Abstract
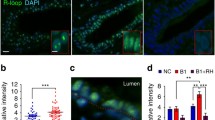
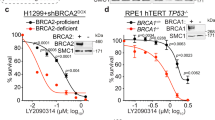
Inactivation of the BRCA1 gene has been found to confer susceptibility to early-onset familial breast and ovarian cancers. BRCA1 regulates DNA repair, chromatin remodeling and affects gene transcription. Transforming growth factor-β (TGFβ) is a potent regulator of growth, apoptosis and invasiveness of tumor cells, including breast cancer cells. Here we show that Smad3 which is a component of the TGFβ signaling pathway, forms a complex with BRCA1 in vitro and in vivo. The interaction is mediated by the MH1 domain of Smad3 and the C-terminal part of BRCA1. We observed a co-localization of Smad3 and BRCA1 in nuclear complexes. We also found that TGFβ1/Smad3 counteracted BRCA1-dependent repair of DNA double-strand breaks in human breast epithelial cells, as evaluated by BRCA1 nuclear foci formation, single-cell gel electrophoresis and cell survival assays. Thus, TGFβ1/Smad3 suppresses BRCA1-dependent DNA repair in response to a DNA damaging agent.





Similar content being viewed by others
References
Boulay J-L, Mild G, Lowly AJ, Reuter J, Lagrange M, Terracciano L, Laffer U, Herrmann R and Rochlitz C . (2002). Br. J. Cancer, 87, 630–634.
de Caestecker M, Piek E and Roberts AB . (2000). J. Natl. Cancer Inst., 92, 1388–1402.
Dennler S, Itoh S, Vivien D, ten Dijke P, Huet S and Gauthier JM . (1998). EMBO J., 17, 3091–3100.
Derynck R, Akhurst RJ and Balmain A . (2001). Nat. Genet., 29, 117–129.
Derynck R and Zhang YE . (2003). Nature, 425, 577–584.
Ewan KB, Henshall-Powel RL, Ravani SA, Pajares MJ, Arteaga C, Warters R, Akhurst RJ and Barcellos-Hoff MH . (2002). Cancer Res., 62, 5627–5631.
Glick A, Popescu N, Alexander V, Ueno H, Botinger E and Yuspa SH . (1999). Proc. Natl. Acad. Sci. USA, 96, 4949–14954.
Kanamoto T, Hellman U, Heldin C-H and Souchelnytskyi S . (2002). EMBO J., 21, 1219–1230.
Kinzler KW and Vogelstein B . (1997). Nature, 386, 761–763.
Kretzschmar M, Doody J, Timokhina I and Massagué J . (1999). Genes Dev., 13, 804–816.
Lomnytska M, Lukiyanchuk V, Hellman U and Souchelnytskyi S . (2004). Proteomics, 4, 995–1006.
Miyajima A, Asano T, Seta K, Asano T, Kakoi N and Hayakawa M. . (2003). Urology, 61, 1072–1077.
Mulder KM . (2000). Cytokine Growth Factor Rev., 11, 23–35.
Muray PA, Barrett-Lee P, Travers M, Luqmani Y, Powles T and Coombes RC . (1993). Br. J. Cancer, 67, 1408–1412.
Ouchi T, Monteiro ANA, August A, Aaronson SA and Hanafusa H . (1998). Proc. Natl. Acad. Sci. USA, 95, 2302–2306.
Paull TT, Cortez D, Bowers B, Elledge SJ and Gellert M . (2001). Proc. Natl. Acad. Sci. USA, 98, 6086–6091.
Preobrazhenska O, Yakymovych M, Kanamoto T, Yakymovych I, Stoika R, Heldin C-H and Souchelnytskyi S . (2002). Oncogene, 21, 5660–5664.
Pulaski L, Landström M, Heldin C-H and Souchelnytskyi S . (2001). J. Biol. Chem., 276, 14344–14349.
Schmied BM, Ulrich AB, Matsuzaki H, El-Metwally TH, Ding X, Fernandes ME, Adrian TE, Chaney WG, Batra SK and Pour PM . (2000). Carcinogenesis, 21, 1121–1127.
Scully R, Chen J, Plug A, Xiao Y, Weaver D, Feunteun J, Ashley T and Livingston DM . (1997). Cell, 88, 265–275.
Siegel PM and Massague J . (2003). Nature Rev., 3, 807–820.
Souchelnytskyi S . (2002). Exp. Oncol., 24, 3–12.
Takaku K, Oshima M, Miyoshi H, Matsui M, Seldin MF and Taketo MM . (1998). Cell, 92, 645–656.
Venkitaraman AR . (2002). Cell, 108, 171–182.
Wang Q, Zhang H, Fishel R and Greene MI . (2000). Oncogene, 19, 6152–6158.
Watanabe T, Wu TT, Catalano PJ, Ueki T, Satriano R, Haller DG, Benson AB and Hamilton SR . (2001). New Engl. J. Med., 344, 1196–1206.
Xiangming C, Natsugoe S, Takao S, Hokita S, Ishigami S, Tanabe G, Baba M, Kuroshima K and Aikou T . (2001). Clin. Cancer Res., 7, 277–282.
Xie W, Mertens JC, Reiss DJ, Rimm DL, Camp RL, Haffty BG and Reiss M . (2002). Cancer Res., 62, 497–505.
Yakymovych I, ten Dijke P, Heldin C-H and Souchelnytskyi S . (2001). FASEB J., 15, 553–556.
Zhu Y, Richardson JA, Parada LF and Graff J . (1998). Cell, 94, 703–714.
Acknowledgements
We are grateful to Hiroyuki Iwahana, Taras Stasyk for comments, Vasyl Lukiyanchuk for help and advises, Mary Helen Barcellos-Hoff for discussions, Toru Ouchi, Dou Liu, Stephen Elledge, Kohei Miyazono, Didier Marot, Napoleone Ferrara and Johan Ericsson for reagents, and Yevhen Filjak for primary amplification of the BRCA1 virus. This work was supported in part by grants from the Royal Swedish Academy of Sciences, the INSERM/MFR/VR, the EU program on angiogenesis (T-ANGIOVASC), the Swedish Cancer Society, and from the Swedish Research Council to Serhiy Souchelnytskyi.
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary Information accompanies the paper on Oncogene website (http://www.nature.com/onc)
Supplementary information
Rights and permissions
About this article
Cite this article
Dubrovska, A., Kanamoto, T., Lomnytska, M. et al. TGFβ1/Smad3 counteracts BRCA1-dependent repair of DNA damage. Oncogene 24, 2289–2297 (2005). https://doi.org/10.1038/sj.onc.1208443
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1208443
- Springer Nature Limited
Keywords
This article is cited by
-
USP9X-mediated KDM4C deubiquitination promotes lung cancer radioresistance by epigenetically inducing TGF-β2 transcription
Cell Death & Differentiation (2021)
-
Downregulation of TGF-β1 suppressed proliferation and increased chemosensitivity of ovarian cancer cells by promoting BRCA1/Smad3 signaling
Biological Research (2018)
-
TGF-β signaling links E-cadherin loss to suppression of nucleotide excision repair
Oncogene (2016)
-
Genetic variants in microRNA and microRNA biogenesis pathway genes and breast cancer risk among women of African ancestry
Human Genetics (2016)
-
Smad7 enhances ATM activity by facilitating the interaction between ATM and Mre11-Rad50-Nbs1 complex in DNA double-strand break repair
Cellular and Molecular Life Sciences (2015)