Abstract
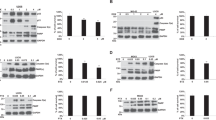
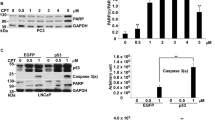
c-Jun N-terminal kinase (JNK) is activated by diverse cell stimuli, including stress, growth factors, and cytokines. Traditionally, activation of JNK by stress treatment is thought to induce cell death. However, our recent data indicate that JNK's ability to sensitize cells to apoptosis may be, in part, cell cycle dependent. Here, we show that the majority of both paclitaxel- and UV-induced apoptosis can be inhibited by the pharmacological JNK inhibitor, SP600125, in MCF-7 cells. However, inhibition of JNK does little to reverse doxorubicin-induced apoptosis in MCF-7 cells or doxorubicin- and UV-mediated death in MDA MB-231 cells. SP treatment causes G2/M arrest of three breast cancer cell lines and results in the endoreduplication (cellular DNA content >4N) of MCF-7 and MDA MB-231 cells. These effects on cell cycle and apoptosis are not significantly altered by the inhibition of p53, indicating that JNK is functioning independently of p53. Lastly, inhibition of JNK using both SP and antisense oligonucleotides targeted to JNK1 and JNK2 reduced proliferation of all three breast cancer cell lines. Taken together, these results suggest that the activation of JNK is important for the induction of apoptosis following stresses that function at different cell cycle phases, and that basal JNK activity is necessary to promote proliferation and maintain diploidy in breast cancer cells.






Similar content being viewed by others
References
Aguirre V, Ucida T, Yenush L, Davis R and White MF . (2000). J. Biol. Chem., 275, 9047–9054.
Aguirre V, Werner ED, Giraud J, Lee YH, Schoelson SE and White MF . (2002). J. Biol. Chem., 277, 1531–1537.
Bacus SS, Gudkov AV, Lowe M, Lyass L, Yung Y, Komarov AP, Keyomarsi K, Yarden Y and Seger R . (2001). Oncogene, 20, 147–155.
Bain J, McLauchlan H, Elliot M and Cohen P . (2003). Biochem. J., 371, 199–204.
Band V, Zajchowski D, Swisshelm K, Trask D, Kulesa V, Cohen C, Connolly J and Sager R . (1990). Cancer Res., 50, 7351–7357.
Bates S, Ryan KM, Phillips AC and Vousden KH . (1998). Oncogene, 17, 1691–1703.
Bennett BL, Sasaki DT, Murray BW, O'Leary EC, Sakata ST, Xu W, Leisten JC, Motiwala A, Pierce S, Satoh Y, Bhagwat SS, Manning AM and Anderson DW . (2001). Proc. Natl. Acad. Sci. USA, 98, 13681–13686.
Brummelkamp TR, Bernards R and Agami R . (2002). Science, 296, 550–553.
Chang B-D, Broude EV, Fang J, Kalinichenko TV, Abdryashitov R, Poole JC and Roninson IB . (2000). Oncogene, 19, 2165–2170.
Chen Y-R, Meyer CF and Tna T-H . (1996). J. Biol. Chem., 271, 631–634.
Courtois S, Verhaegh G, North S, Luciani M-G, Lassus P, Hibner U, Oren M and Hainaut P . (2002). Oncogene, 21, 6722–6728.
Flatt PM, Tang LJ, Scatena CD, Szak ST and Pietenpol JA . (2000). Mol. Cell. Biol., 20, 4210–4223.
Fuchs SY, Adler V, Buschmann T, Yin Z, Wu X, Jones SN and Ronai Z . (1998a). Genes Dev., 12, 2658–2663.
Fuchs SY, Adler V, Pincus MR and Ronai Z . (1998b). Proc. Natl. Acad. Sci. USA, 95, 10541–10546.
Fuchs SY, Fried VA and Ronai Z . (1998c). Oncogene, 17, 1483–1490.
Gupta S, Barrett T, Whitmarsh AJ, Cavanagh J, Sluss HK, Derijard B and Davis RJ . (1996). EMBO J., 15, 2760–2770.
Haq R, Brenton JD, Takahashi M, Finan D, Finkielsztein A, Damaraju S, Rottapel R and Zanke B . (2002). Cancer Res., 62, 5076–5082.
Hibi M, Lin A, Smeal T, Minden A and Karin M . (1993). Genes Dev., 7, 2135–2148.
Hildesheim J, Bulavin DV, Anver MR, Alvord WG, Hollander MC, Vardanian L and Fornace Jr AJ . (2002). Cancer Res., 62, 7305–7315.
Hochedlinger K, Wagner EF and Sabapathy K . (2002). Oncogene, 21, 2441–2445.
Jin S, Antinore MJ, Lung F-D, Dong X, Zhao H, Fan F, Colchagie AM, Blanck P, Roller PP, Fornace Jr AJ and Zhan Q . (2000). J. Biol. Chem., 275, 16602–16608.
Jochum W, Passegue E and Wagner WF . (2001). Oncogene, 20, 2401–2412.
Kim G-Y, Mercer SE, Ewton DZ, Yan Z, Jin K and Friedman E . (2002). J. Biol. Chem., 277, 29792–29802.
MacCorke-Chosnek RA, VanHooser A, Goodrich DW, Brinkley BR and Tan H . (2001). Biochem. Biophys. Res. Commun., 289, 173–180.
Mamay CL, Mingo-Sion AM, Wolf DM, Molina MD and Van Den Berg CL . (2003). Oncogene, 22, 602–614.
Minden A and Karin M . (1997). Biochim. Biophys. Acta, 1333, F85–F104.
Monno S, Newman MV, Cook M and Lowe W . (2000). Endocrinology, 141, 544–550.
Motwani M, Li X-K and Schwartz GK . (2000). Clin. Cancer Res., 6, 924–932.
Osborn MT and Chambers TC . (1996). J. Biol. Chem., 271, 30950–30955.
Patel R, Bartosch B and Blank JL . (1998). J. Cell Sci., 111, 2247–2255.
Potapova O, Gorospe M, Bost F, Dean NM, Gaarde WA, Mercola D and Holbrook NJ . (2000a). J. Biol. Chem., 275, 24767–24775.
Potapova O, Gorospe M, Dougherty RH, Dean NM, Gaarde WA and Holbrook NJ . (2000b). Mol. Cell. Biol., 20, 1713–1722.
Shaulian E and Karin M . (2001). Oncogene, 20, 2390–2400.
Shieh SY, Ikeda M, Taya Y and Prives C . (1997). Cell, 91, 325–334.
Shim J, Lee H, Park J, Kim H and Choi E-J . (1996). Nature, 381, 804–807.
Tournier C, Hess P, Yang DD, Xu J, Turner TK, Nimnual A, Bar-Sagi D, Jones SN, Flavell RA and Davis RJ . (2000). Science, 288, 870–874.
Vogt PK . (2001). Oncogene, 20, 2365–2377.
Wang T-H, Eang H-S, Ichijo H, Giannakakou P, Foster JS, Fojo T and Wimalasena J . (1998). J. Biol. Chem., 273, 4928–4936.
Wang XW, Zhan Q, Coursen JD, Kahn MA, Kontny HU, Yu L, Hollander MC, O'Connor PM, Fornace Jr AJ and Harris CC . (1999). Proc. Natl. Acad. Sci. USA, 96, 3706–3711.
Woodgett JR, Avruch J and Kyriakis J . (1996). Cancer Surv., 27, 127–138.
Xue Y, Ramaswamy NT, Hong X and Pelling JC . (2003). Mol. Carcinogen., 36, 38–44.
Yang Q, Manicone A, Coursen JD, Linke SP, Nagashima M, Forgues M and Wang XW . (2000). J. Biol. Chem., 275, 36892–36898.
Acknowledgements
We thank Karen Helm and Michael Ashton from the UCCC Flow Cytometry Core for their assistance with FACS analysis presented herein. This work was supported in part by the Avon Breast Cancer Foundation and United States Army Medical Research and Command Grant DAMD17-99-9142 (to CLVDB). The content of this information does not necessarily reflect the position or the policy of the Government, and no official endorsement should be inferred.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mingo-Sion, A., Marietta, P., Koller, E. et al. Inhibition of JNK reduces G2/M transit independent of p53, leading to endoreduplication, decreased proliferation, and apoptosis in breast cancer cells. Oncogene 23, 596–604 (2004). https://doi.org/10.1038/sj.onc.1207147
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1207147
- Springer Nature Limited
Keywords
This article is cited by
-
JNK signaling pathway regulates the development of ovaries and synthesis of vitellogenin (Vg) in the swimming crab Portunus trituberculatus
Cell Stress and Chaperones (2020)
-
Cytostatic and Anti-tumor Potential of Ajwa Date Pulp against Human Hepatocellular Carcinoma HepG2 Cells
Scientific Reports (2019)
-
The ERK and JNK pathways in the regulation of metabolic reprogramming
Oncogene (2019)
-
Endogenous c-Jun N-terminal kinase (JNK) activity marks the boundary between normal and malignant granulosa cells
Cell Death & Disease (2018)
-
BPGAP1 spatially integrates JNK/ERK signaling crosstalk in oncogenesis
Oncogene (2017)