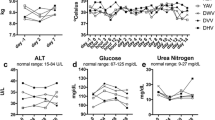
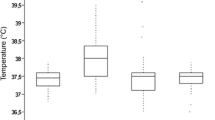
Abstract
We tested the canarypox virus vector ALVAC and the genetically attenuated vaccinia virus vector NYVAC as vehicles for achieving local immunomodulation in domestic animals bearing spontaneous tumours. Following intratumoral administration of ALVAC-, or NYVAC-luciferase in dogs with melanoma, it was demonstrated that viral recombinants remained localized along the needle track, with no virus detectable in the periphery of the tumour. Given these distribution characteristics and their well-documented safety profile, ALVAC- or NYVAC-based recombinants expressing feline or human IL2, respectively, were administered to domestic cats, in order to prevent the recurrence of spontaneous fibrosarcomas. In the absence of immunotherapy, tumour recurrence was observed in 61% of animals within a 12-month follow-up period after treatment with surgery and iridium-based radiotherapy. In contrast, only 39 and 28% of cats receiving either NYVAC-human IL2 or ALVAC-feline IL2, respectively, exhibited tumour recurrences. Based on such results, and in the context of ongoing clinical studies conducted in humans, we discuss the utilization of ALVAC- or NYVAC-based recombinants as viable therapeutic modalities for local immunotherapy or therapeutic vaccination against cancer, both in humans and companion animals.




Similar content being viewed by others
References
Ruby J, Ramsay A, Karupiah G, Ramshaw I . Recombinant virus vectors that coexpress cytokines – A new vaccine strategy. Vaccine Res 1992; 4: 347–356.
Cooney EL et al. Enhanced immunity to human immunodeficiency virus (HIV) envelope elicited by a combined vaccine regimen consisting of priming with a vaccinia recombinant expressing HIV envelope and boosting with gp160 protein. Proc Natl Acad Sci USA 1993; 90: 1882–1886.
Perkus ME, Tartaglia J, Paoletti E . Poxvirus-based vaccine candidates for cancer, AIDS, and infectious diseases. J Leukocyte Biol 1995; 58: 1–13.
Moss B . Genetically engineered pox viruses for recombinant gene expression, vaccination and safety. Proc Natl Acad Sci USA 1996; 93: 11341–11348.
Paoletti E . Application of poxvirus vectors to vaccination: an update. Proc Natl Acad Sci 1996; 93: 11349–11353.
Bonnet MC et al. Recombinant viruses as a tool for therapeutic vaccination against human cancers. Immunol Lett 2000; 74: 11–25.
Meyer H, Sutter G, Mayr A . Mapping of deletions in the genome of the highly attenuated vaccinia virus MVA and their influence on virulence. J Gen Virol 1991; 72: 1031–1038.
Tartaglia J et al. NYVAC: highly attenuated strain of vaccinia virus. Virology 1992; 188: 217–232.
Paoletti E, Tartaglia J, Taylor J . Safe and effective poxvirus vectors – NYVAC and ALVAC. Dev Biol Stand 1994; 82: 65–69.
Plotkin SA et al. The safety and use of canarypox vectored vaccines. Dev Biol Stand 1995; 84: 165–170.
Cadoz M et al. Immunisation with canarypox virus expressing rabies glycoproteins. Lancet 1992; 339: 1429–1432.
Fries LF et al. Human safety and immunogenicity of a canarypox–rabies glycoprotein recombinant vaccine: an alternative poxvirus vector system. Vaccine 1996; 14: 428–434.
Excler J-L, Plotkin S . The prime-boost concept applied to HIV preventive vaccines. AIDS 1997; 11 (Suppl A): S127–137.
Ferrari G et al. Replication-defective canarypox (ALVAC) vectors effectively activate anti-human immunodeficiency virus-1 cytotoxic T lymphocytes present in infected patients: implications for antigen-specific immunotherapy. Blood 1997; 90: 2406–2416.
Konishi E et al. Induction of Japanese encephalitis virus-specific cytotoxic T lymphocytes in humans by poxvirus-based JE vaccine candidates. Vaccine 1998; 16: 842–849.
Ockenhouse CF et al. Phase I/IIa safety, immunogenicity, and efficacy trial of NYVAC-Pf7, a pox-vectored, multiantigen, multistage vaccine candidate for Plasmodium falciparum malaria. J Infect Dis 1998; 177: 1664–1673.
Tartaglia J et al. Therapeutic vaccines against melanoma and colorectal cancer. Vaccine 2001; 19: 2571–2575.
Moingeon P . Cancer vaccines. Vaccine 2001; 19: 1305–1326.
Hurpin C et al. The mode of presentation and route of administration are critical for the induction of immune responses to p53 and antitumour immunity. Vaccine 1998; 16: 208–215.
Puisieux I et al. Canarypox virus-mediated IL12 gene transfer into murine mammary adenocarcinoma induces tumour suppression and long term antitumoural immunity. Hum Gene Ther 1998; 9: 2488–2492.
Odin L et al. Canarypox vitus expressing wild type p53 for gene therapy in murine tumours mutated in p53. Cancer Gene Ther 2000; 8: 1–12.
Van Dyke T, Jacks T . Cancer modeling in the modern era: progress and challenges. Cell 2002; 108: 135–144.
Turrel J, Korlik P . Techniques of afterloading iridium-192 interstitial brachytherapy in veterinary medicine. Vet Radiol 1983; 24: 278–283.
Quintin-Colonna F et al. Gene therapy of spontaneous canine melanoma and feline fibrosarcoma by intratumoural administration of histoincompatible cells expressing human interleukin 2. Gene Therapy 1996; 3: 1104–1112.
Siemens D et al. Evaluation of gene transfer efficiency by viral vectors to murine bladder epithelium. J Urol 2001; 65: 667–671.
Kawakita M et al. Effect of canarypox virus (ALVAC)-mediated cytokine gene expression on murine prostate tumour growth. J Natl Cancer Inst 1997; 89: 428–436.
Rosenberg SA et al. Treatment of patients with metastatic melanoma with autologous tumour infiltrating lymphocytes and interleukin 2. J Natl Cancer Inst 1994; 86: 1159–1166.
Roth J et al. p53 as a target for cancer vaccines: recombinant canarypox virus vectors expressing p53 protect mice against lethal tumour cell challenge. Proc Natl Acad Sci USA 1996; 930: 4781–4786.
Hodge JW, McLaughlin JP, Kantor JA, Schlom J . Diversified prime and boost protocols using recombinant vaccinia virus and recombinant nonreplicating avian pox virus to enhance T-cell immunity and antitumour responses. Vaccine 1997; 16: 759–768.
Marshall JL et al. Phase I study in cancer patients of a replication-defective avipox recombinant vaccine that expresses human carcinoembryonic antigen. J Clin Oncol 1999; 17: 332–333.
Zhu M et al. Specific cytolytic T-cell responses to human CEA from patients immunized with recombinant avipox-CEA vaccine. Clin Cancer Res 2000; 6: 24–33.
Van der Burg SH et al. Induction of p53-specific immune responses in colorectal cancer patients receiving a recombinant ALVAC-p53 candidate vaccine. Clin Cancer Res 2002; 8: 1019–1027.
Von Mehren M et al. Pilot study of a dual gene recombinant avipox vaccine containing both carcinoembryonic antigen (CEA) and B7.1 transgenes in patients with recurrent CEA-expressing adenocarcinomas. Clin Cancer Res 2000; 6: 2219–2228.
Perkus M, Limbach K, Paoletti E . Cloning and expression of foreign genes in vaccinia virus using a host range selection system. J Virol 1989; 63: 3829–3836.
Konishi E et al. A highly attenuated range-restricted vaccinia virus strain, NYVAC, encoding the prM, E and NS1 genes of Japonese encephalitis virus prevents JEV viremia in swine. Virology 1992; 190: 454–458.
Owen L . World Health Organization TNM classification of tumours in domestic animals. In: Harmer M (ed). TNM Classification of Malignant Tumours. UICC: Geneva, 1980, pp 448–487.
Acknowledgements
We acknowledge Jean-Louis Bachan, Marie-Clotilde Bernard, Emmanuelle Bonnet-Piro, Christophe Charnay, Maryse Philipona and Christelle Serraille, at Aventis Pasteur, Marcy l'Etoile, France, as well as Joëlle Gibouin (ENVA, Maisons Alfort, France) for their excellent technical assistance. We also thank Drs Bernard Cornet and Olivier Level for supplying reagents and Dr Florence Boudet for serological analyses. We are thankful as well to Drs Michel Chevalier and François Verdier for critically reviewing the manuscript.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jourdier, TM., Moste, C., Bonnet, MC. et al. Local immunotherapy of spontaneous feline fibrosarcomas using recombinant poxviruses expressing interleukin 2 (IL2). Gene Ther 10, 2126–2132 (2003). https://doi.org/10.1038/sj.gt.3302124
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3302124
- Springer Nature Limited
Keywords
This article is cited by
-
Genetic Therapy in Veterinary Medicine
BioNanoScience (2022)
-
Safety studies and viral shedding of intramuscular administration of oncolytic vaccinia virus TG6002 in healthy beagle dogs
BMC Veterinary Research (2020)
-
Utilizing Microbes to Treat Naturally Occurring Cancer in Veterinary Species
Current Clinical Microbiology Reports (2019)
-
IL-12 based gene therapy in veterinary medicine
Journal of Translational Medicine (2012)
-
Oncolytic virotherapy in veterinary medicine: current status and future prospects for canine patients
Journal of Translational Medicine (2012)