Abstract
OBJECTIVE: Intravenous steroids improve the respiratory course in ventilator-dependent preterm infants but have adverse effects. We hypothesized that inhaled steroids would be as effective, but with less systemic effects.
STUDY DESIGN: We conducted a randomized, prospective trial comparing inhaled beclomethasone, either 400 or 800 μg/d, to intravenous dexamethasone in preterm infants dependent on conventional mechanical ventilation and supplemental oxygen at 2 weeks of age.
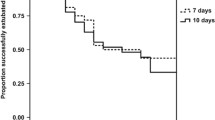
RESULTS: Seventy-eight infants were randomized. By day three of therapy, the intravenous steroid group had significantly decreased ventilator and oxygen requirements compared to either inhaled group. The inhaled 800-μg/d group trended toward more rapid decreases in ventilator and oxygen requirements than the 400-μg/d group. By day 14, all groups had similar reductions in ventilator and oxygen requirements. The incidence of adverse effects did not differ between groups.
CONCLUSIONS: In this small, randomized study, inhaled steroids conferred no advantages to intravenous steroids in the management of ventilator-dependent preterm infants.
Similar content being viewed by others
References
Cummings JJ, D'Eugenio DB, Gross SJ . Controlled trial of dexamethasone in preterm infants at high risk for bronchopulmonary dysplasia N Engl J Med 1989 320: 1505–10
Ng PC . The effective and side effects of dexamethasone in preterm infants with bronchopulmonary dysplasia Arch Dis Child 1993 68: 3 Spec No 330–6
Wilson NM, Silverman M . Treatment of acute, episodic asthma in preschool children using intermittent high dose inhaled steroids at home Arch Dis Child 1990 65: 4 407–10
Konig P, Shatley M, Levine C, Mawhinney TP . Clinical observations of nebulized flunisolide in infants and young children with asthma and bronchopulmonary dysplasia Pediatr Pulmonol 1992 13: 209–14
LaForce WR, Brudno DS . Controlled trial of beclomethasone diproprionate by nebulization in oxygen and ventilator dependent infants J Pediatr 1993 122: 285–8
Cloutier MM . Nebulized steroid therapy in bronchopulmonary dysplasia Pediatr Pulmonol 1993 15: 2 111–6
Kamada AK, Szefler SJ . Inhaled therapy in infants: why not nebulize glucocorticoids? Pediatr Pulmonol 1992 13: 4 198–9
Gal P, Ransom JR, Carlos RQ et al. Beclomethasone for treating premature infants with BPD: Guidelines for patients selection (letter) J Pediatr 1993 123: 3 490–1
O'Callaghan C, Hardy J, Stammers J, Stephenson TJ, Hull D . Evaluation of techniques for delivery of steroids to lungs of neonates using a rabbit model Arch Dis Child 1992 67: 20–4
Rozycki H, Bryon P, Dailey K et al. Evaluation of a system for delivery of inhaled beclomethasone to intubated neonates Dev Pharmacol Ther 1991 16: 65–70
Tan KL . Blood pressure in very low birth weight infants in the first seventy days of life J Pediatr 1988 112: 266–70
Wilson DM, Baldwin RB, Ariagno RL . A randomized, placebo-controlled trial of effects of dexamethasone on hypothalamic–pituitary–adrenal axis in preterm infants J Pediatr 1988 113: 764–8
Thomas S, Murphy JF, Dyas J, Ryalls M, Hughes IA . Response to ACTH in the newborn Arch Dis Child 1986 61: 57–60
Mammel MC, Green TP, Johnson DE, Thompson TR . Controlled trial of dexamethasone therapy in infants with bronchopulmonary dysplasia Lancet 1983 i: 1356–8
Dimitriou G, Greenough A, Giffin FJ, Kavadia V . Inhaled versus systemic steroids in chronic oxygen dependency of preterm infants Eur J Pediatr 1997 156: 51–5
Avent ML, Gal P, Ransom JL . The role of inhaled steroids in the treatment of bronchopulmonary dysplasia Neonat Network 1994 13: 3 63–9
Groneck P, Goetze-Spear B, Speer CP . Effects of inhaled beclomethasone compared to systemic dexamethasone on lung inflammation in preterm infants at risk of chronic lung disease Pediatr Pulmonol 1999 27: 383–7
Ford LR, Willi SM, Hollis BW, Wright NM . Suppression and recovery of the neonatal hypothalamic–pituitary–adrenal axis after prolonged dexamethasone use J Pediatr 1997 131: 722–6
Cole CH, Shah B, Abbasi S et al. Adrenal function in premature infants during inhaled beclomethasone therapy J Pediatr 1999 135: 65–70
Korte C, Styne D, Merrit A, Mayes D, Wertz A, Helbock HJ . Adrenocortical function in the very low birthweight infant: improved testing sensitivity and association with neonatal outcome J Pediatr 1996 128: 257–63
O'Shea TM, Kothadia JM, Klinepeter KL et al. Randomized placebo-controlled trial of a 42-day tapering course of dexamethasone to reduce the duration of ventilator dependency in very low birth weight infants: outcome of study participants at 1-year adjusted age Pediatr 1999 104: 415–21
Yeh TF, Lin YJ, Huang CC et al. Early dexamethasone therapy in preterm infants: a follow-up study Pediatr 1998 101: 5 e7
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Suchomski, S., Cummings, J. A Randomized Trial of Inhaled Versus Intravenous Steroids in Ventilator-Dependent Preterm Infants. J Perinatol 22, 196–203 (2002). https://doi.org/10.1038/sj.jp.7210705
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.jp.7210705
- Springer Nature America, Inc.
This article is cited by
-
Airway administration of corticosteroids for prevention of bronchopulmonary dysplasia in premature infants: a meta-analysis with trial sequential analysis
BMC Pulmonary Medicine (2017)
-
Glucocorticoids in the treatment of neonatal meconium aspiration syndrome
European Journal of Pediatrics (2011)