Abstract
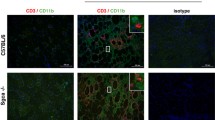
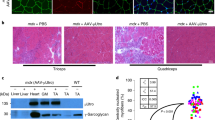
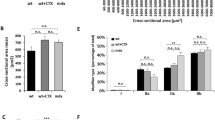
Duchenne muscular dystrophy (DMD) is an X-linked, lethal muscular disorder caused by a defect in the DMD gene. AAV vector-mediated micro-dystrophin cDNA transfer is an attractive approach to treatment of DMD. To establish effective gene transfer into skeletal muscle, we examined the transduction efficiency of an AAV vector in skeletal muscles of dystrophin-deficient mdx mice. When an AAV vector encoding the LacZ gene driven by a CMV promoter (AAV-CMVLacZ) was introduced, β-galactosidase expression markedly decreased in mdx muscle 4 weeks after injection due to immune responses against the transgene product. We also injected AAV-CMVLacZ into skeletal muscles of mini-dystrophin-transgenic mdx mice (CVBA3’), which show ameliorated phenotypes without overt signs of muscle degeneration. AAV vector administration, however, evoked substantial immune responses in CVBA3’ muscle. Importantly, AAV vector using muscle-specific MCK promoter also elicited responses in mdx muscle, but at a considerably later period. These results suggested that neo-antigens introduced by AAV vectors could evoke immune reactions in mdx muscle, since increased permeability allowed a leakage of neo-antigens from the dystrophin-deficient sarcolemma of muscle fibers. However, resident antigen-presenting cells, such as myoblasts, myotubes and regenerating immature myofibers, might also play a role in the immune response.








Similar content being viewed by others
References
Emery AEH . Duchenne Muscular Dystrophy 2nd edn Oxford University Press 1993
Rogot T et al. Efficient adenovirus-mediated transfer of a human minidystrophin gene to skeletal muscle of mdx mice Nature 1993 361: 647–650
Acsadi G et al. Dystrophin expression in muscles of mdx mice after adenovirus-mediated in vivo gene transfer Hum Gene Ther 1996 7: 129–140
Kochanek S et al. A new adenoviral vector: replacement of all viral coding sequences with 28 kb of DNA independently expressing both full-length dystrophin and β-galactosidase Proc Natl Acad Sci USA 1996 93: 5731–5736
Tuner G, Duncley MG, Dickson G . Gene therapy of Duchenne muscular dystrophy Brown SC, Lucy JA (eds); Dystrophin: Gene, Protein and Cell Biology Cambridge University Press 1997 pp 274–309
Chen H-H et al. DNA from both high-capacity and first generation adenoviral vectors remains intact in skeletal muscle Hum Gene Ther 1999 10: 365–373
Acsadi G et al. Human dystrophin expression in mdx mice after intramuscular injection of DNA constructs Nature 1991 352: 815–818
Gussoni E, Blau HM, Kunkel LM . The fate of individual myoblasts after transplantation into muscles of DMD patients Nature Med 1997 3: 970–977
Gussoni E et al. Dystrophin expression in mdx mouse restored by stem cell transplantation Nature 1999 40: 390–394
Rando TA, Disatnik MH, Zhou LZ . Rescue of dystrophin expression in mdx mouse muscle by RNA/DNA oligonucleotides Proc Natl Acad Sci USA 2000 97: 5363–5368
Bartlett RJ et al. In vivo targeted repair of a point mutation in the canine dystrophin gene by a chimeric RNA/DNA oligonucleotide Nat Biotechnol 2000 18: 615–622
Dunckley MG et al. Modification of splicing in dystrophin gene in cultured Mdx muscle cells by antisense oligonucleotides Hum Mol Genet 1998 7: 1083–1090
Barton-Davis ER et al. Aminoglycoside antibiotics restore dystrophin function to skeletal muscles of mdx mice J Clin Invest 1999 104: 375–381
Wagner KR et al. Gentamicin treatment of Duchenne and Becker muscular dystrophy due to nonsense mutations Ann Neurol 2001 49: 706–711
Yamamoto K et al. Immune response to adenovirus-delivered antigens upregulates utrophin and results in mitigation of muscle pathology in mdx mice Hum Gene Ther 2000 11: 669–680
Xiao X, Li J, Samulski RJ . Efficient long-term gene transfer into muscle tissue of immunocompetent mice by adeno-associated virus vector J Virol 1996 70: 8098–8108
Kessler PD et al. Gene delivery to skeletal muscle results in sustained expression and systemic delivery of a therapeutic protein Proc Natl Acad Sci USA 1996 93: 14082–14087
Fisher KJ et al. Recombinant adeno-associated virus for muscle directed gene therapy Nat Med 1997 3: 306–312
Kaplitt MG et al. Long-term gene expression and phenotypic correction using adeno-associated virus vectors in mammalian brain Nat Genet 1994 8: 148–153
Jooss K, Yang Y, Fisher KJ, Wilson JM . Transduction of dendritic cells by DNA viral vectors directs the immune response to transgene products in muscle cells J Virol 1998 72: 4212–4223
Greelish JP et al. Stable restoration of the sarcoglycan complex in dystrophic muscle perfused with histamine and a recombinant adeno-associated viral vector Nat Med 1999 5: 439–443
Li J et al. rAAV vector-mediated sarcoglycan gene transfer in a hamster model for limb girdle muscular dystrophy Gene Therapy 1999 6: 74–82
Cordier L et al. Rescue of skeletal muscles of γ-sarcoglycan-deficient mice with adeno-associated virus-mediated gene transfer Mol Ther 2000 1: 119–129
Cordier L et al. Muscle-specific promoters may be necessary for adeno-associated virus-mediated gene transfer in the treatment of muscular dystrophies Hum Gene Ther 2001 12: 205–215
England SB et al. Very mild muscular dystrophy associated with the deletion of 46% of dystrophin Nature 1990 343: 180–182
Phelps SF et al. Expression of full-length and truncated dystrophin mini-gene in transgenic mdx mice Hum Gene Ther 1995 4: 1251–1258
Yuasa K et al. Effective restoration of dystrophin-associated proteins in vivo by adenovirus-mediated transfer of truncated dystrophin cDNAs FEBS Lett 1998 425: 329–336
Wang B, Li J, Xiao X . Adeno-associated virus vector carrying human minidystrophin genes effectively ameliorates muscular dystrophy in mdx mouse model Proc Natl Acad Sci USA 2000 97: 13714–13719
Straub V, Rafael JA, Chamberlain JS, Campbell KP . Animal models for muscular dystrophy show different patterns of sarcolemmal disruption J Cell Biol 1997 139: 375–385
Zhang Y, Chirmule N, Gao G-P, Wilson J . CD40 ligand-dependent activation of cytotoxic T lymphocytes by adeno-associated virus vectors in vivo: role of immature dendritic cells J Virol 2000 74: 8003–8010
Witmer-Pack MD et al. Tissue distribution of the DEC-205 protein that is detected by the monoclonal antibody NLDC-145 Cell Immunol 1995 163: 157–162
Khan MA . Corticosteroid therapy in Duchenne muscular dystrophy J Neurol Sci 1993 120: 8–14
Pimorady-Esfahani A, Grounds MD, McMenamin PG . Macrophages and dendritic cells in normal and regenerating murine skeletal muscle Muscle Nerve 1997 20: 158–166
Hartigan-O'Connor D et al. Immune evasion by muscle-specific gene expression in dystrophic muscle Mol Ther 2001 4: 525–533
Hohlfeld R, Engel AG . The immunobiology of muscle Immunol Today 1994 15: 269–274
Tinsley JM et al. Amelioration of the dystrophic phenotype of mdx mice using a truncated utrophin transgene Nature 1996 28: 349–353
Xiao X, Li J, Samulski RJ . Production of high-titer recombinant adeno-associated virus vectors in the absence of helper adenovirus J Virol 1998 72: 2224–2232
Shield MA et al. E-box sites and a proximal regulatory region of the muscle creatine kinase gene differentially regulate expression in diverse skeletal muscles and cardiac muscle of transgenic mice Mol Cell Biol 1996 16: 5058–5068
Snyder R, Xiao X, Samulski RJ . Production of recombinant adeno-associated viral vectors Dracopoli N (eds); Current Protocols in Human Genetics John Wiley 1996 pp 1–2
Ishii A et al. Effective adenovirus-mediated gene expression in adult murine skeletal muscle Muscle Nerve 1999 22: 592–599
Acknowledgements
We are grateful to Dr M Yoshida for providing mdx mice. We also thank colleagues in our laboratory for useful discussion and suggestions on this work. This work is supported by Grants-in-Aid for Scientific Research for Center of Excellence, Research on Nervous and Mental Disorders (10B-1, 13B-1) Health Sciences Research Grants for Research on the Human Genome and Gene Therapy (H10-genome-015, H13-genome-001), for Research on Brain Science (H12-Brain-028) from the Ministry of Heath, Labor and Welfare, Grant-in-Aids for Scientific Research (10557065, 11470153, 11170264 and 14657158) from the Ministry of Education, Culture, Sports, Science and Technology, and a Research Grant from Human Frontier Science Project.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Yuasa, K., Sakamoto, M., Miyagoe-Suzuki, Y. et al. Adeno-associated virus vector-mediated gene transfer into dystrophin-deficient skeletal muscles evokes enhanced immune response against the transgene product. Gene Ther 9, 1576–1588 (2002). https://doi.org/10.1038/sj.gt.3301829
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.gt.3301829
- Springer Nature Limited
Keywords
This article is cited by
-
Molecular Approaches for the Treatment of Pompe Disease
Molecular Neurobiology (2020)
-
Fukutin is prerequisite to ameliorate muscular dystrophic phenotype by myofiber-selective LARGE expression
Scientific Reports (2015)
-
Intra-Amniotic rAAV-Mediated Microdystrophin Gene Transfer Improves Canine X-Linked Muscular Dystrophy and May Induce Immune Tolerance
Molecular Therapy (2015)
-
Plasmapheresis Eliminates the Negative Impact of AAV Antibodies on Microdystrophin Gene Expression Following Vascular Delivery
Molecular Therapy (2014)
-
AAV-mediated Overexpression of Human α7 Integrin Leads to Histological and Functional Improvement in Dystrophic Mice
Molecular Therapy (2013)