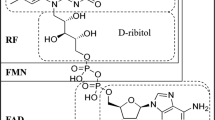
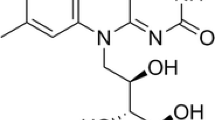
A novel process for riboflavin production using a recombinant Bacillus subtilis strain has been developed. Here we describe a down-stream processing procedure to obtain riboflavin qualities having a minimal content of 96% (‘feed-grade’) and 98% (‘food/pharma-grade’) riboflavin, respectively. Compared to riboflavin produced by chemical synthesis, products with improved chemical purity were obtained. All compounds representing more than 0.1% of the final products were identified. Feed-grade riboflavin material ex fermentation contained small amounts of amino acids and amino sugars and the biosynthetic riboflavin precursor dimethyl-ribityl-lumazine. All other side products found were derived from riboflavin, resulted from the purification procedure and were also found in riboflavin obtained by chemical synthesis. The Bacillus-produced riboflavin does not contain DNA. The data presented here were used to obtain product approval for the commercial application in the USA, Japan and the UK.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received 22 July 1998/ Accepted in revised form 8 November 1998
Rights and permissions
About this article
Cite this article
Bretzel, W., Schurter, W., Ludwig, B. et al. Commercial riboflavin production by recombinant Bacillus subtilis: down-stream processing and comparison of the composition of riboflavin produced by fermentation or chemical synthesis. J Ind Microbiol Biotech 22, 19–26 (1999). https://doi.org/10.1038/sj.jim.2900604
Issue Date:
DOI: https://doi.org/10.1038/sj.jim.2900604