Abstract
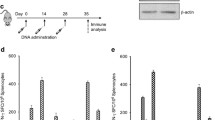
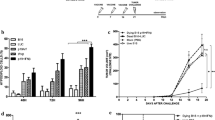
A mouse glioblastoma cell line, termed GL261, was shown to express high levels of proteins involved in melanin biosynthesis such as the tyrosinase-related protein-2 (TRP-2), which is commonly overexpressed in melanoma cells. Mice injected with GL261 cells developed a CD8+ T-cell response to TRP-2 and a DNA vaccine expressing human (h)TRP-2 induced CD8+ T cells that recognized TRP-2 expressed by GL261 cells indicating that this melanoma-associated antigen may be suited for active immunotherapy of glioblastoma. Mice vaccinated with a DNA vaccine expressing TRP-2 were partially protected against subcutaneous, intravenous, or intracerebral challenge with the glioblastoma cells. Vaccine-induced protection against intracerebral challenge required both CD4+ and CD8+ T cells. Vaccine efficacy was enhanced upon addition of IL-12 as a genetic adjuvant. These results indicate that this well-defined melanoma-associated antigen can induce an adaptive immune response, which limits the intracerebral progression of a glioblastoma.








Similar content being viewed by others
References
Mahaley MS, Mettlin C, Natarajan N, et al. National survey of patterns of care for brain-tumor patients. J Neurosurg. 1989;71:826–836.
Soiffer R, Lynch T, Mihm M, et al. Vaccination with irradiated autologous melanoma cells engineered to secrete human granulocyte–macrophage colony-stimulating factor generates potent antitumor immunity in patients with metastatic melanoma. Proc Natl Acad Sci USA. 1998;95:13141–13146.
Gilboa E, Nair SK, Lyerly HK . Immunotherapy of cancer with dendritic-cell-based vaccines. Cancer Immunol Immunother. 1998;46:82–87.
Tuting T, Gambotto A, DeLeo A, et al. Induction of tumor antigen-specific immunity using plasmid DNA immunization in mice. Cancer Gene Ther. 1999;6:73–80.
Hawkins WG, Gold JS, Blachere NE, et al. Xenogeneic DNA immunization in melanoma models for minimal residual disease. J Surg Res. 2002;102:137–143.
Bronte V, Apolloni E, Ronca R, et al. Genetic vaccination with “self” tyrosinase-related protein 2 causes melanoma eradication but not vitiligo. Cancer Res. 2000;60:253–258.
Mendiratta SK, Thai G, Eslahi, NK, et al. Therapeutic tumor immunity induced by polyimmunization with melanoma antigens gp100 and TRP-2. Cancer Res. 2001;61:859–863.
Tanaka M, Kaneda Y, Fujii S, et al. Induction of a systemic immune response by a polyvalent melanoma-associated antigen DNA vaccine for prevention and treatment of malignant melanoma. Mol Ther. 2002;5:291–299.
O I, Ku G, Ertl HC, et al. A dendritic cell vaccine induces protective immunity to intracranial growth of glioma. Anticancer Res. 2002;22:613–621.
Kowalczyk D, Wlazlo AP, Giles-Davis W, et al. Staining of antigen activated lymphocytes (SAAL): a highly specific method for amplification of tumor-specific CD8+ T cells. J Immunol Methods 2000;241:131–139.
Kleinman HK, McGarvey ML, Liotta LA, et al. Isolation and characterization of type IV procollagen, laminin and heparan sulfate proteoglycan from the EHS sarcoma. Biochemistry. 1982;21: 6188–6193.
Parkhurst MR, Fitzgerald EB, Southwood S, et al. Identification of a shared HLA-A*0201-restricted T-cell epitope from the melanoma antigen tyrosinase-related protein 2 (TRP2). Cancer Res. 1998;58:4895–4901.
Yokoyama K, Suzuki H, Yasumoto K, et al. Molecular cloning and functional analysis of a cDNA coding for human DOPAchrome tautomerase/tyrosinase-related protein-2. Biochem Biophys Acta. 1994;1217:317–321.
He Z, Wlazlo AP, Kowalczyk DW, et al. Viral recombinant vaccines to the E6 and E7 antigens of HPV-16. Virology. 2000;270:146–161.
Overwijk WW, Restifo NP . Autoimmunity and the immunotherapy of cancer: targeting the “self” to destroy the “other”. Crit Rev Immunol. 2000;20:433–450.
Steitz J, Bruck J, Steinbrink K, et al. Genetic immunization of mice with human tyrosinase-related protein 2: implications for the immunotherapy of melanoma. Int J Cancer. 2000;86:89–94.
Overwijk WW, Lee DS, Surman DR, et al. Vaccination with a recombinant vaccinia virus encoding a “self” antigen induces autoimmune vitiligo and tumor cell destruction in mice: requirement for CD4(+) T lymphocytes. Proc Acad Natl Sci USA. 1999;96:2982–2987.
Pertmer TM, Oran AE, Madorin CA, et al. Th1 genetic adjuvants modulate immune responses in neonates. Vaccine. 2001;19:1764–1771.
Deng H, Kowalczyk D, O I, et al. A modified DNA vaccine to p53 induces protective immunity to challenge with a chemically induced sarcoma cell line. Cell Immunol. 2002;215:20–31.
Pisetsky DS . Mechanisms of immune stimulation by bacterial DNA. Springer Sem Immunopathol 2000;22:21–33.
Krieg AM . CpG motifs in bacterial DNA and their immune effects. Annu Rev Immunol. 2002;20:709–760.
Tuting T, Gambotto A, DeLeo A, et al. Induction of tumor antigen-specific immunity using plasmid DNA immunization in mice. Cancer Gene Ther. 1999;6:73–80.
Le TP, Coonan KM, Hedstrom RC, et al. Safety, tolerability and humoral immune responses after intramuscular administration of a malaria DNA vaccine to healthy adult volunteers. Vaccine. 2000;18:1893–1901.
Wang R, Doolan, DL, Le TP, et al. Induction of antigen-specific cytotoxic T lymphocytes in humans by a malaria DNA vaccine. Science. 1998;282:476–480.
MacGregor RR, Boyer JD, Ugen KE, et al. First human trial of a DNA-based vaccine for treatment of human immunodeficiency virus type 1 infection: safety and host response. J Infect Dis. 1998;178:92–100.
Ulmer JB, Donnelly JJ, Parker SE, et al. Heterologous protection against influenza by injection of DNA encoding a viral protein. Science. 1993;259:1745–1749.
Xiang ZQ, Ertl HCJ . Manipulation of the immune response to a plasmid-encoded viral antigen by coinoculation with plasmid expressing cytokines. Immunity. 1995;2:129–136.
Barouch DH, Craiu A, Santra S, et al. Elicitation of high-frequency cytotoxic T-lymphocyte responses against both dominant and subdominant simian–human immunodeficiency virus epitopes by DNA vaccination of rhesus monkeys. J Virol. 2001;75:2462–2467.
Xiang ZQ, Pasquini S, Ertl HCJ . Induction of genital immunity by DNA priming and intranasal booster immunization with a replication-defective adenoviral recombinant. J Immunol. 1999;162:6716–6723.
Huang SK, Okamoto T, Morton DL, et al. Antibody responses in melanoma/melanocytes autoantigens in melanoma patients. J Int Dermatol. 1998;111:662–667.
Reynolds SR, Celis E, Sette A, et al. HLA-independent heterogeneity of CD8+ T cell responses to MAGE-3, Melan-A/MART-1, gp100, tyrosinase, MC1R and TRP-2 in vaccine-treated melanoma patients. J Immunol. 1998;161:6970–6976.
Sgadari C, Angiolillo AL, Tosato G . Inhibition of angiogenesis by interleukin-12 is mediated by the interferon-inducible protein 10. Blood. 1996;87:3877–3882.
Rubin LL, Staddon JM . The cell biology of the blood–brain barrier. Annu Rev Neurosci. 1999;2:11–28.
Joly E, Mucke L, Oldstone M . Viral persistence in neurons explained by lack of major histocompatibility class I expression. Science. 1991;253:1283–1285.
Frei K, Fontana A . Antigen presentation in the CNS. Mol Psychol. 1997;2:96–98.
De Micco C . Immunology of central nervous system tumors. J Neuroimmunol. 1989;25:93–108.
De Micco C . Immunology of tumors of the central nervous system. Bull Cancer. 1989;76:17–31.
Saas P, Walker PR, Hahne M, et al. Fas ligand expression by astrocytoma in vivo: maintaining immune privilege in the brain? J Clin Invest. 1997;99:1173–1178.
Weller M, Fontana A . The failure of current immunotherapy for malignant glioma. Tumor-derived TGF-beta, T-cell apoptosis, and the immune privilege of the brain. Brain Res Rev. 1995;21:128–151.
Fontana A, Bodmer S, Frei K, et al. Expression of TGF-beta 2 in human glioblastoma: a role in resistance to immune rejection? Ciba Found Symp. 1991;157:232–241.
Weller M, Constam DB, Malipiero U, et al. Transforming growth factor-beta 2 induces apoptosis of murine T cell clones without down-regulating bcl-2 mRNA expression. Eur J Immunol. 1994;24:1293–1300.
Zou JP, Morford LA, Chougnet C, et al. Human glioma-induced immunosuppression involves soluble factor(s) that alters monocyte cytokine profile and surface markers. J Immunol. 1999;162:4882–4892.
Pazmany T, Kosa JP, Tomasi TB, et al. Effect of transforming growth factor-beta1 on microglial MHC-class II expression. J Neuroimmunol. 2000;103:122–130.
Aloisi F, De Simone R, Columba-Cabezas S, et al. Opposite effects of interferon-gamma and prostaglandin E2 on tumor necrosis factor and interleukin-10 production in microglia: a regulatory loop controlling microglia pro- and anti-inflammatory activities. J Neurosci Res. 1999;56:571–580.
Janabi N, Hau I, Tardieu M . Negative feedback between prostaglandin and alpha- and beta-chemokine synthesis in human microglial cells and astrocytes. J Immunol. 1999;162:1701–1706.
Hishii M, Nitta T, Ishida H, et al. Human glioma-derived interleukin-10 inhibits antitumor immune responses in vitro. Neurosurgery. 1995;37:1160–1166.
Knolle PA, Uhrig A, Hegenbarth S, et al. IL-10 down-regulates T cell activation by antigen-presenting liver sinusoidal endothelial cells through decreased antigen uptake via the mannose receptor and lowered surface expression of accessory molecules. Clin Exp Immunol. 1998;114:427–433.
Frei K, Lins H, Schwerdel C, et al. Antigen presentation in the central nervous system. The inhibitory effect of IL-10 on MHC class II expression and production of cytokines depends on the inducing signals and the type of cell analyzed. J Immunol. 1994;152:2720–2728.
Allegretta M, Albertini RJ, Howell MD, et al. Homologies between T cell receptor junctional sequences unique to multiple sclerosis and T cells mediating experimental allergic encephalomyelitis. J Clin Invest. 1994;94:105–109.
Soderstrom M, Link H, Sun JB, et al. Autoimmune T cell repertoire in optic neuritis and multiple sclerosis: T cells recognizing multiple myelin proteins are accumulated in cerebrospinal fluid. J Neurol Neurosurg Psychiatry. 1994;55:544–551.
Kalman B, Knobler RL, Lublin FD . Preferential but not exclusive T cell receptor V beta chain utilization of myelin basic protein and peptide-specific T cell clones in mice. Cell Immunol. 1994;153:206–213.
Dranoff G, Jaffee E, Lazenby A, et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte–macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA. 1993;90:3539–3543.
Wakimoto H, Yoshida Y, Aoyagi M, et al. Efficient retrovirus-mediated cytokine-gene transduction of primary-cultured human glioma cells for tumor vaccination therapy. J Cancer Res. 1997;88:296–305.
Acknowledgements
We would like to thank Ms W Giles-Davis for technical assistance and Ms Cole for preparation of the manuscript. This work was supported by a grant from the Katie's Kids Brain Tumor Foundation.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
O, I., Blaszczyk-Thurin, M., Shen, C. et al. A DNA vaccine expressing tyrosinase-related protein-2 induces T-cell-mediated protection against mouse glioblastoma. Cancer Gene Ther 10, 678–688 (2003). https://doi.org/10.1038/sj.cgt.7700620
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7700620
- Springer Nature America, Inc.
Keywords
This article is cited by
-
Surface display of glycosylated Tyrosinase related protein-2 (TRP-2) tumour antigen on Lactococcus lactis
BMC Biotechnology (2015)
-
Incomplete tumour control following DNA vaccination against rat gliomas expressing a model antigen
Acta Neurochirurgica (2013)
-
Downregulation of KIF23 suppresses glioma proliferation
Journal of Neuro-Oncology (2012)
-
In vivo bioluminescence imaging in an experimental mouse model for dendritic cell based immunotherapy against malignant glioma
Journal of Neuro-Oncology (2009)
-
DNA vaccines for cancer too
Cancer Immunology, Immunotherapy (2006)