.
Weterings E, Verkaik NS, Bruggenwirth HT, Hoeijmakers JH, van Gent DC . (2003). The role of DNA dependent protein kinase in synapsis of DNA ends. Nucleic Acids Res 31: 7238–7246.
Wu PY, Frit P, Malivert L, Revy P, Biard D, Salles B et al. (2007). Interplay between cernunnos-XLF and NHEJ proteins at DNA ends in the cell. J Biol Chem, (in press).
Xu X, Stern DF . (2003). NFBD1/MDC1 regulates ionizing radiation-induced focus formation by DNA checkpoint signaling and repair factors. FASEB J 17: 1842–1848.
Yan CT, Boboila C, Souza EK, Franco S, Hickernell TR, Murphy M et al. (2007). IgH class switching and translocations use a robust non-classical end-joining pathway. Nature 449: 478–482.
Yaneva M, Kowalewski T, Lieber MR . (1997). Interaction of DNA-dependent protein kinase with DNA and with Ku: biochemical and atomic-force microscopy studies. EMBO J 16: 5098–5112.
Zha S, Alt FW, Cheng HL, Brush JW, Li G . (2007). Defective DNA repair and increased genomic instability in Cernunnos-XLF-deficient murine ES cells. Proc Natl Acad Sci USA 104: 4518–4523.
Zhou BB, Elledge SJ . (2000). The DNA damage response: putting checkpoints in perspective. Nature 408: 433–439.
Zhu C, Bogue MA, Lim DS, Hasty P, Roth DB . (1996). Ku86-deficient mice exhibit severe combined immunodeficiency and defective processing of V(D)J recombination intermediates. Cell 86: 379–389.
Acknowledgements
We thank Drs PO Mari, SP Perseng and NGJ Jaspers for useful comments. Research in our laboratories is supported by the European Union (Projects ‘RISC-RAD’ and ‘DNA Repair’) and the Dutch Organization for Scientific Research (NWO/ZonMw veni grant 916.56.107).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
van Gent, D., van der Burg, M. Non-homologous end-joining, a sticky affair. Oncogene 26, 7731–7740 (2007). https://doi.org/10.1038/sj.onc.1210871
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1210871
- Springer Nature Limited
Keywords
This article is cited by
-
Analysis on the desert adaptability of indigenous sheep in the southern edge of Taklimakan Desert
Scientific Reports (2022)
-
Control of DNA integrity in skeletal muscle under physiological and pathological conditions
Cellular and Molecular Life Sciences (2017)
-
Copy Number Variation Disorders
Current Genetic Medicine Reports (2017)
-
Circulating T Cells of Patients with Nijmegen Breakage Syndrome Show Signs of Senescence
Journal of Clinical Immunology (2017)
-
Decreased IL7Rα and TdT expression underlie the skewed immunoglobulin repertoire of human B-cell precursors from fetal origin
Scientific Reports (2016)