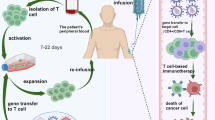
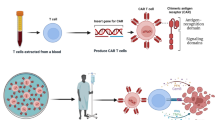
Abstract
Immune-isolation of nonautologous cells with microencapsulation protects these cells from graft rejection, thus allowing the same recombinant therapeutic cell line to be implanted in different recipients. This approach was successful in treating HER2/neu-expressing tumors in mice by delivering an interleukin-2 fusion protein (sFvIL-2), or angiostatin. However, treatment with interleukin-2 led to profuse inflammation, while angiostatin delivery did not result in long-term tumor suppression, in part due to endothelial cell-independent neovascularization (vascular mimicry). We hypothesize that coencapsulating the two producer cells in the same microcapsules may enhance the efficacy and ameliorate the above side effects. Hence, B16-F0/neu tumor-bearing mice were implanted with sFvIL-2- and angiostatin-secreting cells coencapsulated in the same alginate-poly-L-lysine-alginate microcapsules. However, this protocol only produced an incremental but not synergistic improvement, as measured with greater tumor suppression and improved survival. Compared to the single sFvIL-2 treatment, the coencapsulation protocol showed improved efficacy associated with: mobilization of sFvIL-2 from the spleen; a higher level of cytokine delivery systemically and to the tumors; increased tumor and tumor-associated endothelial cell apoptosis; and a reduced host inflammatory response. However, compared to the single angiostatin treatment, the efficacy was reduced, primarily due to a “bystander” effect in which the angiostatin-secreting cells suffered similar transgene silencing as the coencapsulated cytokine-secreting cells. Nevertheless, the level of “vascular mimicry” of the single angiostatin treatment was significantly reduced. Hence, while there was no synergy in efficacy, an incremental improvement and some reduction in undesirable side effects of inflammation and vascular mimicry were achieved over the single treatments.








Similar content being viewed by others
References
al Hendy A, Hortelano G, Tannenbaum GS, Chang PL . Correction of the growth defect in dwarf mice with nonautologous microencapsulated myoblasts — an alternate approach to somatic gene therapy. Hum Gene Ther. 1995;6:165–175.
Ross CJ, Ralph M, Chang PL . Somatic gene therapy for a neurodegenerative disease using microencapsulated recombinant cells. Exp Neurol. 2000;166:276–286.
Ross CJ, Bastedo L, Maier SA, Sands MS, Chang PL . Treatment of a lysosomal storage disease, mucopolysaccharidosis VII, with microencapsulated recombinant cells. Hum Gene Ther. 2000;11:2117–2127.
Van Raamsdonk JM, Ross CJ, Potter MA, et al. Treatment of hemophilia B in mice with nonautologous somatic gene therapeutics. J Lab Clin Med. 2002;139:35–42.
Dautzenberg H, Schuldt U, Grasnick G, et al. Development of cellulose sulfate-based polyelectrolyte complex microcapsules for medical applications. Ann N Y Acad Sci. 1999;875:46–63.
Muller P, Jesnowski R, Karle P, et al. Injection of encapsulated cells producing an ifosfamide-activating cytochrome P450 for targeted chemotherapy to pancreatic tumors. Ann NY Acad Sci. 1999;880:337–351.
Lohr M, Muller P, Karle P, et al. Targeted chemotherapy by intratumour injection of encapsulated cells engineered to produce CYP2B1, an ifosfamide activating cytochrome P450. Gene Ther. 1998;5:1070–1078.
Kroger JC, Bergmeister H, Hoffmeyer A, et al. Intraarterial instillation of microencapsulated cells in the pancreatic arteries in pig. Ann NY Acad Sci. 1999;880:374–378.
Fujiki T, Futatsuki K, Akazawa S, et al. [Ifosfamide chemotherapy ineffective for advanced pancreatic carcinoma]. Gan To Kagaku Ryoho. 1997;24:569–572.
Cirone P, Bourgeois JM, Austin RC, Chang PL . A novel approach to tumor suppression with microencapsulated recombinant cells. Hum Gene Ther. 2002;13:1157–1166.
Folkman J . Tumor angiogenesis: therapeutic implications. N Engl J Med. 1971;285:1182–1186.
O'Reilly MS, Holmgren L, Shing Y, et al. Angiostatin: a novel angiogenesis inhibitor that mediates the suppression of metastases by a Lewis lung carcinoma. Cell. 1994;79:315–328.
MacDonald NJ, Murad AC, Fogler WE, Lu Y, Sim BK . The tumor-suppressing activity of angiostatin protein resides within kringles 1 to 3. Biochem Biophys Res Commun. 1999;264:469–477.
Cao Y, Ji RW, Davidson D, et al. Kringle domains of human angiostatin. Characterization of the anti-proliferative activity on endothelial cells. J Biol Chem. 1996;271:29461–29467.
Claesson-Welsh L, Welsh M, Ito N, et al. Angiostatin induces endothelial cell apoptosis and activation of focal adhesion kinase independently of the integrin-binding motif RGD. Proc Natl Acad Sci USA. 1998;95:5579–5583.
Eriksson K, Magnusson P, Dixelius J, Claesson-Welsh L, Cross MJ . Angiostatin and endostatin inhibit endothelial cell migration in response to FGF and VEGF without interfering with specific intracellular signal transduction pathways. FEBS Lett. 2003;536:19–24.
Stack MS, Gately S, Bafetti LM, Enghild JJ, Soff GA . Angiostatin inhibits endothelial and melanoma cellular invasion by blocking matrix-enhanced plasminogen activation. Biochem J. 1999;340:77–84.
Barendsz-Janson AF, Griffioen AW, Muller AD, Dam-Mieras MC, Hillen HF . In vitro tumor angiogenesis assays: plasminogen lysine binding site 1 inhibits in vitro tumor-induced angiogenesis. J Vasc Res. 1998;35:109–114.
Griscelli F, Li H, Bennaceur-Griscelli A, et al. Angiostatin gene transfer: inhibition of tumor growth in vivo by blockage of endothelial cell proliferation associated with a mitosis arrest. Proc Natl Acad Sci USA. 1998;95:6367–6372.
O'Reilly MS, Holmgren L, Chen C, Folkman J . Angiostatin induces and sustains dormancy of human primary tumors in mice. Nat Med. 1996;2:689–692.
Gately S, Twardowski P, Stack MS, et al. The mechanism of cancer-mediated conversion of plasminogen to the angiogenesis inhibitor angiostatin. Proc Natl Acad Sci USA. 1997;94:10868–10872.
Sim BK, O'Reilly MS, Liang H, et al. A recombinant human angiostatin protein inhibits experimental primary and metastatic cancer. Cancer Res. 1997;57:1329–1334.
Wu Z, O'Reilly MS, Folkman J, Shing Y . Suppression of tumor growth with recombinant murine angiostatin. Biochem Biophys Res Commun. 1997;236:651–654.
Cao Y, O'Reilly MS, Marshall B, Flynn E, Ji RW, Folkman J . Expression of angiostatin cDNA in a murine fibrosarcoma suppresses primary tumor growth and produces long-term dormancy of metastases. J Clin Invest. 1998;101:1055–1063.
Kirsch M, Strasser J, Allende R, Bello L, Zhang J, Black PM . Angiostatin suppresses malignant glioma growth in vivo. Cancer Res. 1998;58:4654–4659.
Griscelli F, Li H, Cheong C, et al. Combined effects of radiotherapy and angiostatin gene therapy in glioma tumor model. Proc Natl Acad Sci USA. 2000;97:6698–6703.
Sacco MG, Caniatti M, Cato EM, et al. Liposome-delivered angiostatin strongly inhibits tumor growth and metastatization in a transgenic model of spontaneous breast cancer. Cancer Res. 2000;60:2660–2665.
Joki T, Machluf M, Atala A, et al. Continuous release of endostatin from microencapsulated engineered cells for tumor therapy. Nat Biotechnol. 2001;19:35–39.
Read TA, Sorensen DR, Mahesparan R, et al. Local endostatin treatment of gliomas administered by microencapsulated producer cells. Nat Biotechnol. 2001;19:29–34.
Cirone P, Bourgeois JM, Chang PL . Antiangiogenic cancer therapy with microencapsulated cells. Hum Gene Ther. 2003;14:1065–1077.
Heike Y, Takahashi M, Ohira T, et al. Genetic immunotherapy by intrapleural, intraperitoneal and subcutaneous injection of IL-2 gene-modified Lewis lung carcinoma cells. Int J Cancer. 1997;73:844–849.
Peirone MA, Ross CJD, Hortelano G, Brash JL, Chang PL . Encapsulation of various recombinant mammalian cell types in different alginate microcapsules. J Biomed Mater Res. 1998;42:587–596.
Van Raamsdonk JM, Chang PL . Osmotic pressure test: a simple, quantitative method to assess the mechanical stability of alginate microcapsules. J Biomed Mater Res. 2001;54:264–271.
Benelli R, Morini M, Carrozzino F, et al. Neutrophils as a key cellular target for angiostatin: implications for regulation of angiogenesis and inflammation. FASEB J. 2002;16:267–269.
Tarui T, Miles LA, Takada Y . Specific interaction of angiostatin with integrin alpha(v)beta(3) in endothelial cells. J Biol Chem. 2001;276:39562–39568.
Griffioen AW, Damen CA, Blijham GH, Groenewegen G . Tumor angiogenesis is accompanied by a decreased inflammatory response of tumor-associated endothelium. Blood. 1996;88:667–673.
Griffioen AW, Damen CA, Mayo KH, et al. Angiogenesis inhibitors overcome tumor induced endothelial cell anergy. Int J Cancer. 1999;80:315–319.
Cirone P, Bourgeois JM, Shen F, Chang PL . Combined immuno- and antiangiogenic therapy of cancer with microencapsulated cells. Hum Gene Ther. 2004;15:945–959.
Cirone P, Saller RM, Chang PL . Immuno-isolation in oncology — a mini review. Curr Pharm Biotechnol. 2001;2:269–277.
Acknowledgements
We would like to thank the Canadian Breast Cancer Research Alliance and the Canadian Institute of Health Research for grant support and advice from Dr J Bourgeois in the pilot studies. Pasquale Cirone is the recipient of The David and Grace Prosser Scholarship, the McMaster University Graduate Scholarship and the Lee Nielson Roth Award for Medical Science (Cancer).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cirone, P., Shen, F. & Chang, P. A multiprong approach to cancer gene therapy by coencapsulated cells. Cancer Gene Ther 12, 369–380 (2005). https://doi.org/10.1038/sj.cgt.7700786
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700786
- Springer Nature America, Inc.
Keywords
This article is cited by
-
Design of an Adhesive Film-Based Microfluidic Device for Alginate Hydrogel-Based Cell Encapsulation
Annals of Biomedical Engineering (2020)
-
A Perspective on Bioactive Cell Microencapsulation
BioDrugs (2012)